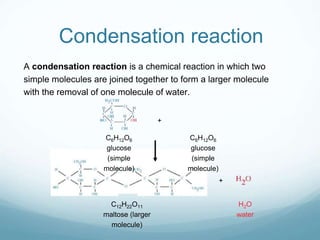
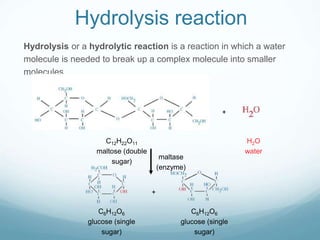
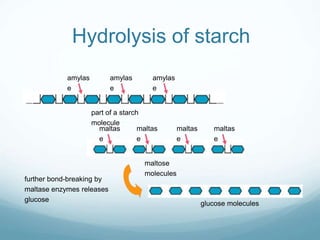
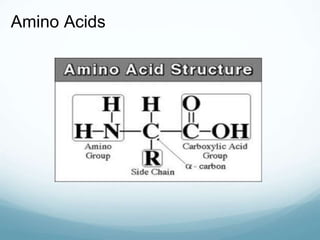
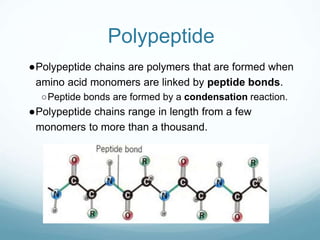
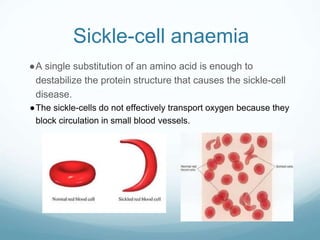
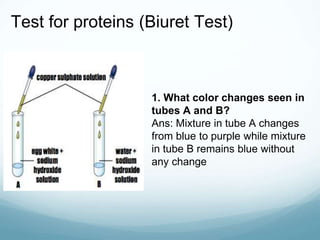
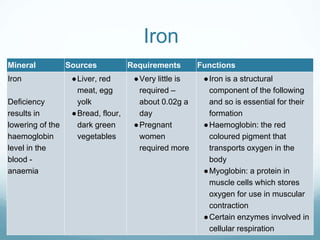
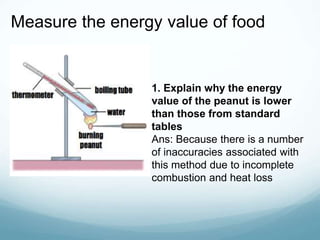
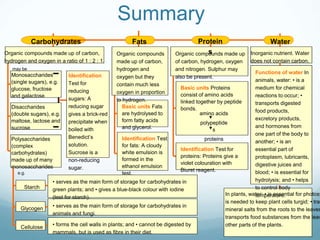
Organisms require nutrients from food for energy, growth, repair, and health. There are six main types of nutrients: carbohydrates, proteins, lipids, vitamins, minerals, and water. Carbohydrates, proteins, and lipids provide energy and are made up of smaller units called monosaccharides, amino acids, and fatty acids, respectively. These macronutrients are broken down into their smaller subunits before the body can use them. In contrast, water and minerals can be absorbed directly.