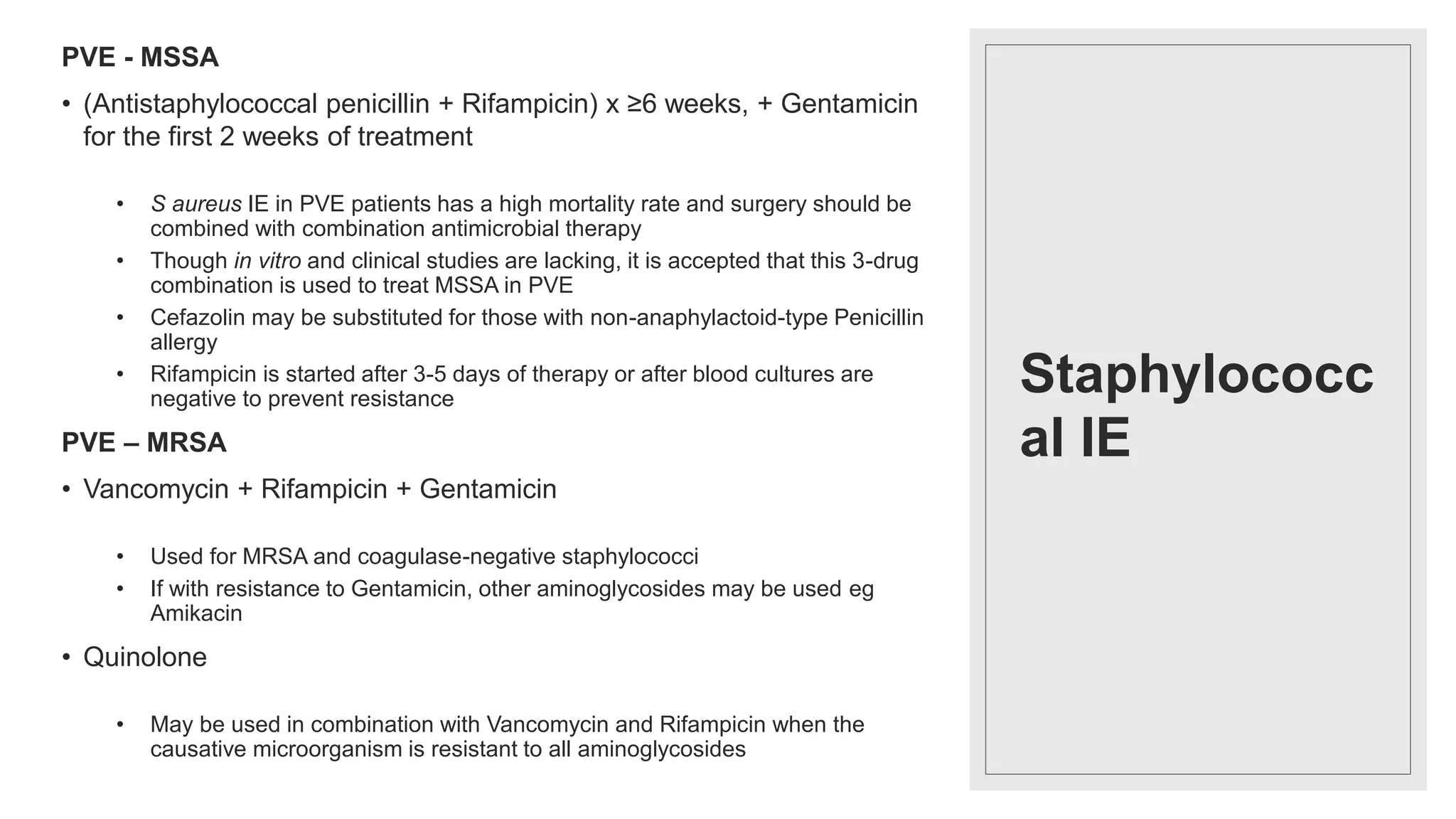
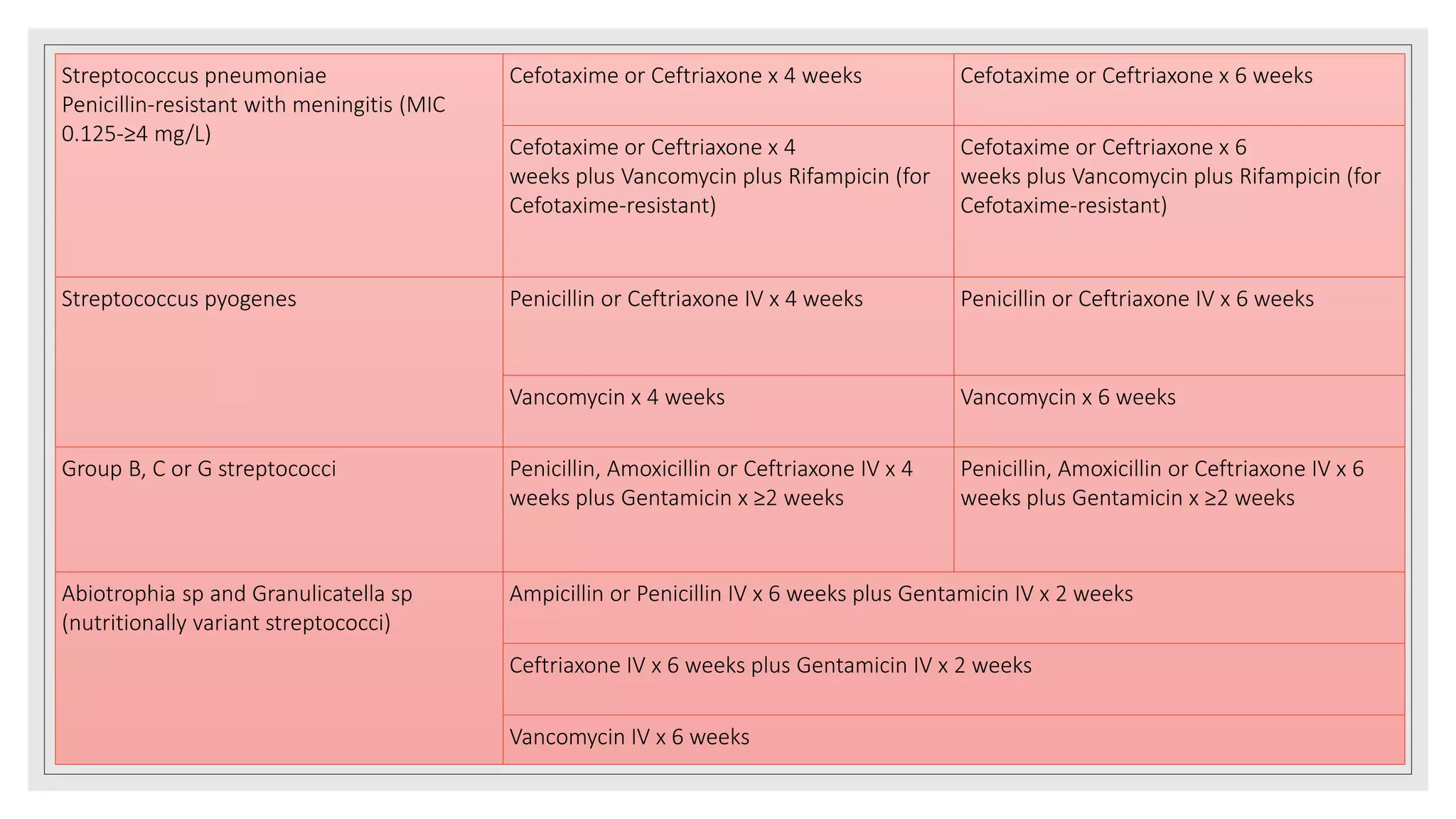
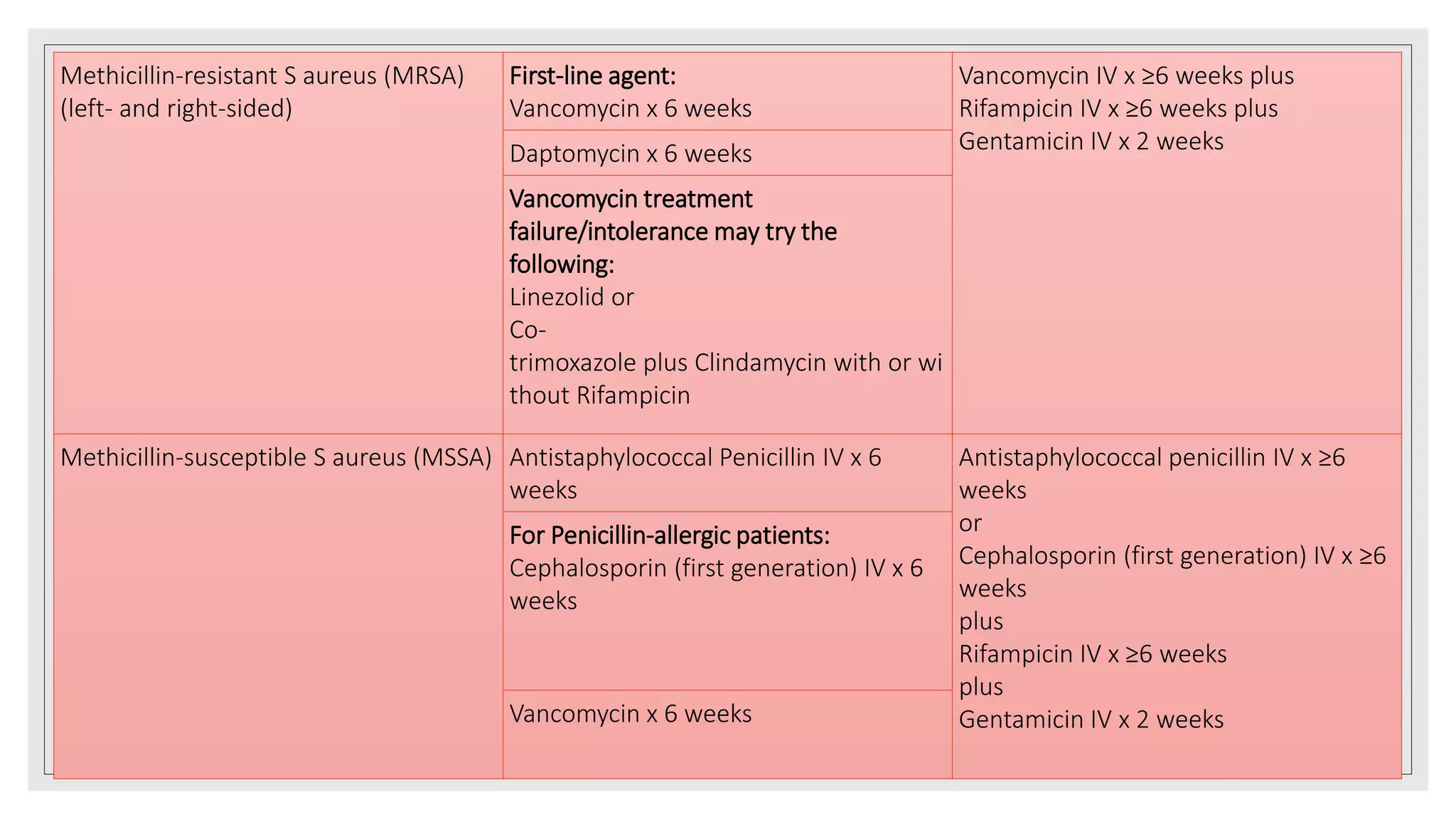
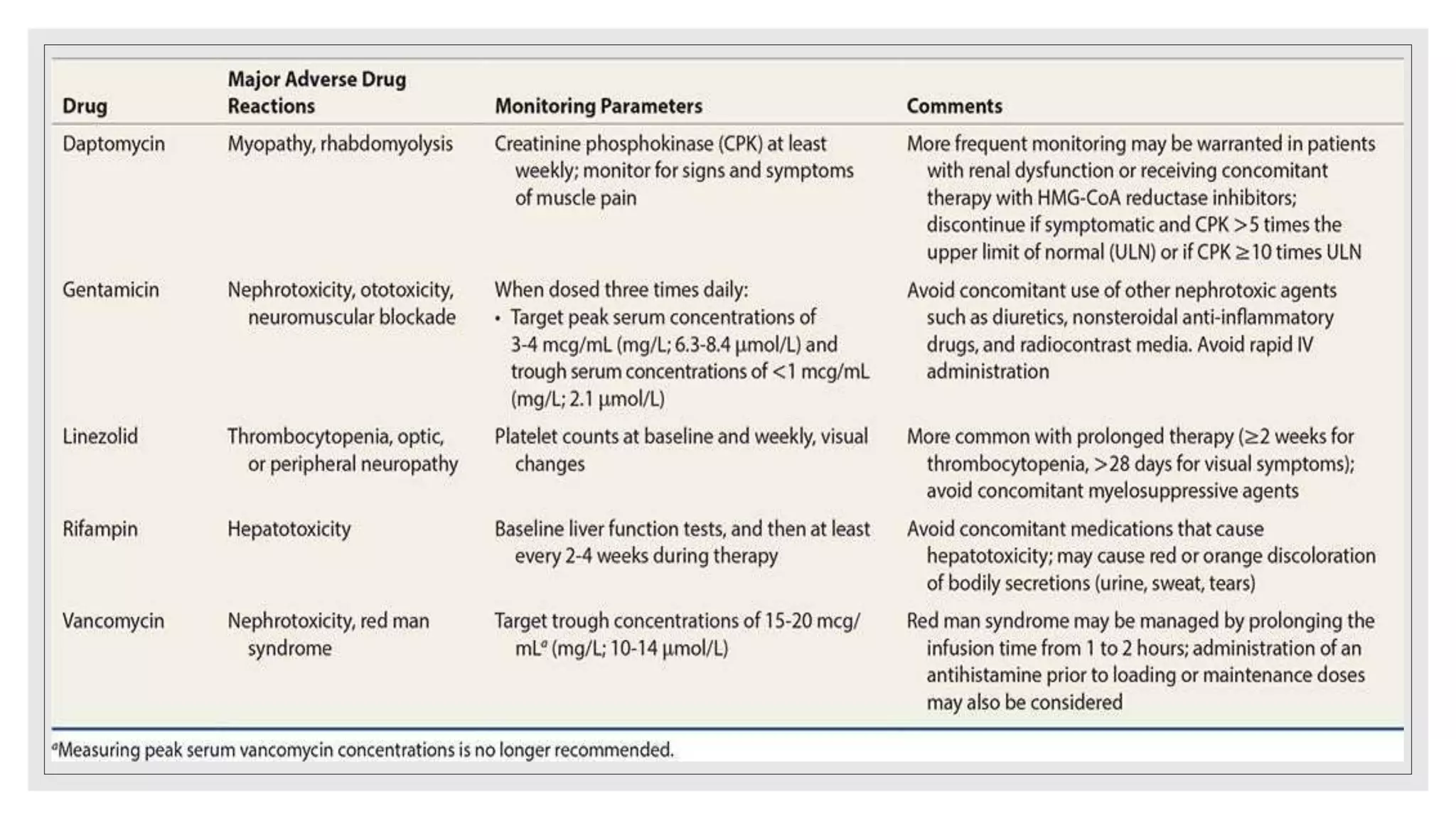
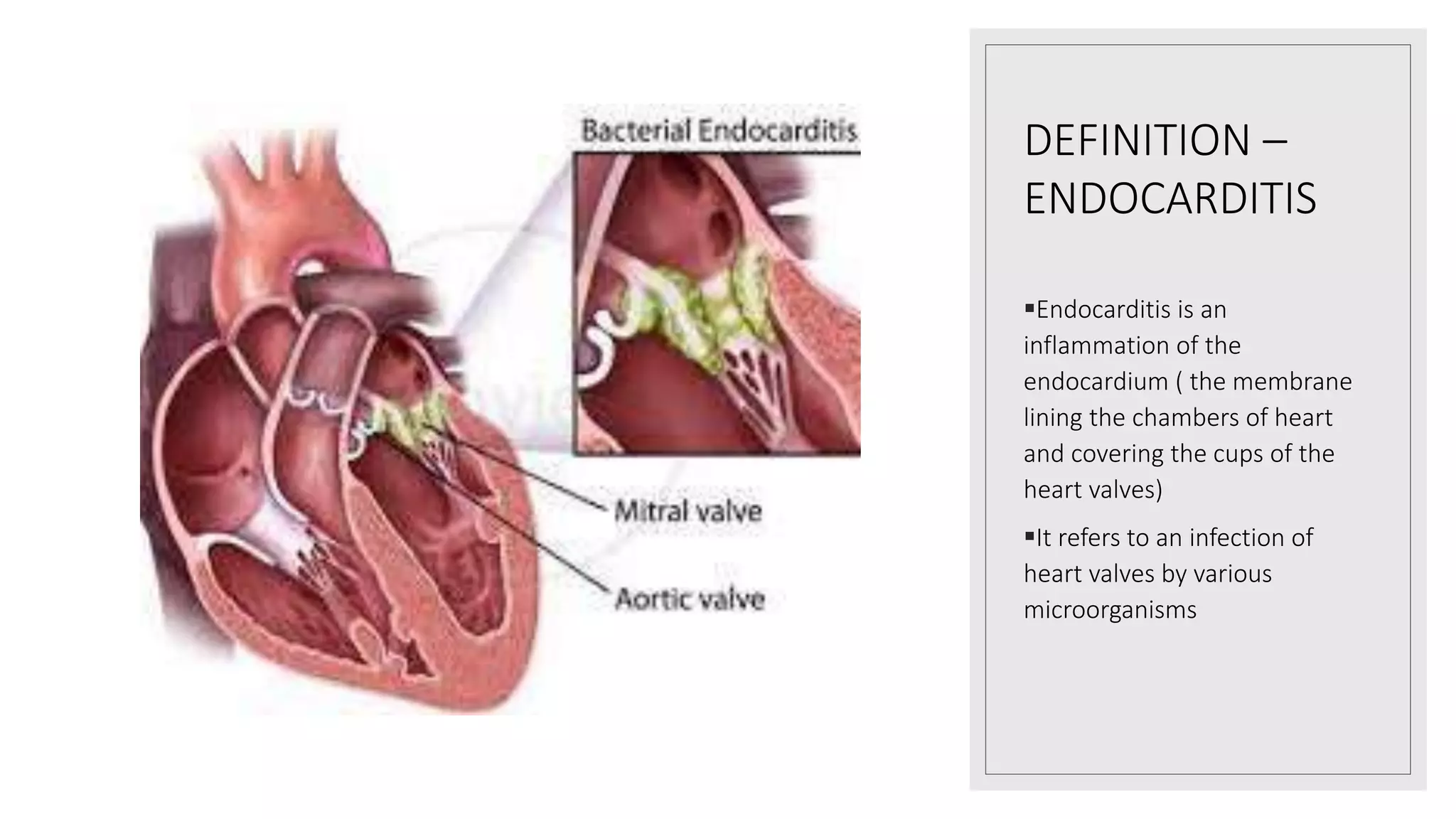
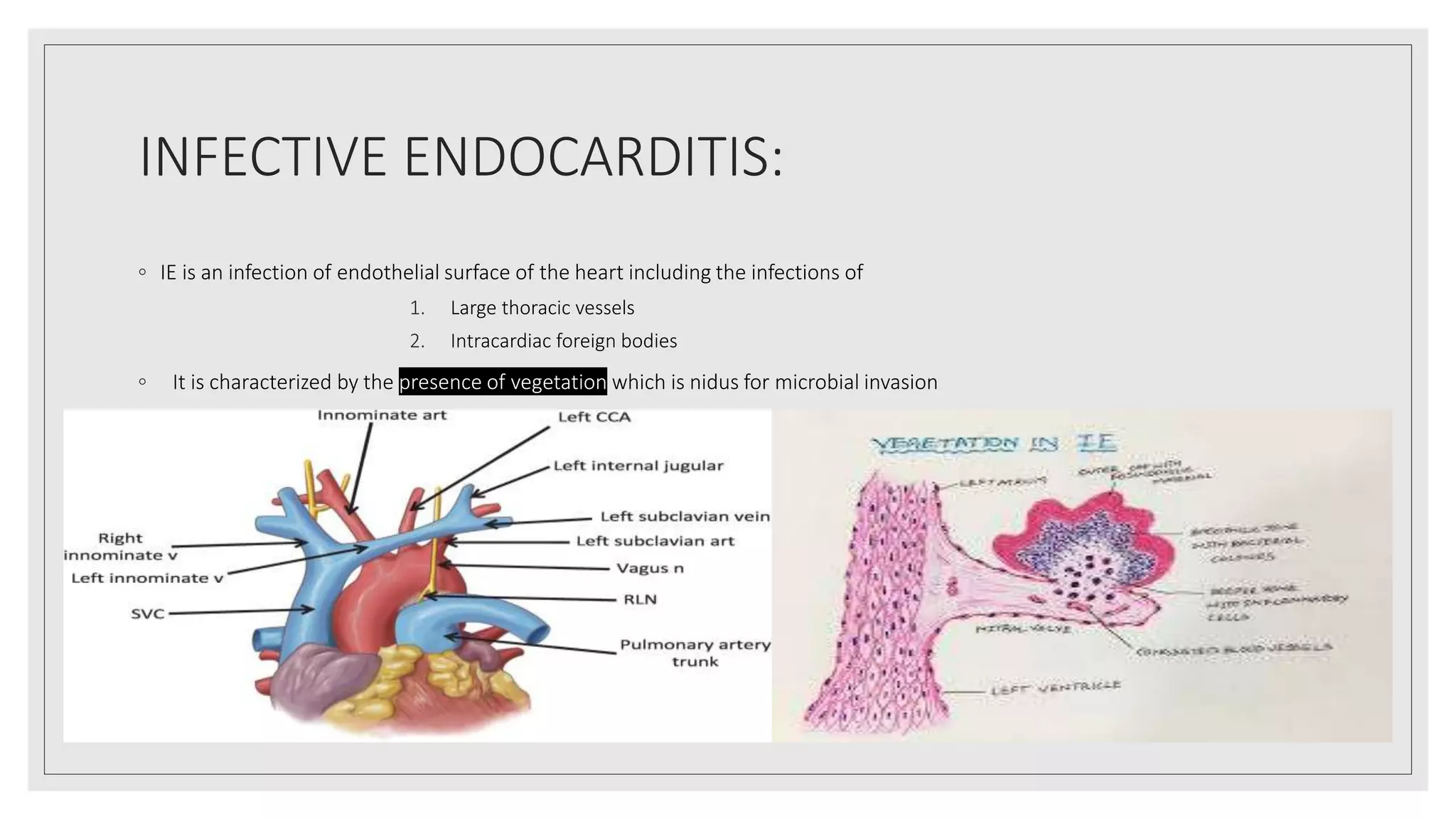
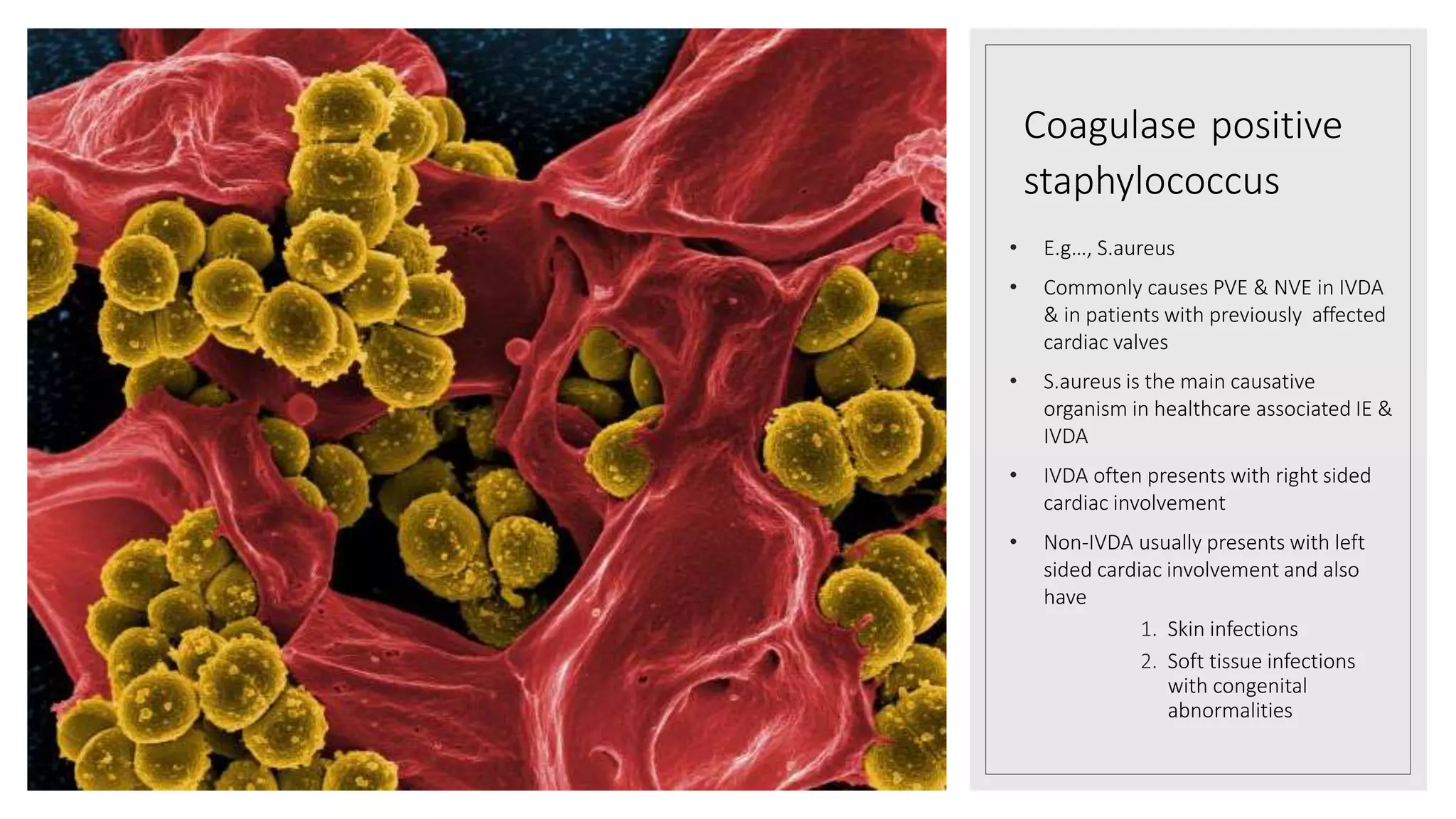
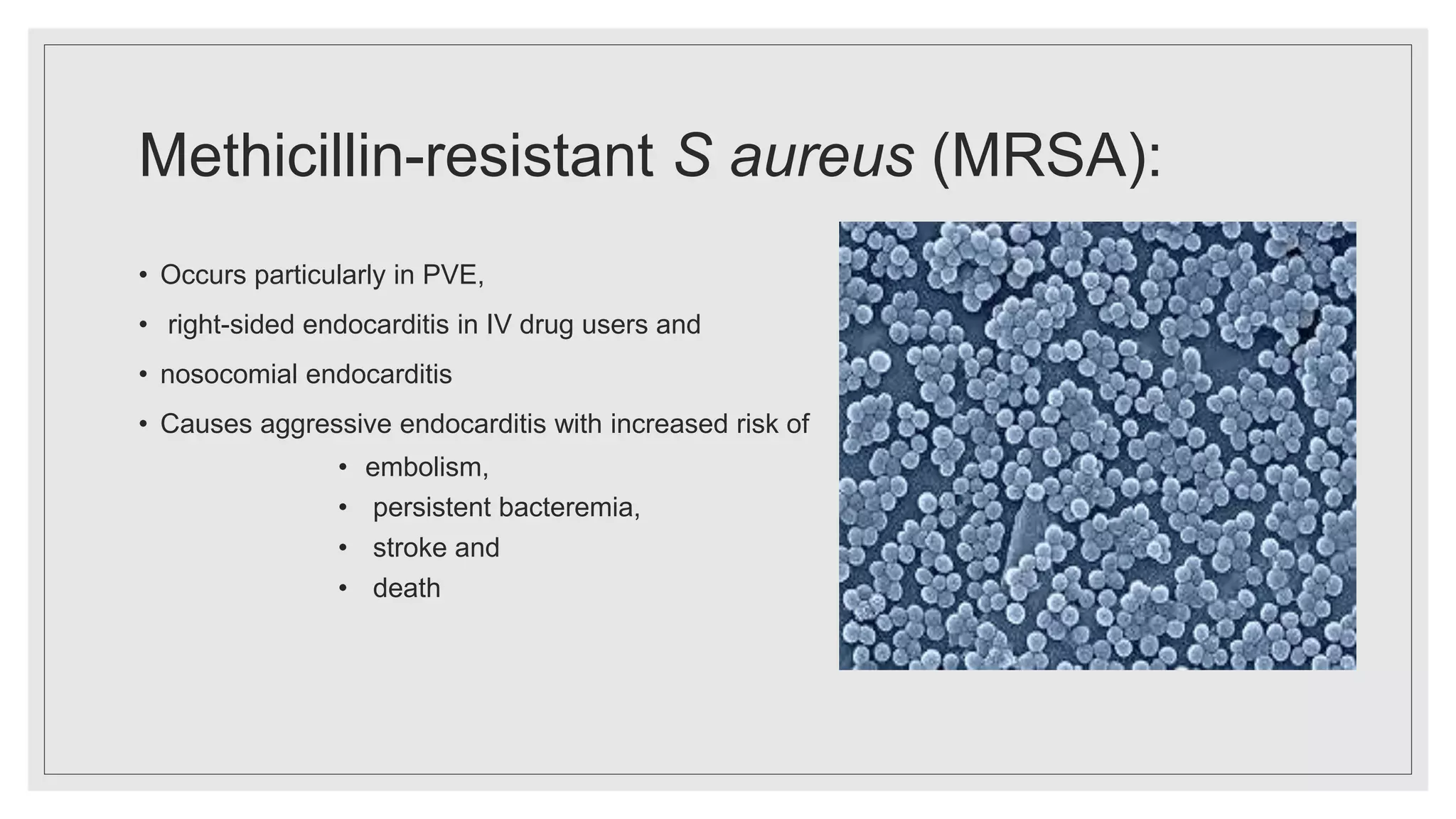
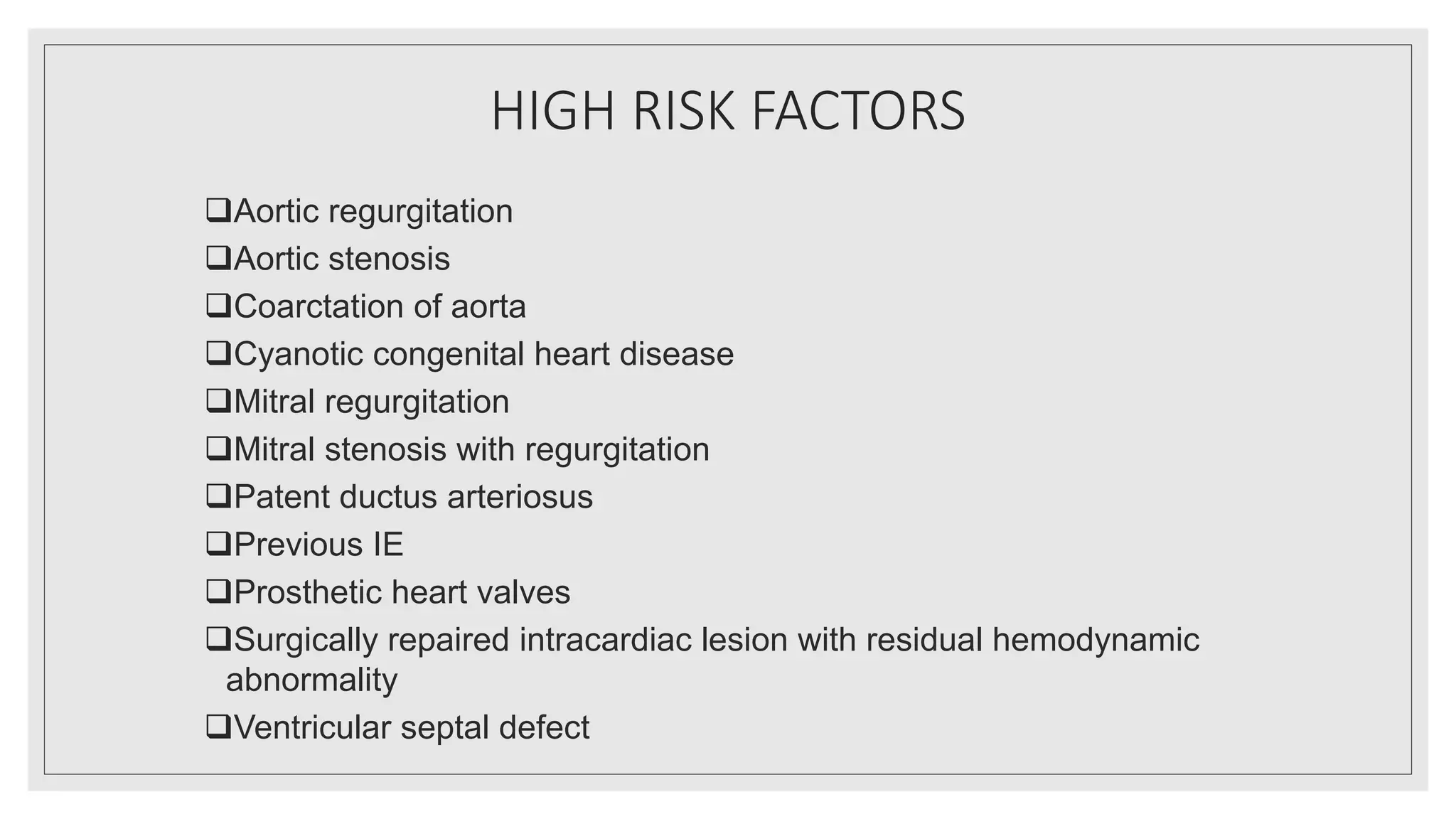
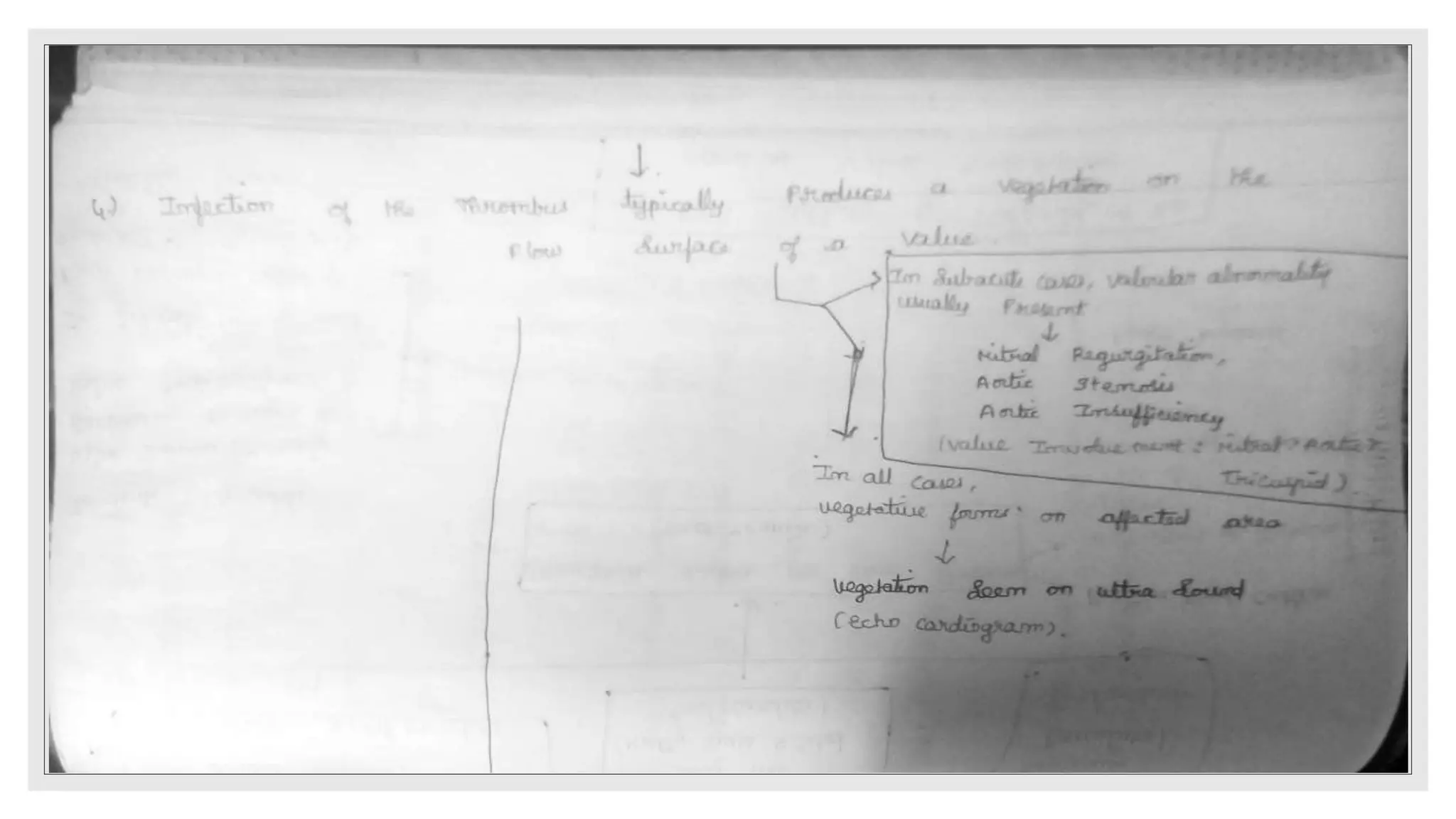
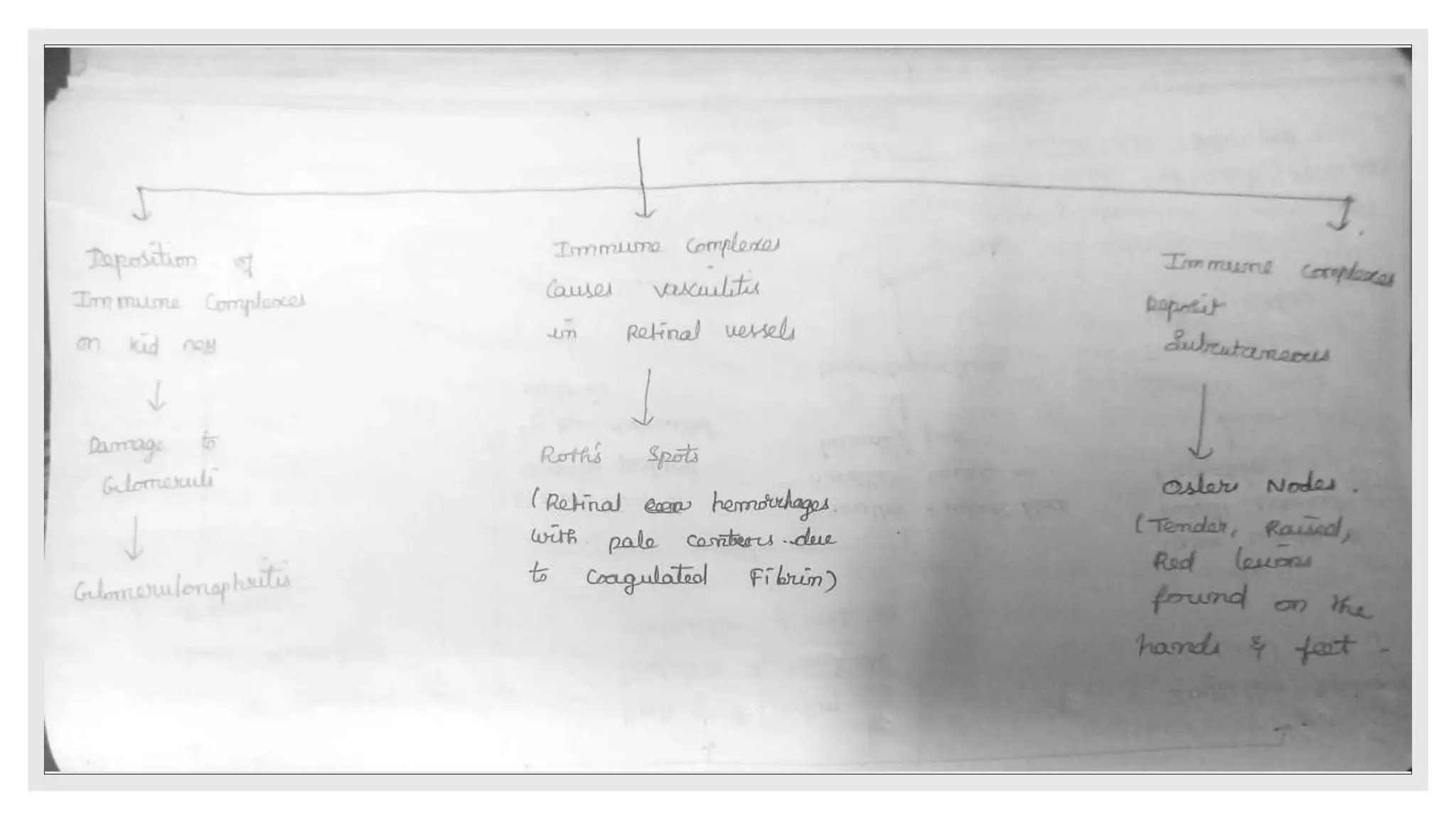
