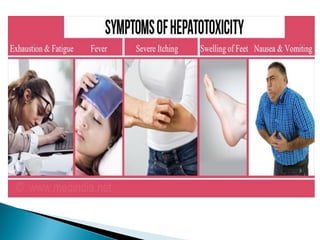
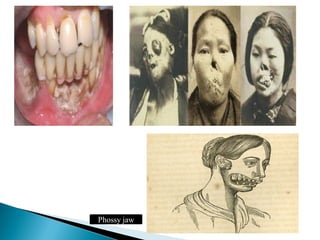
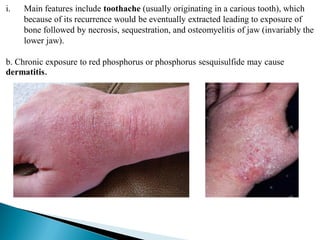
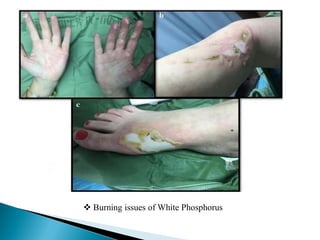
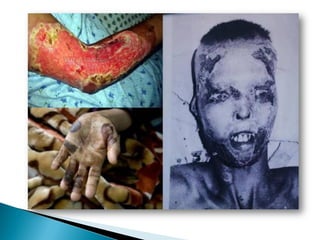
The document discusses the toxicology of phosphorus, highlighting its different types (yellow, red, and black), sources, and applications, particularly in industries and agriculture. It explains the symptoms and modes of poisoning, diagnostic procedures, and treatment protocols for acute and chronic exposures. Additionally, it reviews the regulatory framework surrounding industrial phosphorus use and the forensic implications of phosphorus poisoning.