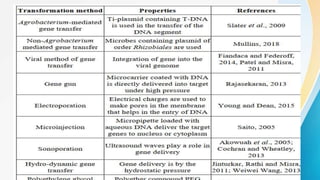
Genetic transformation methods allow for the insertion of foreign genes into plant genomes. This document outlines various genetic transformation techniques, including Agrobacterium-mediated transformation, particle bombardment, electroporation, and chemical methods using PEG or DEAE dextran. Agrobacterium is a natural gene transfer agent for many plants, while particle bombardment can transform a wide variety of plant species. Genetic transformation has important applications for improving crop resistance to stresses and pests, increasing yields, and enhancing nutritional quality.