Endocarditis is an inflammation of the heart's endocardium, primarily caused by microbial infections, and is classified into native valve endocarditis (NVE) and prosthetic valve endocarditis (PVE). It can lead to serious complications such as embolism and death if not treated promptly, with varying clinical presentations and risk factors linked to underlying heart conditions and non-cardiac issues. Diagnosis relies on clinical criteria, positive blood cultures, and echocardiographic findings of vegetations or abscesses.
























![Major Criteria
Positive blood culture for IE
• Typical microorganism consistent with IE from 2
separate BCs as noted below:
Viridans streptococci, Streptococcus
gallolyticus (formerly known as S bovis), HACEK
group, or Staphylococcus aureus
Community-acquired enterococci in the absence
of a primary focus; or
• Microorganisms consistent with IE from
persistently positive BCs defined as
≥2 positive cultures of blood samples drawn >12
hours apart; or
All of 3 or a majority of ≥4 separate cultures of
blood (with first and last sample drawn ≥1 hour
apart)
• Single positive BC for Coxiella burnetii or
antiphase I IgG antibody titer ≥1:800
Evidence of endocardial involvement
• Positive echocardiogram for IE [transesophageal
echocardiogram (TEE) recommended in patients
with prosthetic valves and rated at least “possible
IE” by clinical criteria, or complicated IE
(paravalvular abscess); TTE as first test in other
patients] defined as:
Oscillating intracardiac mass on valve or
supporting structures, in the path of
