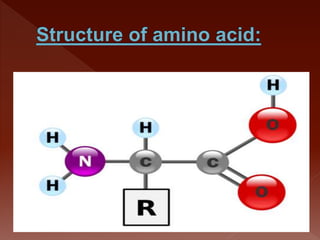
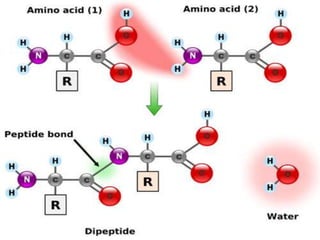
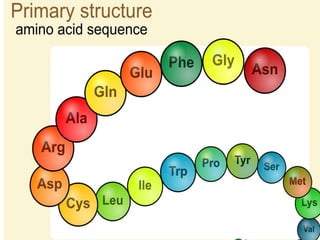
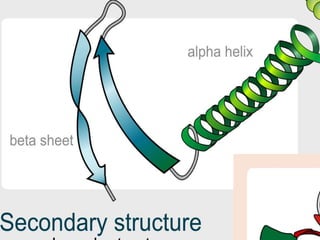
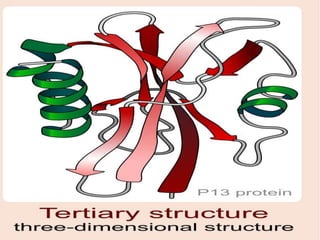
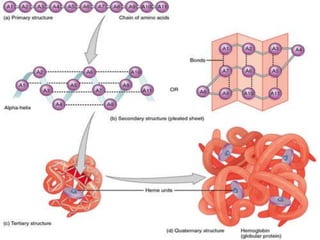
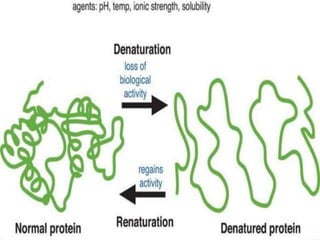
The document summarizes a biochemistry group project on proteins. The group, led by Mohammad Raes, discussed the definition, chemistry, structure, classification, properties, and functions of proteins. Key points included that proteins are polymers of amino acids and have four levels of structure - primary, secondary, tertiary, and quaternary. Proteins perform important roles such as enzymes, hormones, antibodies, and structure.