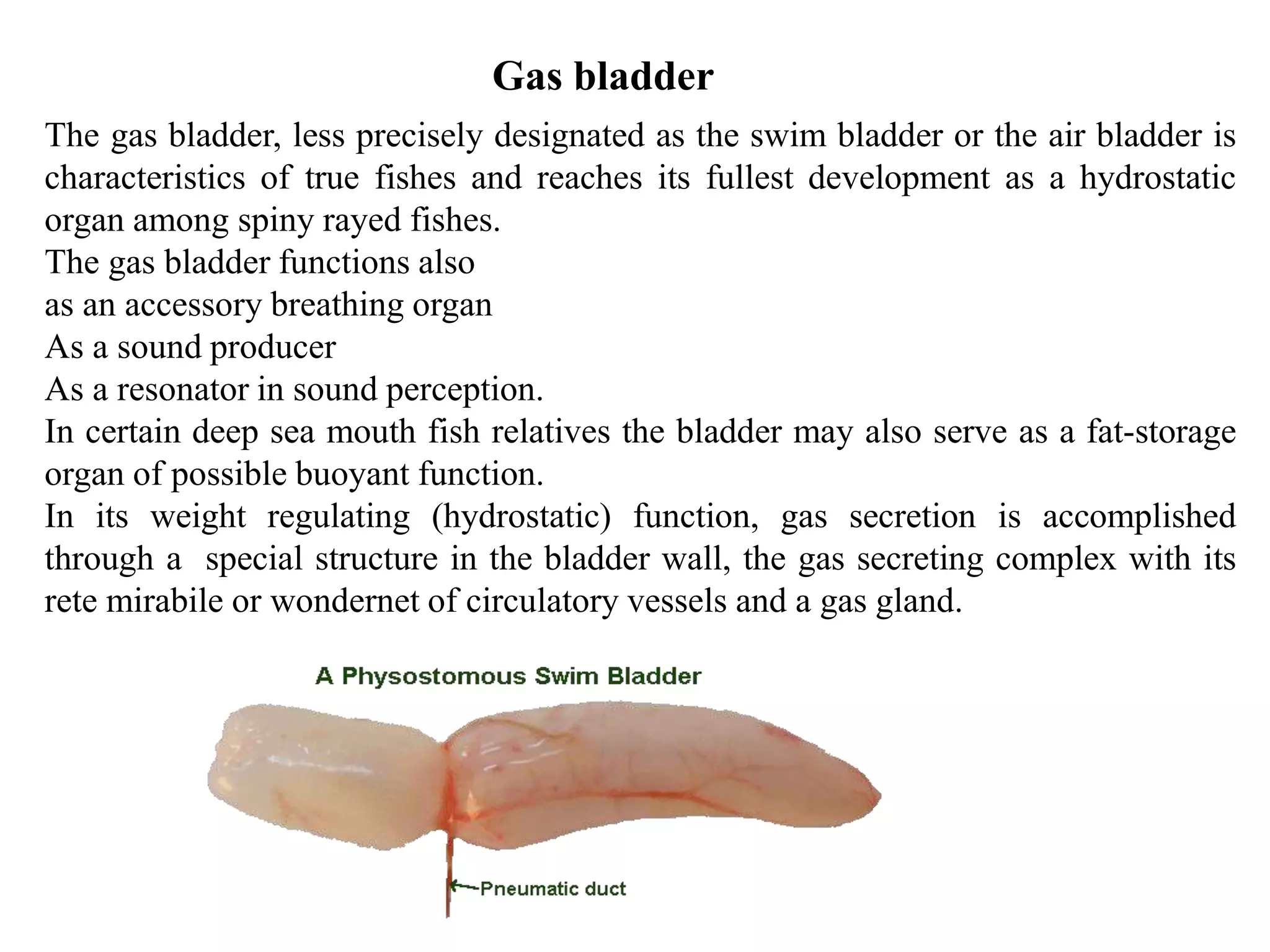
