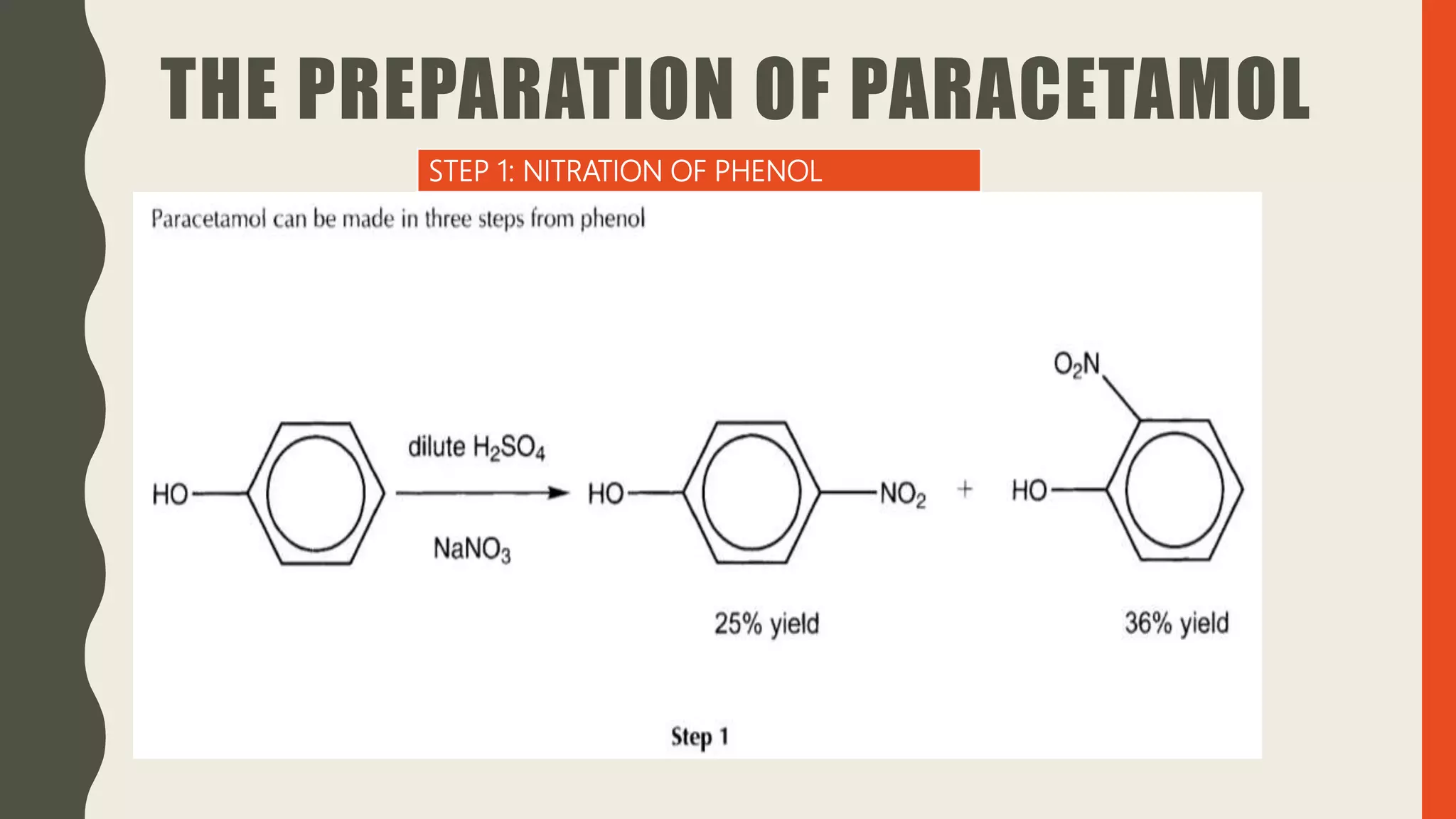
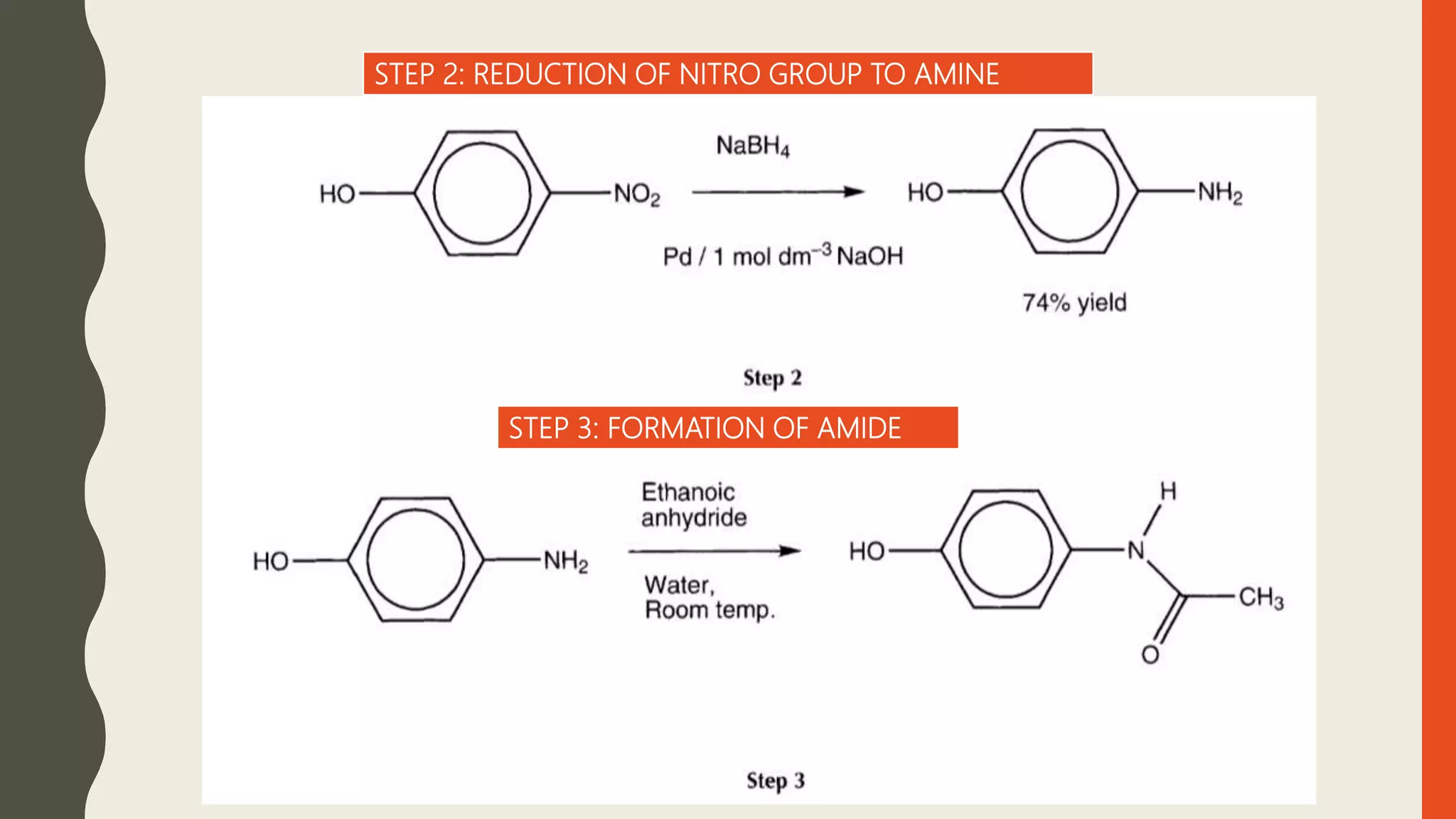
Paracetamol is a widely used analgesic and antipyretic medication, available in over 90 formulations and generally safe at recommended doses, though high doses pose significant toxicity risks. Initially derived from acetanilide in the 1880s, its development progressed through various derivatives, with paracetamol marketed in the 1950s as a safer alternative to aspirin for children. It is effective for treating fever and mild to moderate pain but has potential interactions and side effects, including liver damage from overdose.