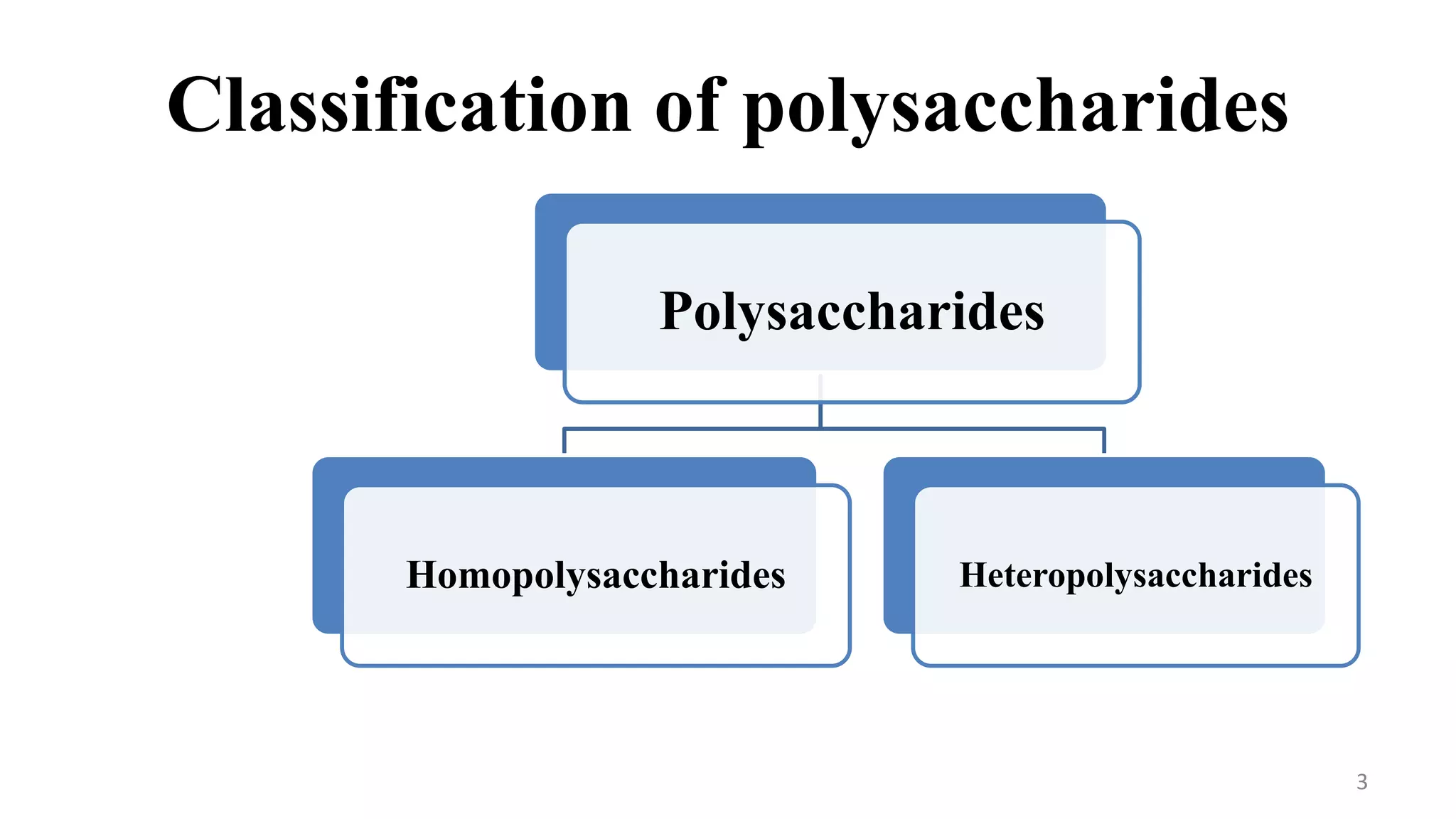
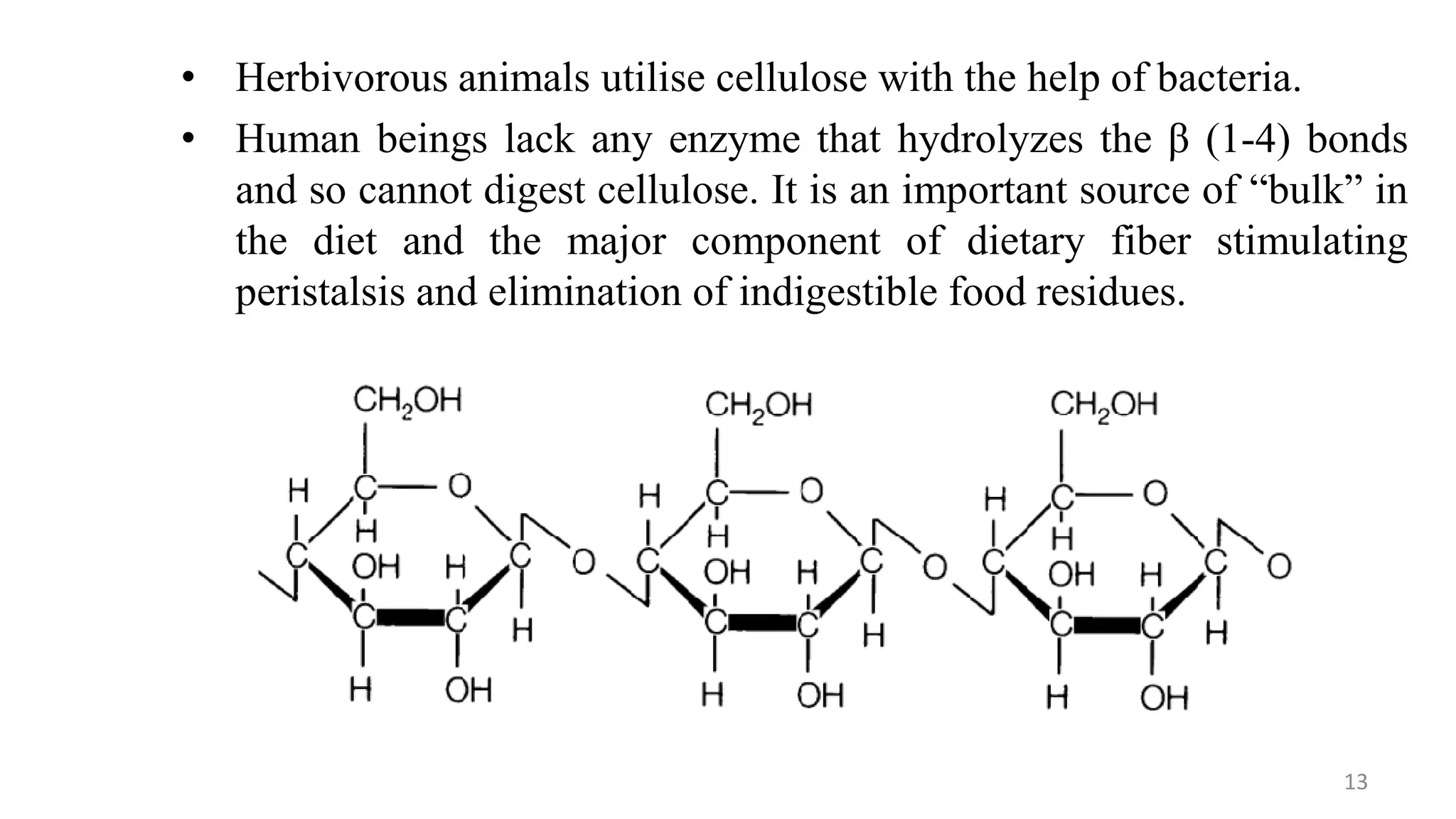
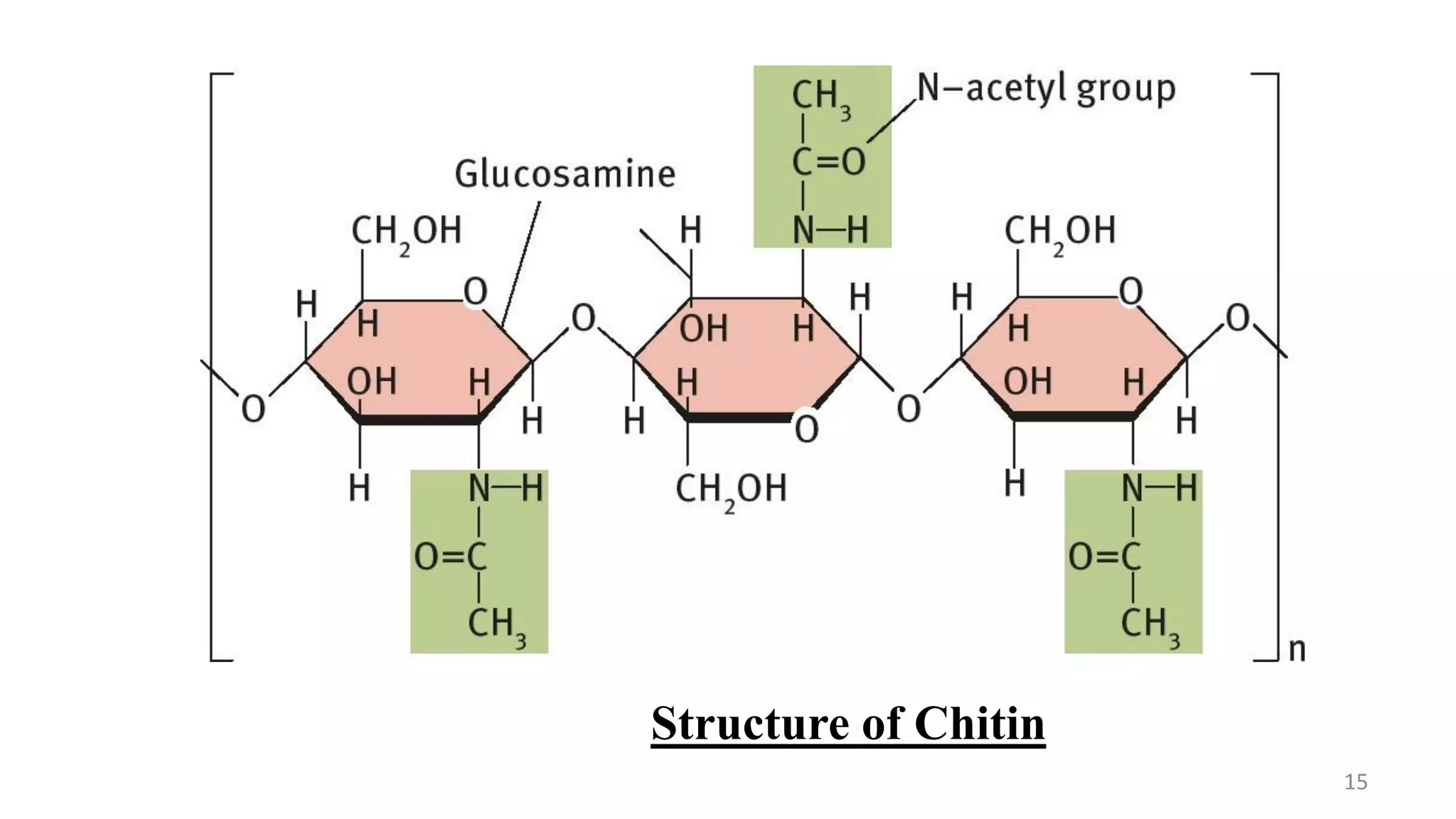
This document provides an overview of polysaccharides, including their definitions, classifications into homopolysaccharides and heteropolysaccharides, and specific examples like starch, glycogen, cellulose, chitin, and various glycoproteins. It details their structures, functions, and applications in biological systems, highlighting the roles of sugars in energy storage, cell structure, and communication. Additionally, it discusses the mechanisms of glycosylation and the significance of glycans in protein function and regulation.








































