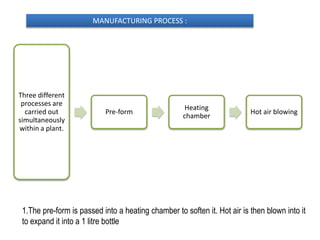
The document provides information on packaged drinking water, including its definition, types, manufacturing process, and quality evaluation. It discusses the major types of bottled water such as mineral water, spring water, and purified water. The manufacturing process involves treating raw water through various purification methods like reverse osmosis and ozonization before filling into bottles or jars. Quality is evaluated using various physical, chemical, and microbiological tests to ensure the water meets safety standards.