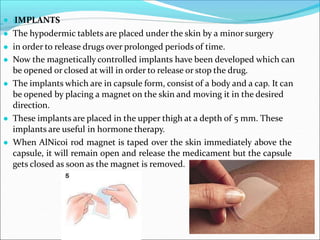
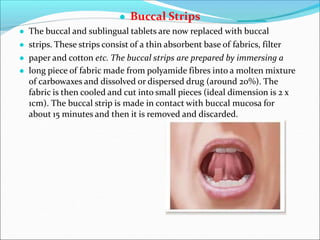
Novel drug delivery systems refer to approaches for transporting pharmaceutical compounds in the body to achieve therapeutic effects safely. They are classified as implants, films/strips, liposomes, controlled delivery modules, erythrocytes, nanoparticles, and prodrugs. Implants are placed under the skin to release drugs over time. Films and strips are meant for topical application of slow drug release. Liposomes can transport both hydrophilic and hydrophobic drugs. Controlled delivery modules release drugs slowly from a polymeric matrix. Erythrocytes allow drugs to circulate in the body for prolonged release. Nanoparticles are colloidal systems that deposit drugs at target sites. Prodrugs undergo biotransformation before showing pharmacological activity