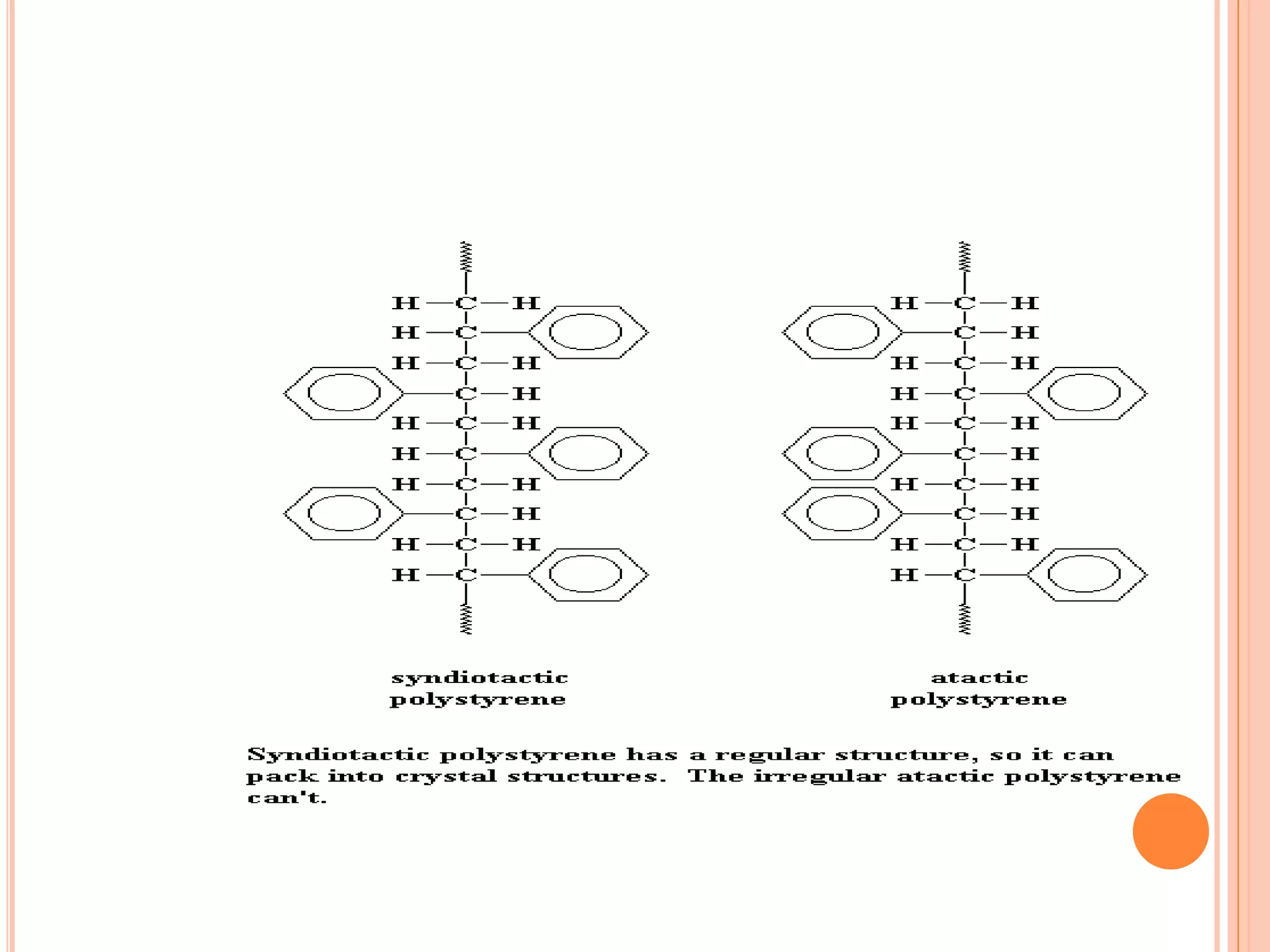
Group members Rahamat Ullah, MD. Ashraful Amin, and Neamul Haque from Daffodil International University presented on crystallinity in polymers. They discussed that crystallinity refers to the structural order of atoms or molecules in a solid. There are two types of polymers: crystalline polymers, which are rigid and high melting, and amorphous polymers. The regular structure of polymer molecules allows them to form lamellar crystals. A polymer's crystallinity depends on its structure - orderly polymers easily form crystals while irregular polymers are amorphous. The degree of crystallinity influences various material properties such as mechanical strength, opacity, and thermal behavior.