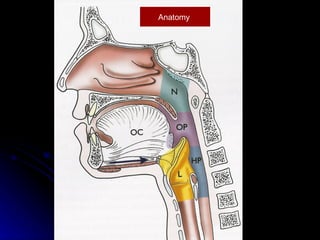
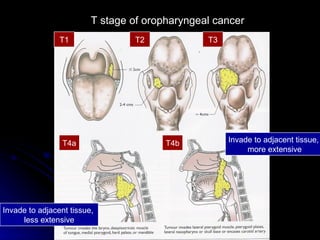
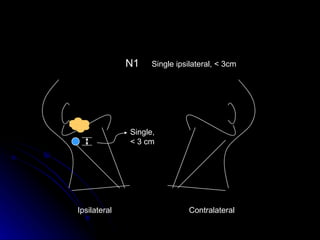
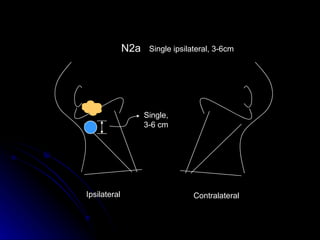
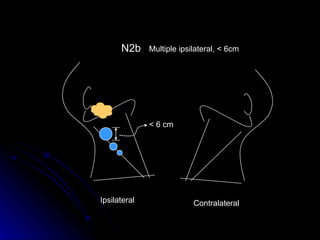
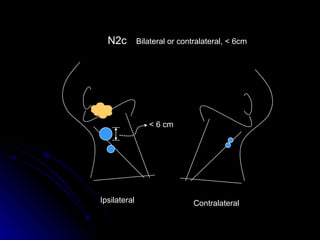
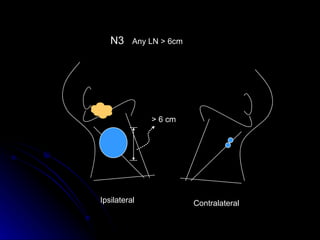
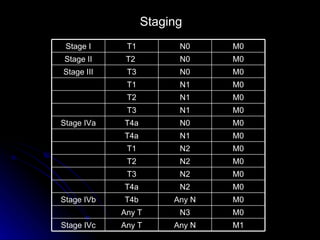
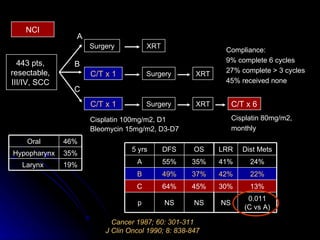
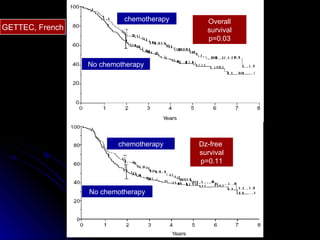
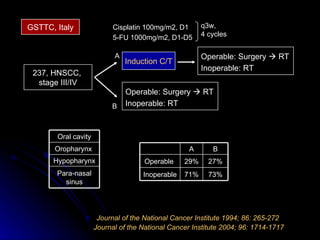
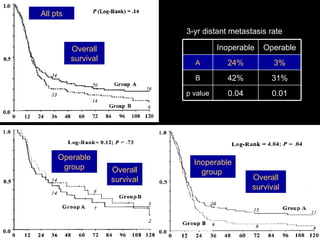
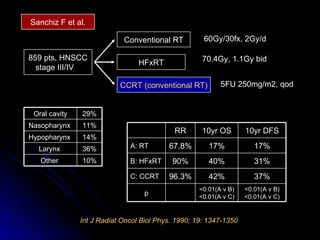
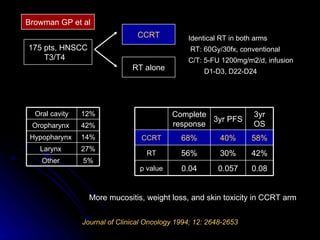
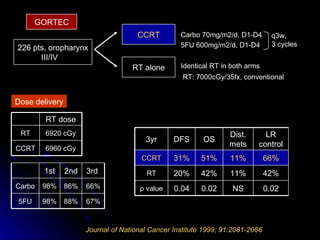
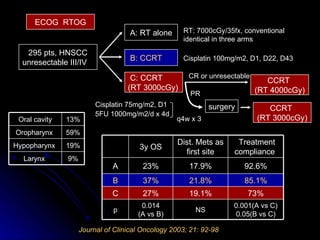
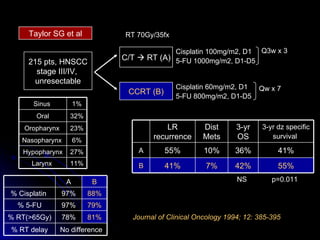
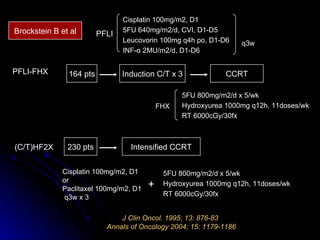
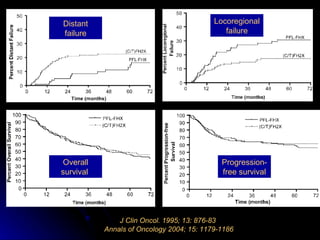
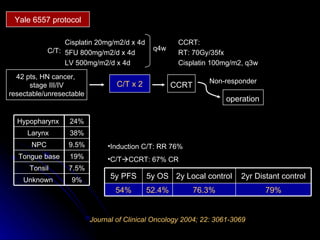
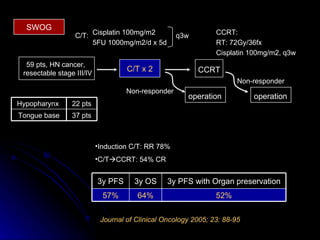
