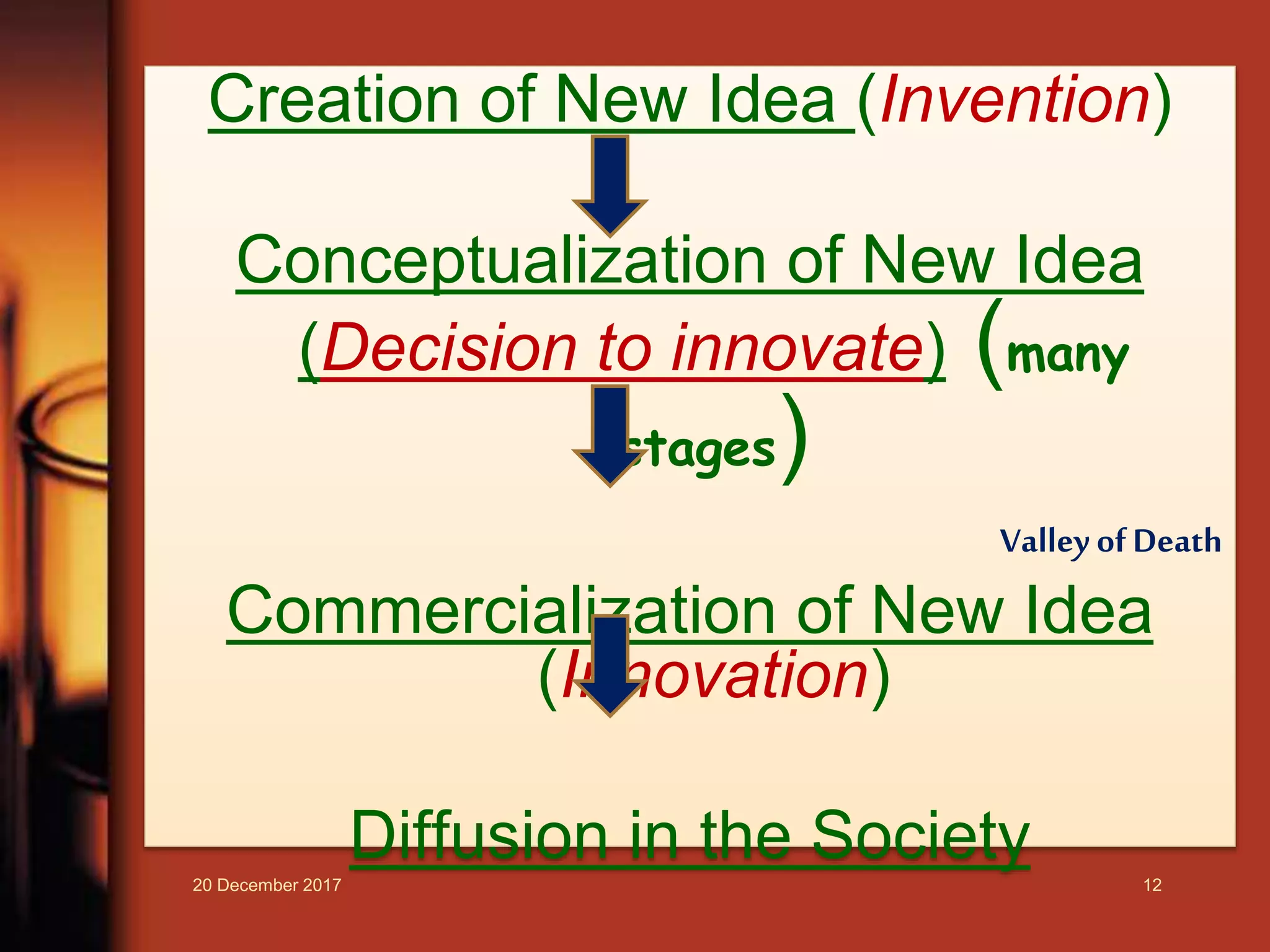
1. The document discusses various topics related to intellectual property and life science innovations, including what constitutes creativity, principles of creative diversity, the difference between discovery and invention, and the importance of intellectual property rights (IPRs) in protecting innovations.
