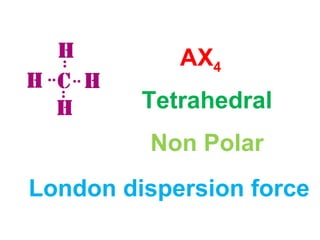
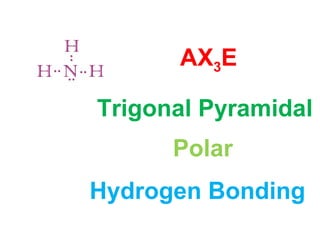
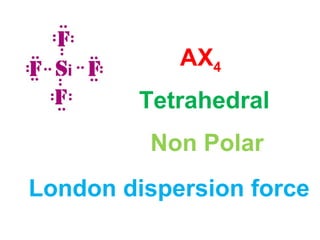
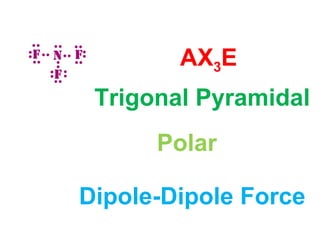
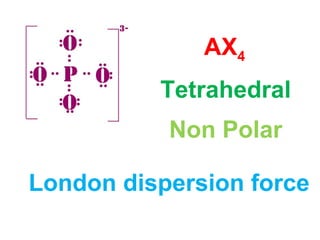
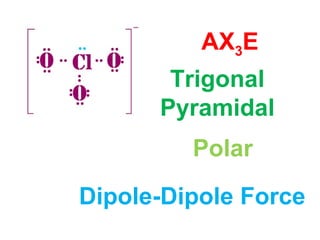
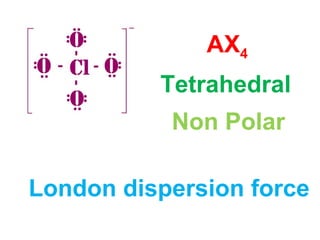
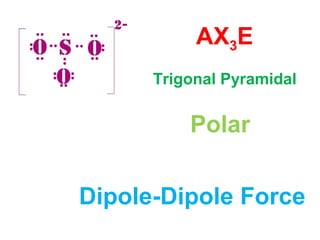
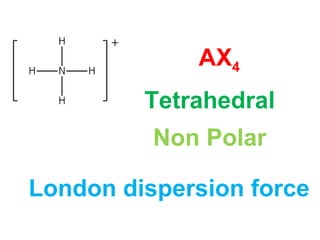
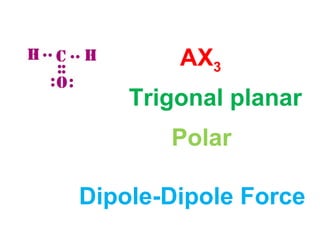
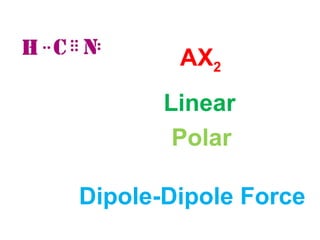
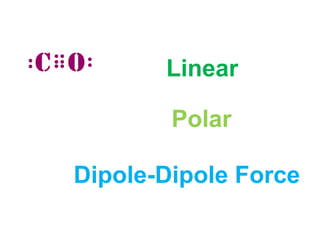
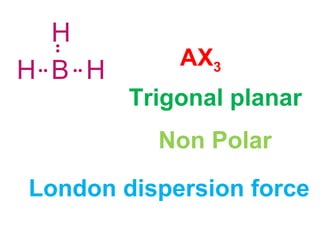
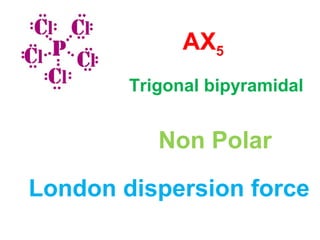
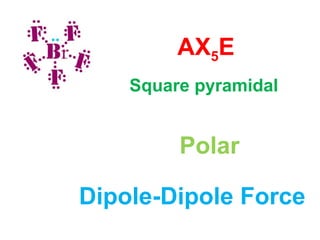
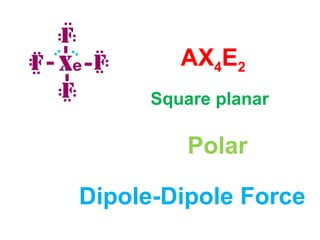
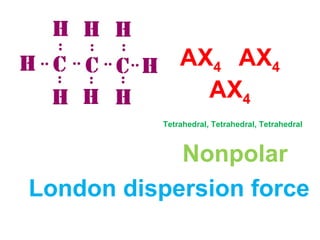
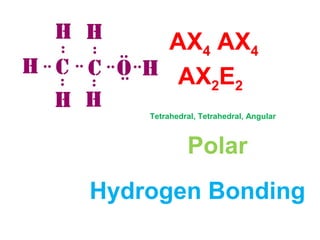
The document describes various Lewis dot structures for different molecular geometries, noting their polarity and type of intermolecular forces. It discusses structures such as tetrahedral (AX4), trigonal pyramidal (AX3E), angular (AX2E2), trigonal planar (AX3), and linear (AX2) geometries. It notes whether each structure is polar or nonpolar and indicates the dominant intermolecular force as either London dispersion, hydrogen bonding, or dipole-dipole interactions. It also covers exceptions and more complex structures formed from combinations of simpler structures.