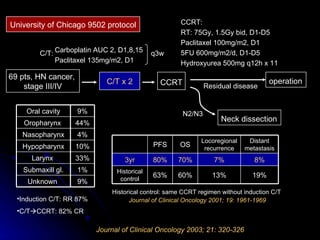
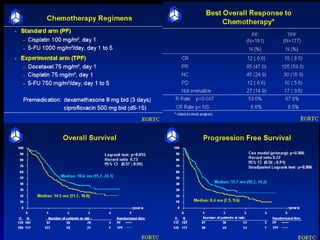
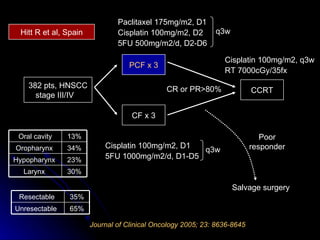
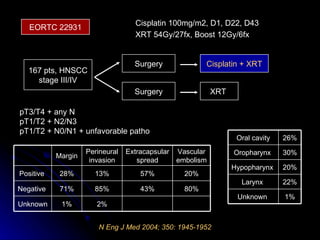
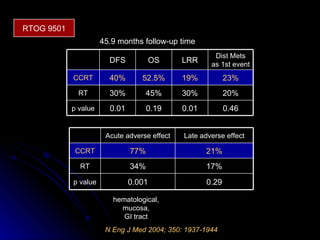
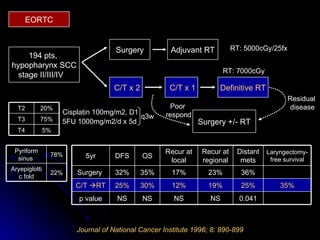
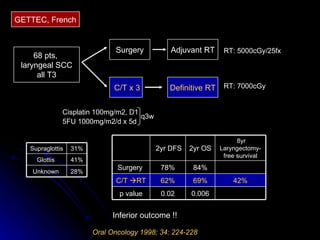
Multimodality treatment of head and neck squamous cell carcinoma (SCC) involves incorporating chemotherapy into definitive local treatments such as surgery or radiotherapy (RT) to improve outcomes. Concurrent chemoradiotherapy (CCRT) has been shown to enhance locoregional control compared to RT alone with minimal effects on distant metastasis. CCRT also improves survival outcomes compared to sequential chemoradiotherapy or RT alone. Induction or adjuvant chemotherapy may help reduce distant failure rates when combined with CCRT by providing more systemic treatment.