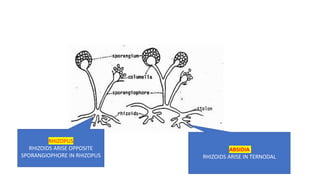
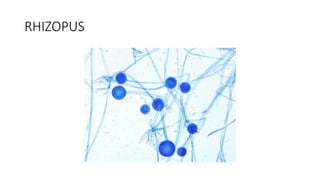
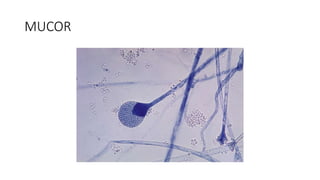
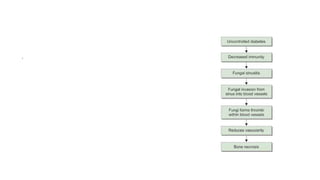
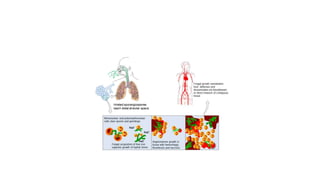
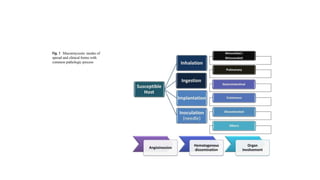
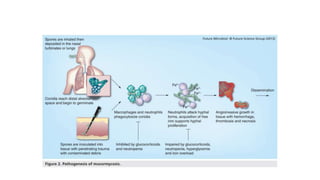
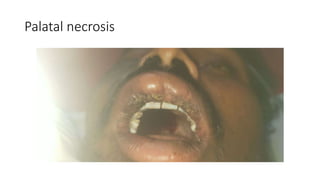
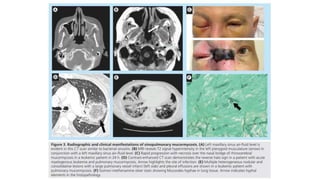
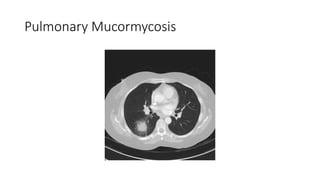
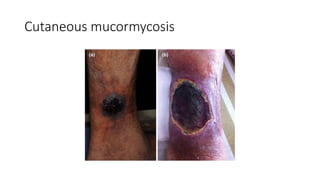
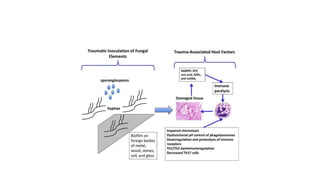
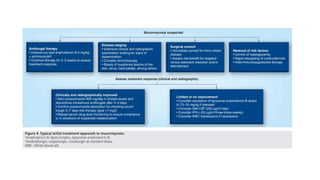
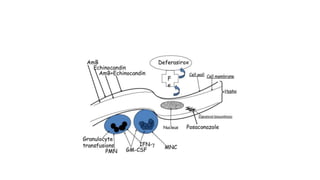
Mucormycosis is a rare but aggressive fungal infection caused by fungi of the class Zygomycetes, including Rhizopus, Mucor, and Absidia. It mainly affects immunocompromised patients or those with uncontrolled diabetes. The fungi thrive in high glucose, acidic conditions. Common forms include rhinocerebral affecting the sinuses and orbit, pulmonary, gastrointestinal, cutaneous from skin injuries, and disseminated infection of multiple organs. Diagnosis involves tissue biopsy and culture. Treatment requires intravenous amphotericin B antifungal therapy and surgical debridement of infected tissues.