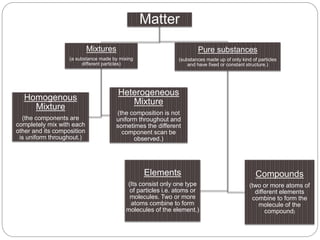
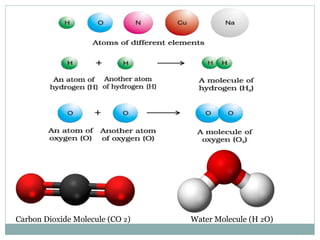
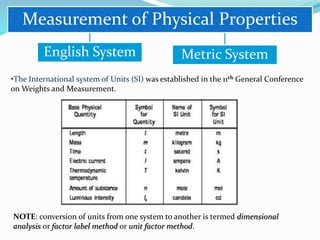
Chemistry is the study of atoms and molecules and their interactions and transformations. It plays an important role in meeting human needs through products like medicines, foods, and other materials. Chemistry concepts include the three states of matter, mixtures and pure substances, chemical and physical properties, and measurement systems. Key ideas in chemistry are the laws of conservation of mass, definite proportions, and multiple proportions. Chemistry also involves the concepts of atoms, molecules, ions, and chemical reactions.