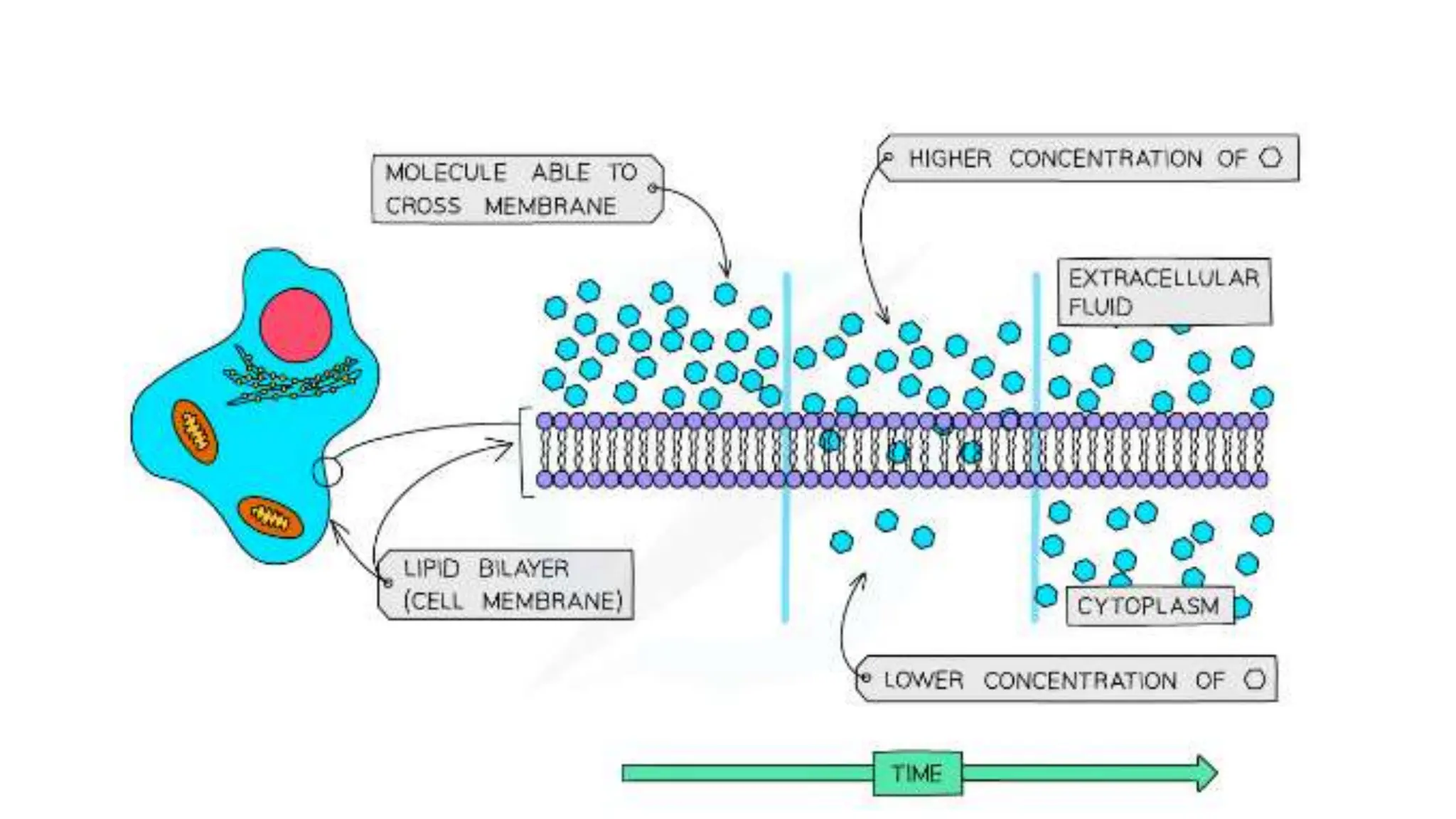
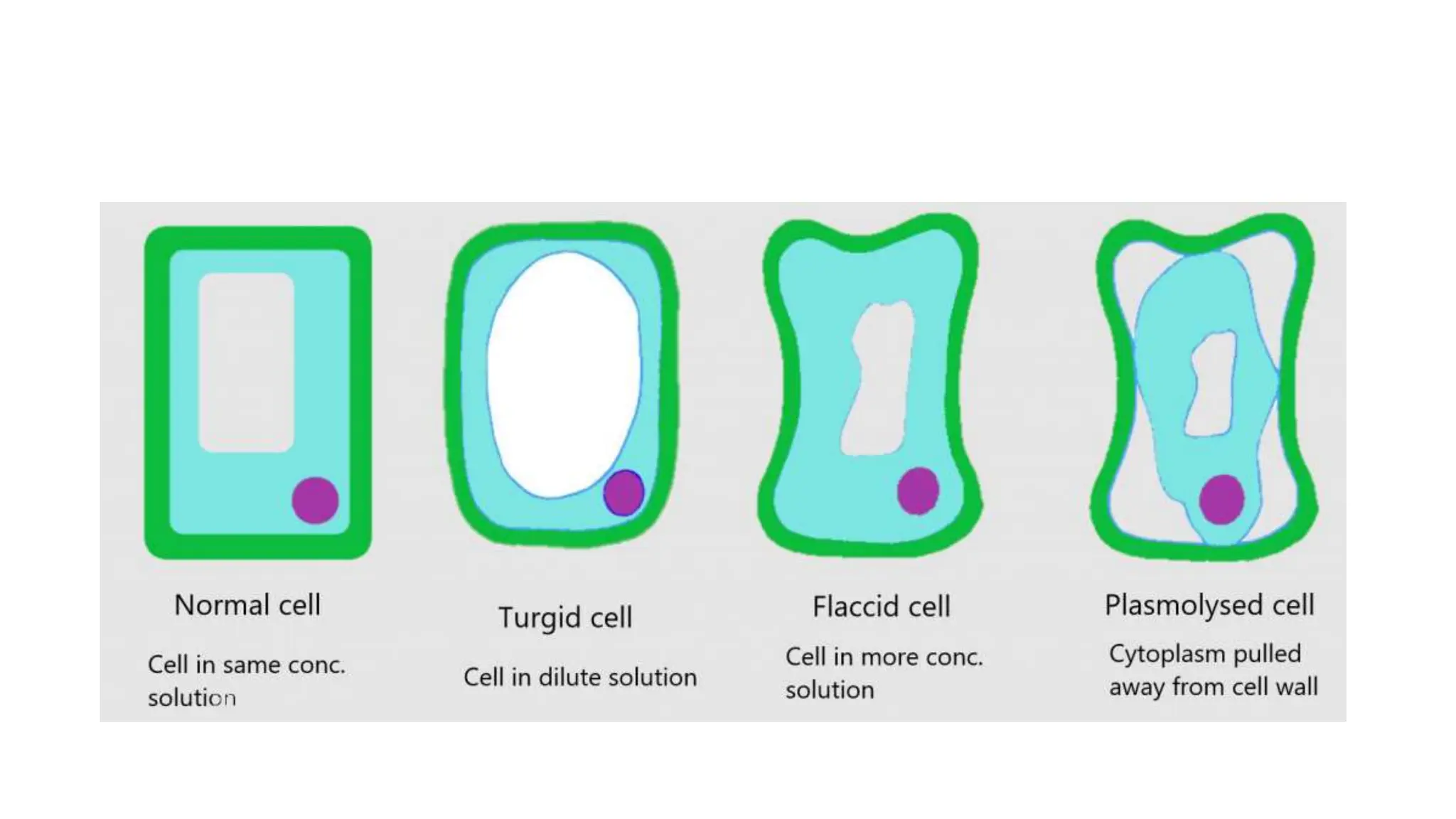
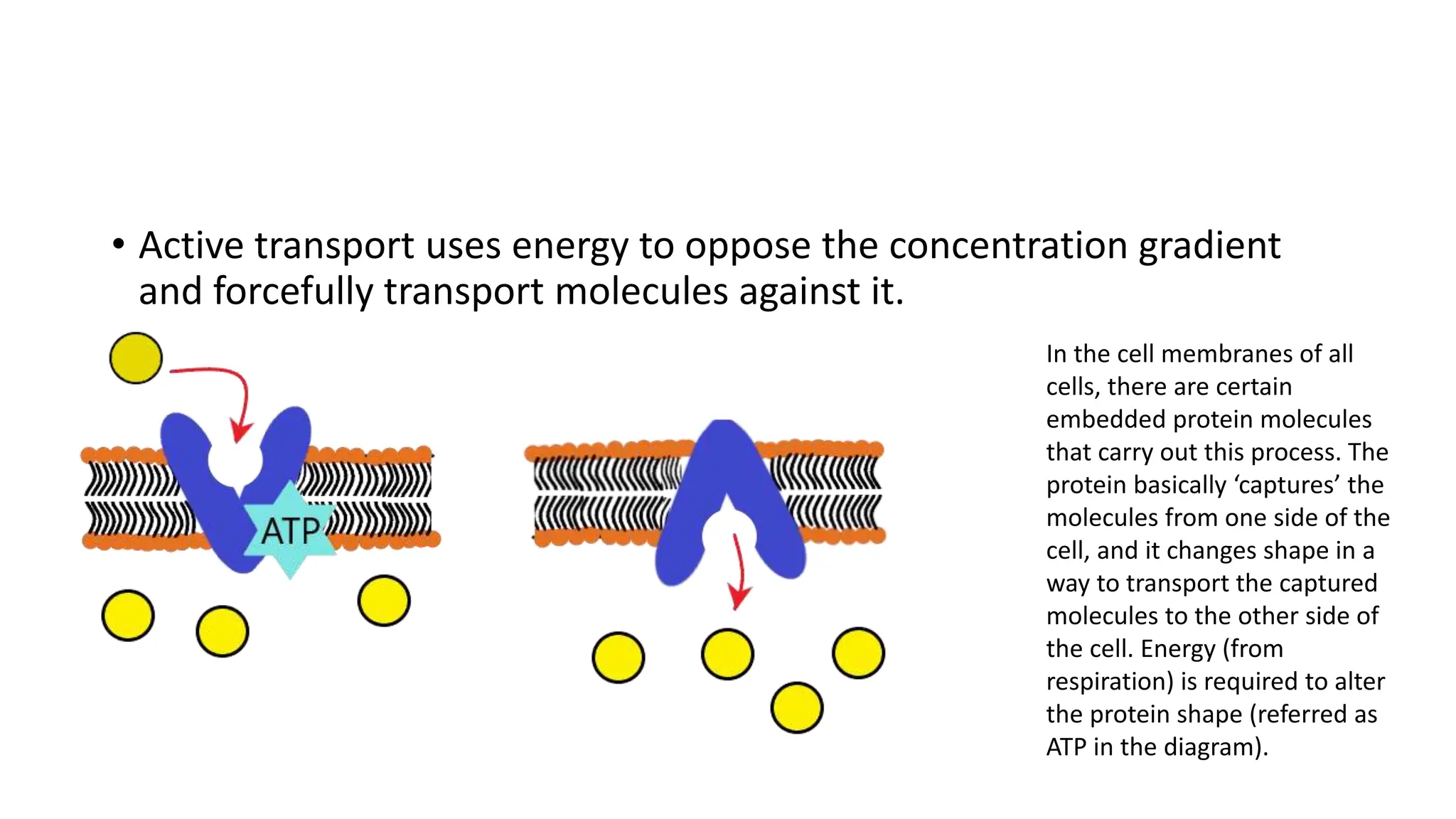
Diffusion, osmosis, and active transport are three important ways that molecules move across cell membranes. Diffusion is the passive movement of molecules from an area of higher concentration to lower concentration down a concentration gradient. Osmosis is the diffusion of water across a partially permeable membrane from an area of higher water potential to lower water potential. Active transport requires energy and transports molecules against their concentration gradient from lower to higher concentration using protein carriers in the cell membrane.