regurgitant jets, or on implanted material in
the absence of an alternative anatomic
explanation; or
Abscess; or
New partial dehiscence of prosthetic valve,
or
• New valvular regurgitation (worsening or
changing of preexisting murmur not sufficient)](https://image.slidesharecdn.com/ieseminar-210516152006/75/Infective-endocarditis-endocarditis-32-2048.jpg)













![•If combination antimicrobial therapy is used, then the agents should be
administered close together to improve synergistic killing effect
•Antibiotic prophylaxis has been limited to patients undergoing an invasive dental
procedure in whom exists a history of infective endocarditis (IE), prosthetic valve,
a heart transplant with abnormal heart valve function, or congenital heart disease
with the following: Unrepaired cyanotic congenital heart disease, congenital heart
defect completely repaired with prosthetic material or device for the first 6 months
post procedure, or repaired congenital heart disease with residual defects
• Patients with prosthetic valves are at the highest risk of developing IE
• Recommended prophylaxis regimens include the standard Amoxicillin,
Ampicillin if unable to take PO medications, and Clindamycin [PO or
intravenous (IV)], Cefazolin or Cefalexin if with penicillin allergy
• Recommended prophylaxis regimen for genitourinary and gastrointestinal
procedures should include drugs against enterococci eg Ampicillin or
Vancomycin](https://image.slidesharecdn.com/ieseminar-210516152006/75/Infective-endocarditis-endocarditis-49-2048.jpg)







![Streptococca
l IE
Abiotrophia and Granulicatella [formerl
y nutritionally variant streptococci
(NVS)]
• Antimicrobial susceptibility is technically difficult
to determine because it is slow growing
• Recommended regimen includes Penicillin,
Ampicillin, Ceftriaxone or Vancomycin for 6
weeks plus an aminoglycoside (eg Gentamicin)
for at least the first 2 weeks](https://image.slidesharecdn.com/ieseminar-210516152006/75/Infective-endocarditis-endocarditis-57-2048.jpg)