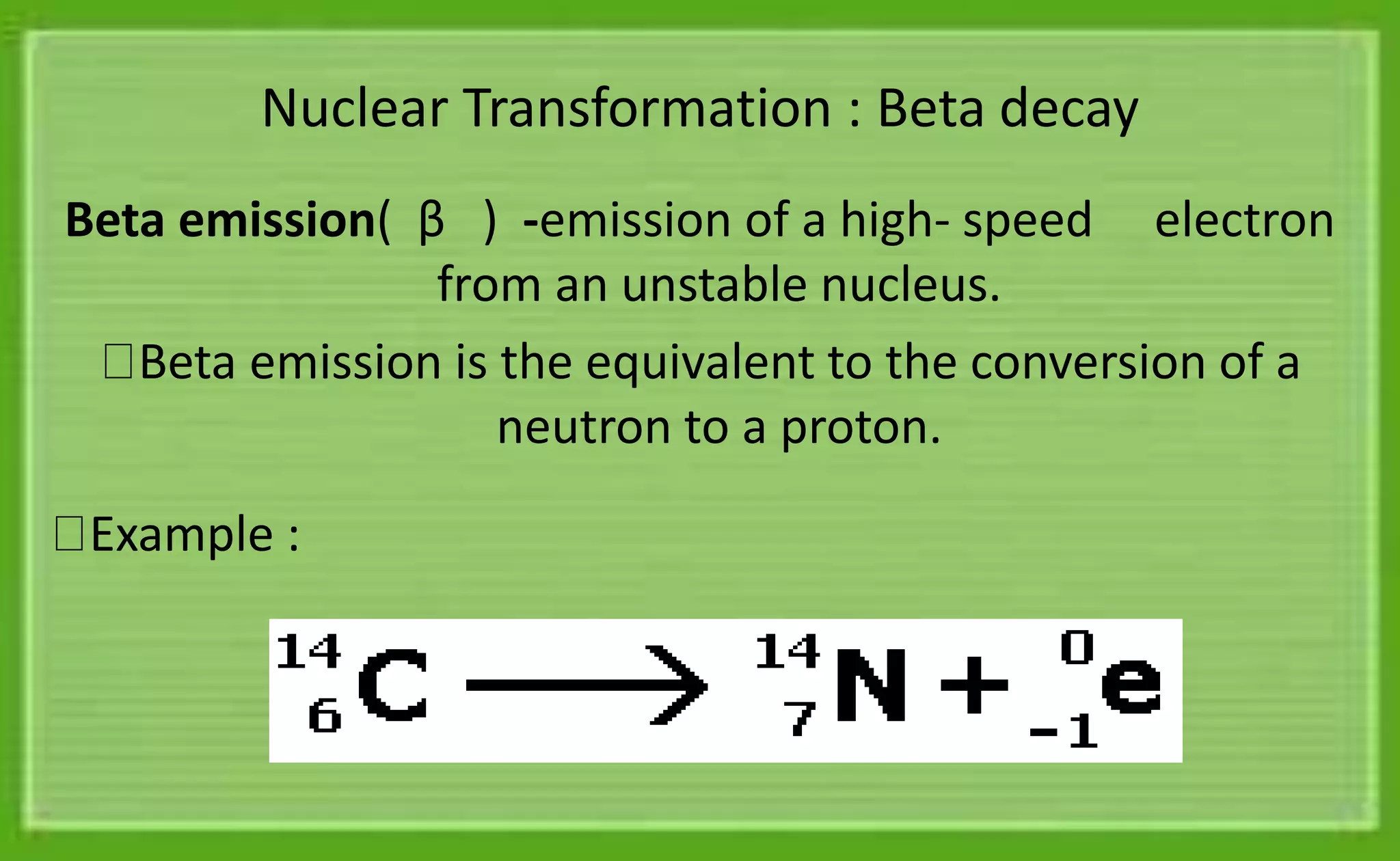
The document discusses different types of radioactive decay:
- Radioactive decay occurs when an unstable atomic nucleus transforms spontaneously through emission of radiation. If the daughter nucleus is stable, decay ends, but it can continue with further unstable daughters until stability.
- Rutherford discovered two types of radiation, alpha and beta rays, emitted from uranium that differed from X-rays in penetrating power.
- There are five main types of radioactive decay: alpha, beta-minus, beta-plus, electron capture, and isomeric transition. Alpha decay involves emitting a helium nucleus. Beta decay involves converting a neutron to a proton with electron emission. Gamma emission does not change proton or neutron number but involves releasing energy from an excited