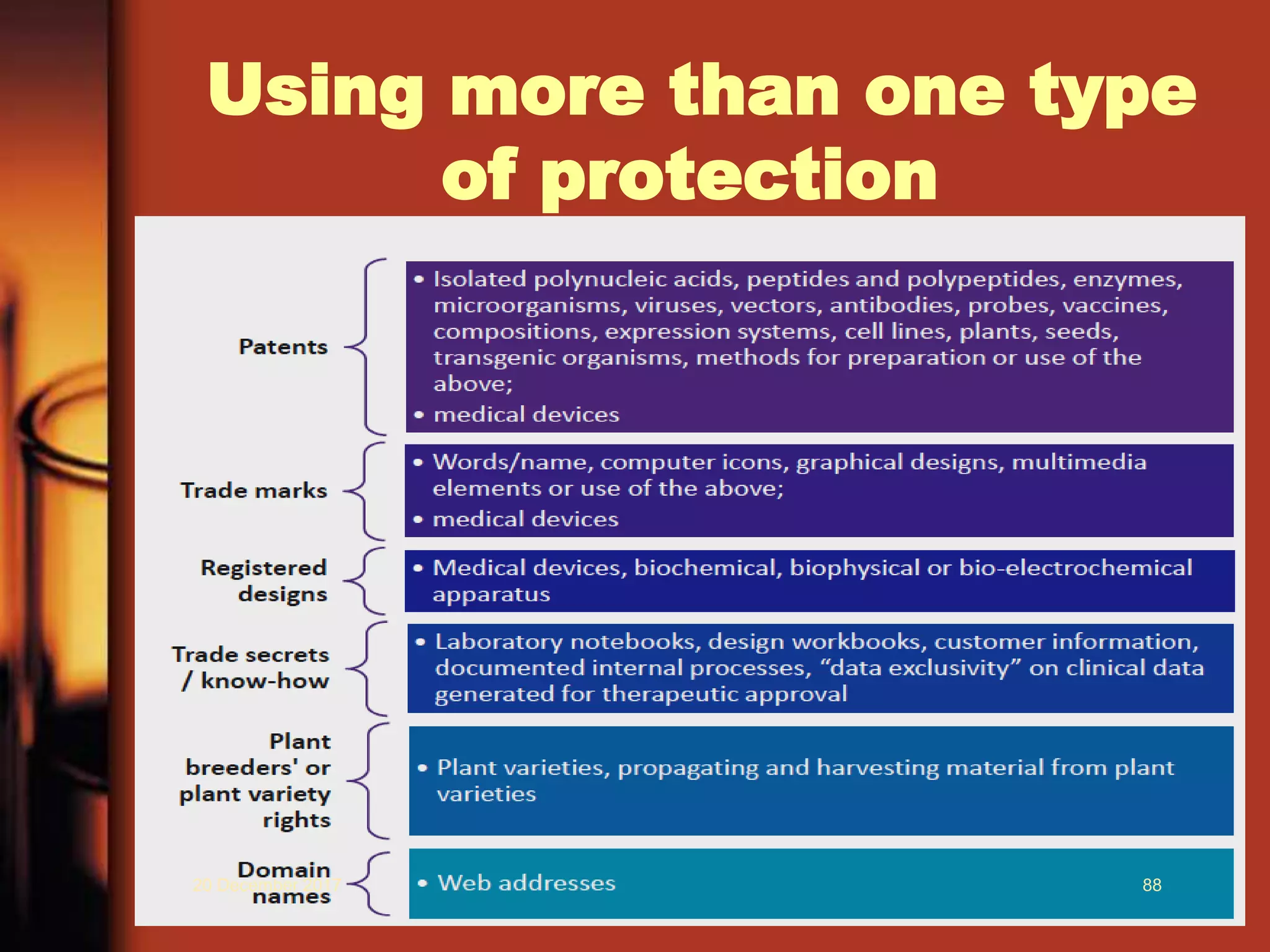
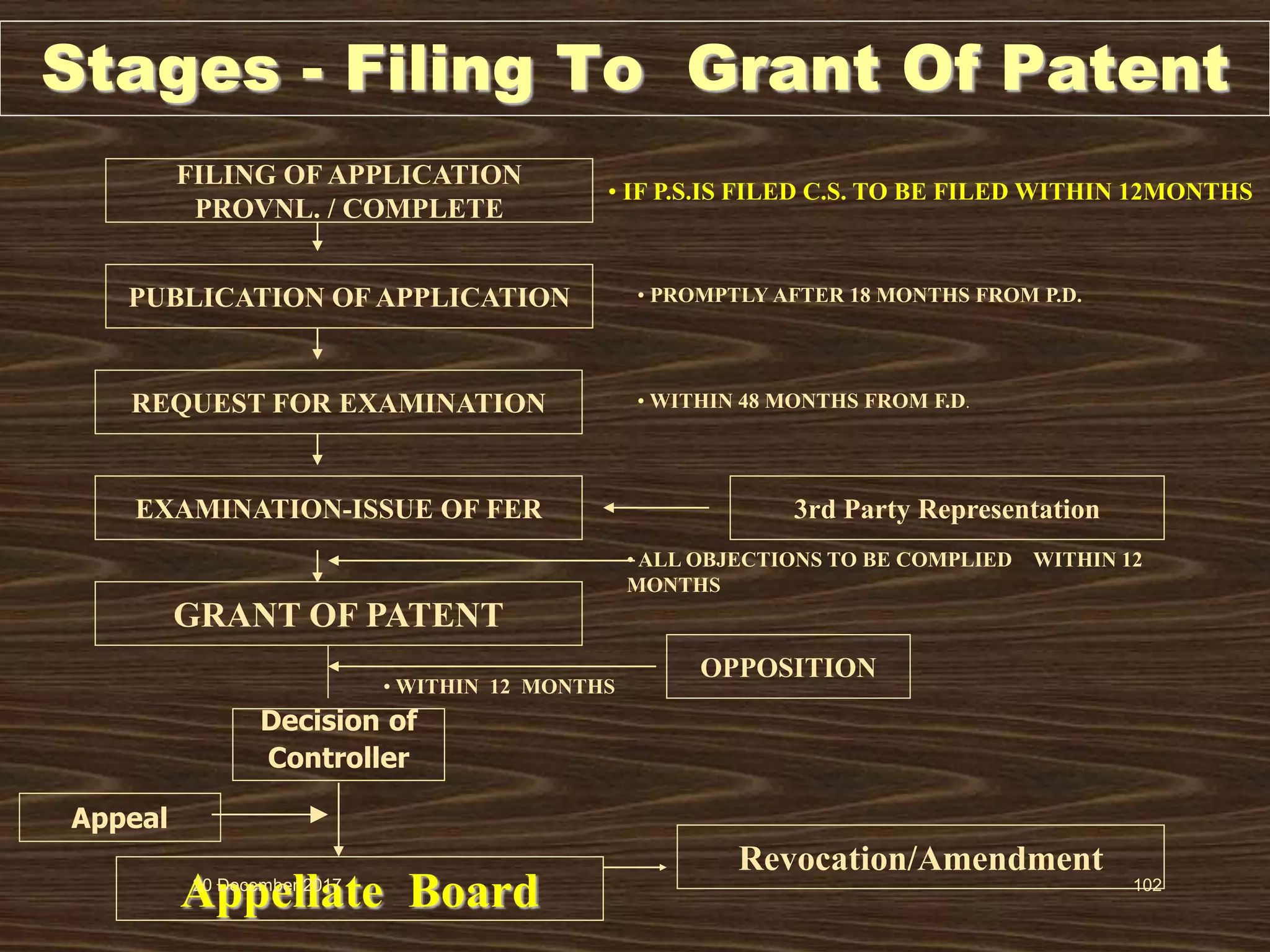
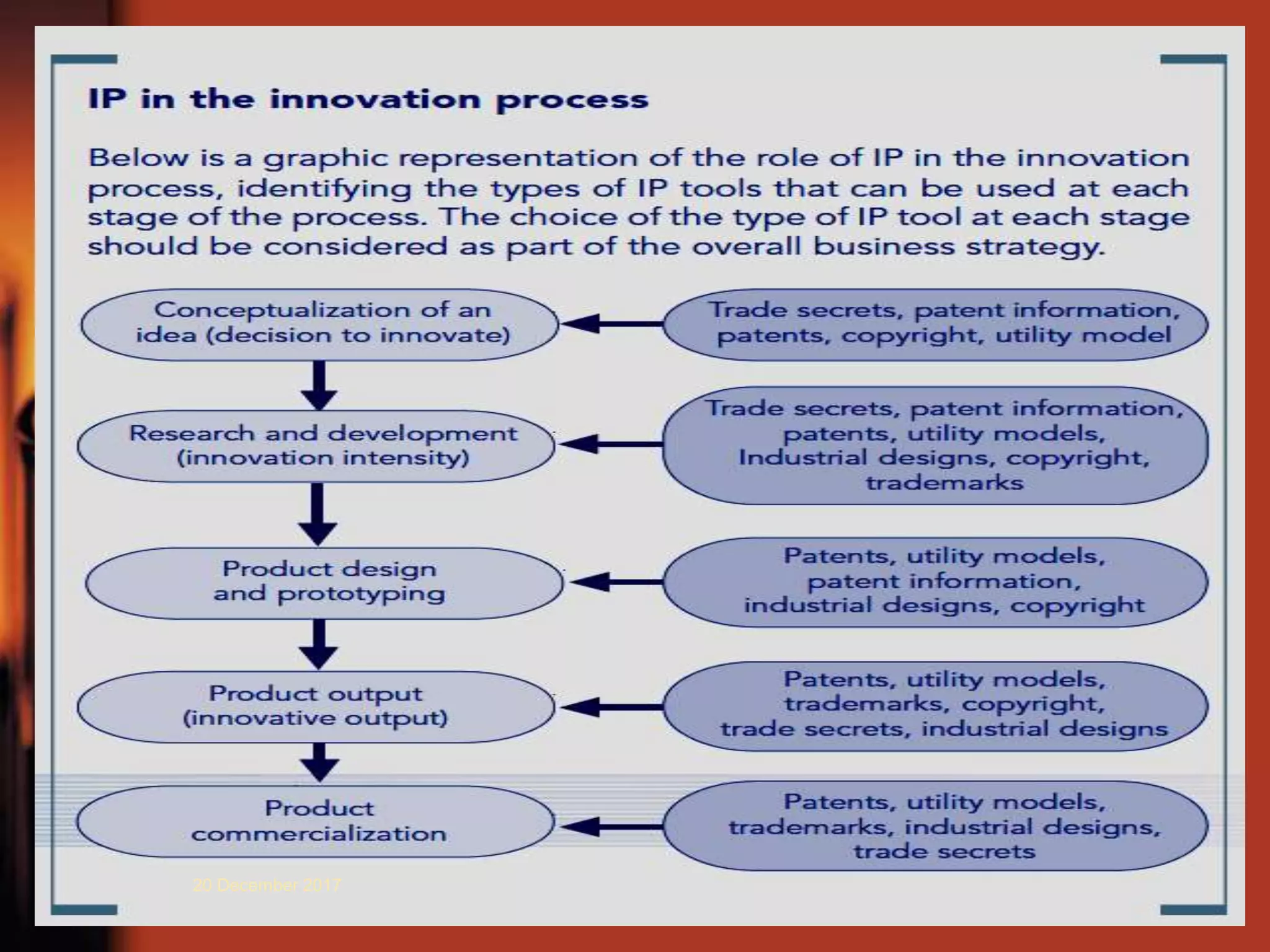
2. It provides an overview of different types of IPRs including patents, trademarks, designs, geographical indications, and copyrights. Key requirements for patentability like novelty, inventive step, and industrial application are also explained.
3. Examples of important life science innovations that have been patented are provided, such as aspirin, streptomycin, and polymerase chain reaction. Landmark patents including the first patent granted for a microorganism and the first patent for a genetically





























































![Biodiversity Act, 2002 &
Biological Resources
• “Biological Resources”
means plants, animals
and microorganisms or
parts thereof, their
genetic material and by-
products with actual or
potential use or value
does not include human
genetic material [Chapter
I Clause 2(b) and 2(c)].
20 December 2017 64](https://image.slidesharecdn.com/iplifescinnovations-171220163219/75/Intellectual-property-Life-Science-Innovations-64-2048.jpg)