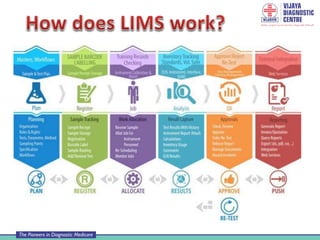
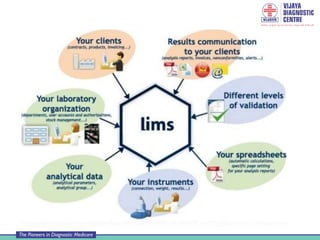
A Laboratory Information Management System (LIMS) is a software solution used in laboratories to manage large amounts of sample data and laboratory workflow. It typically handles five key phases: sample receipt and login, assignment and tracking, processing and quality control, data storage, and result reporting. A LIMS provides advantages like faster access to reports, reduced errors, improved efficiency and productivity. However, it also requires training, time for adaptation, costs for purchase and maintenance, and backup systems in case of data loss. Laboratories must ensure proper management, security and validation of any LIMS.