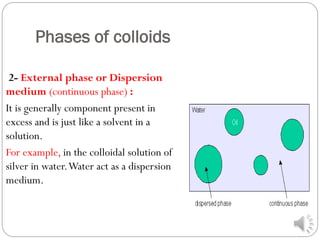
A colloid solution is a heterogeneous mixture whose dispersed particles are larger than molecules but smaller than what can be seen with the naked eye, ranging from 1-1000 nm. Colloids exhibit unique optical properties like the Tyndall effect where a beam of light is scattered when passing through the colloidal solution. Colloids can be classified as hydrophilic or hydrophobic depending on whether the particles are attracted to or repelled by water. Common examples are emulsions like milk or gels.