Radioactivity
•Download as PPTX, PDF•
0 likes•12 views
This presentation is presented by a physics Scholar Ayat Ullah Department of Physics University: Kohat University of science and Technology Kohat
Report
Share
Report
Share
Recommended
Recommended
More Related Content
What's hot
What's hot (20)
Radioactivity ( Tajuk : Astronomi & Fizik Moden_Tugasan Kumpulan Sem 1_UTHM)

Radioactivity ( Tajuk : Astronomi & Fizik Moden_Tugasan Kumpulan Sem 1_UTHM)
Similar to Radioactivity
Similar to Radioactivity (20)
Chapter 21 Nuclear Chemistry Section 2 1Updated (1).pptx

Chapter 21 Nuclear Chemistry Section 2 1Updated (1).pptx
Recently uploaded
Recently uploaded (20)
Hire 💕 9907093804 Hooghly Call Girls Service Call Girls Agency

Hire 💕 9907093804 Hooghly Call Girls Service Call Girls Agency
9654467111 Call Girls In Raj Nagar Delhi Short 1500 Night 6000

9654467111 Call Girls In Raj Nagar Delhi Short 1500 Night 6000
GUIDELINES ON SIMILAR BIOLOGICS Regulatory Requirements for Marketing Authori...

GUIDELINES ON SIMILAR BIOLOGICS Regulatory Requirements for Marketing Authori...
VIRUSES structure and classification ppt by Dr.Prince C P

VIRUSES structure and classification ppt by Dr.Prince C P
Discovery of an Accretion Streamer and a Slow Wide-angle Outflow around FUOri...

Discovery of an Accretion Streamer and a Slow Wide-angle Outflow around FUOri...
Botany krishna series 2nd semester Only Mcq type questions

Botany krishna series 2nd semester Only Mcq type questions
Asymmetry in the atmosphere of the ultra-hot Jupiter WASP-76 b

Asymmetry in the atmosphere of the ultra-hot Jupiter WASP-76 b
Disentangling the origin of chemical differences using GHOST

Disentangling the origin of chemical differences using GHOST
Kochi ❤CALL GIRL 84099*07087 ❤CALL GIRLS IN Kochi ESCORT SERVICE❤CALL GIRL

Kochi ❤CALL GIRL 84099*07087 ❤CALL GIRLS IN Kochi ESCORT SERVICE❤CALL GIRL
Biopesticide (2).pptx .This slides helps to know the different types of biop...

Biopesticide (2).pptx .This slides helps to know the different types of biop...
Radioactivity
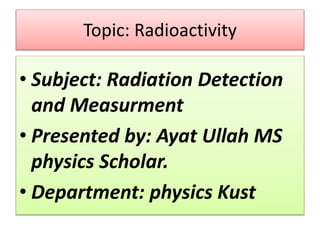
- 1. Topic: Radioactivity • Subject: Radiation Detection and Measurment • Presented by: Ayat Ullah MS physics Scholar. • Department: physics Kust
- 2. RADIOACTIVITY: “The phenomena of spontaneous disintegration of unstable nuclei followed by emission of nuclear radiation is called as Radioactivity” “OR” The spontaneous release of subatomic particles or gamma rays by unstable atoms as their nuclei tend to break apart into other particles to attain stability is called radioactivity. An element which possesses such property is called radioactive element.
- 3. Alpha Particle: • Alpha particle consists of two proton and two neutron i.e., these are positively charged helium nuclei. An alpha particle is emitted by very a large unstable nucleus. • It is easily stopped by cardboard or thin metal. • Has a range in air of no more than a few centimeter. • Ionizes air molecules much more strongly than the other two types of radioactive radiation.
- 4. Emission of Alpha Particle: • When the ratio of neutron to proton in the nucleus is too low, certain atoms restore the balance by emitting alpha particles. • When an atom disintegrates by alpha emission, its atomic number is reduces by 2 and mass number is reduces by 4 units. The disintegration reaction is written by. • ZXA Z-2YA-4 + 2He4 +Q
- 5. Examples of Alpha decay: + Q + Q
- 6. Beta Particle: • Beta particle consists of electrons, each emitted when a nucleus with too many neutron disintegrates. A neutron in such a nucleus suddenly and unexpectedly changes to a proton; in the process, an electron is created and instantly emitted from the nucleus. Beta particles. • Is stopped by 5-10 mm of metal. • Has a range in air of about 1m. • Ionizes air molecules less strongly than alpha- particles.
- 7. Emission of Beta: • Beta decay doesn’t change the mass number. In essence, beta decay changes a neutron into a proton and an electron. The proton stays in the nucleus and the electron is ejected energetically. • The disintegration reaction is written by • ZXA Z+1YA + -1β0 +Q
- 8. Examples of Beta decay: + Q 6C14 7N14 + -1β0 + Q
- 9. Gamma Emission: • Most frequently the alpha or beta emission leaves the daughter nuclide in an exited state. Such a nuclide may go back to a more stable configuration and eventually to its ground state by emitting one or more γ-rays. Since γ-rays are massless photon, their emission will cause no change either in A or Z of the parent nuclide. The γ-decay process is written as follows. • ZXA (ZXA)* ZXA + γ