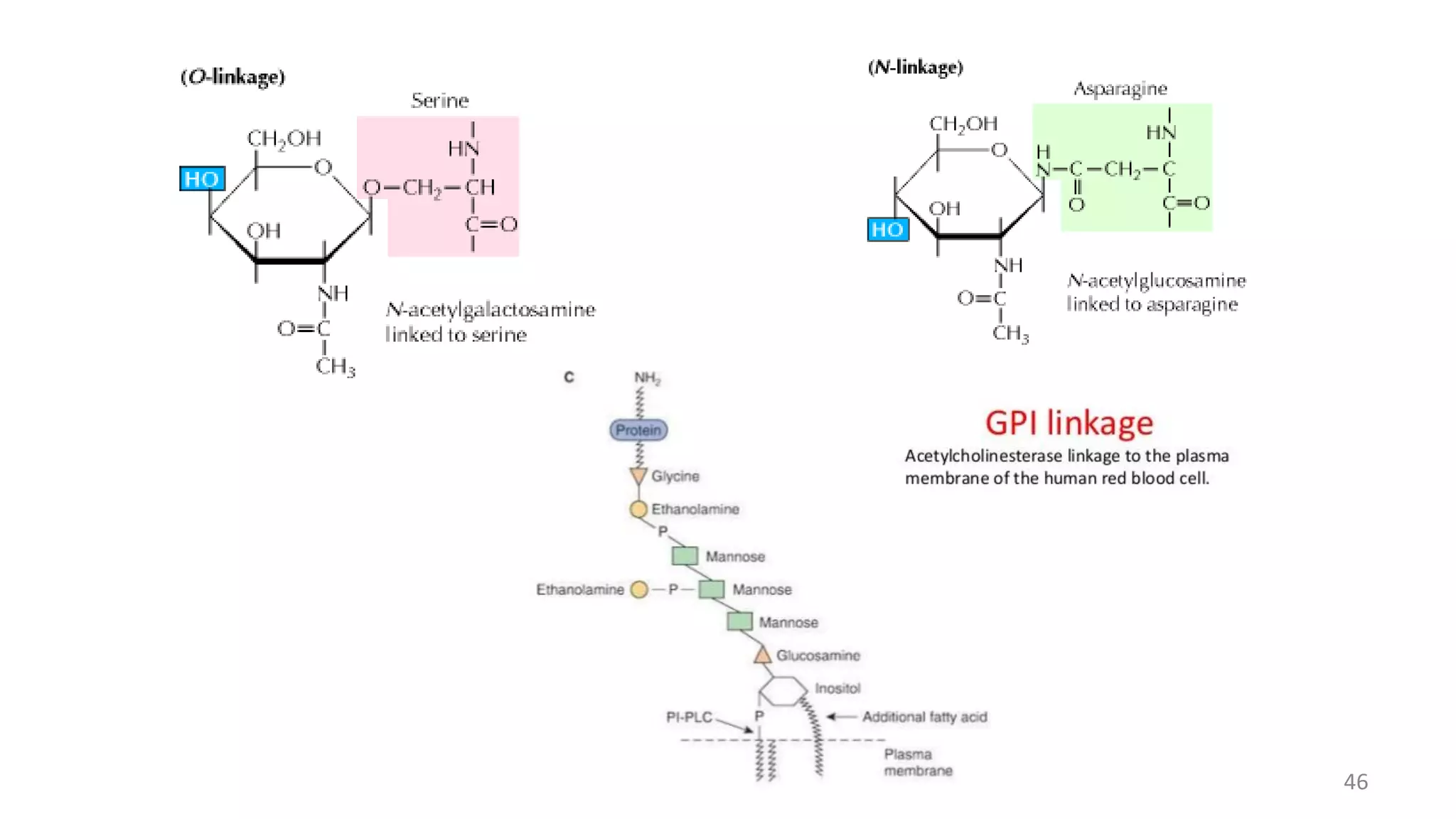
![2. O – linked glycans
Hydroxyl side chain of serine or threonine and a sugar such as N –
acetylgalactosamine (GalNAc – Ser[Thr])
Anomeric carbon of NAG – attached to O of serine or threonine.
Mucins which are found extensively in salivary secretions, contain many
short O – linked glycans.
Increase the viscosity of the fluids in which they are dissolved.
3. GPI – anchored or GPI – linked (Glycosylphosphatidylinositol –
anchored)
Carboxyl terminal amino acid of a protein via a phosphoryl – ethanolamine
moiety joined to an oligosaccharide (glycan), which in turn is linked via
glucosamine to phosphatidylinositol.
The GPI anchor may allow greatly enhanced mobility of a protein in the
plasma membrane.
Some GPI anchors may connect with signal transduction pathways.
Some examples include Acetylcholinesterase, Alkaline phosphatase
45](https://image.slidesharecdn.com/polysaccharides-201210152630/75/Polysaccharides-Biochemistry-for-Msc-Students-45-2048.jpg)