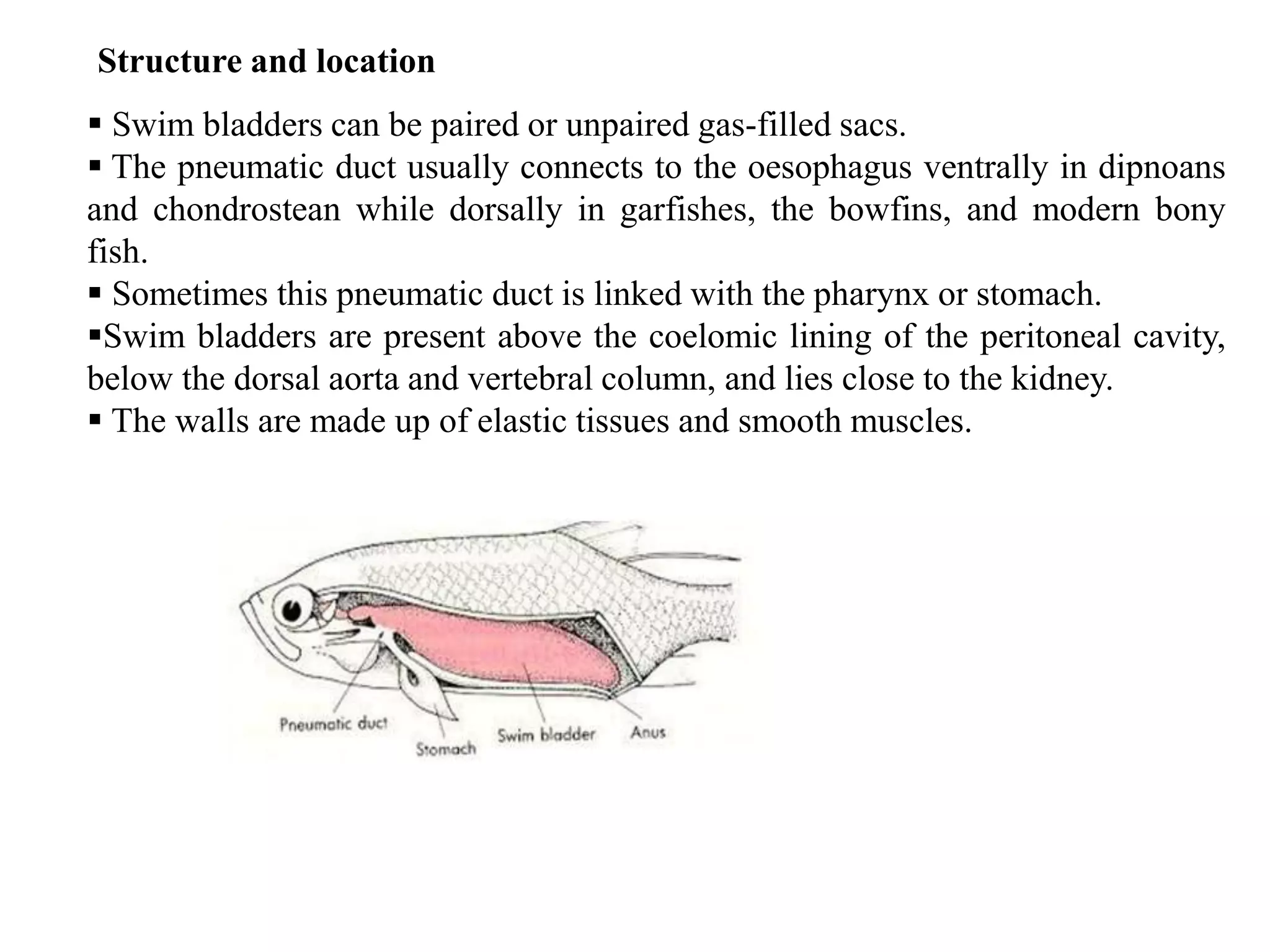
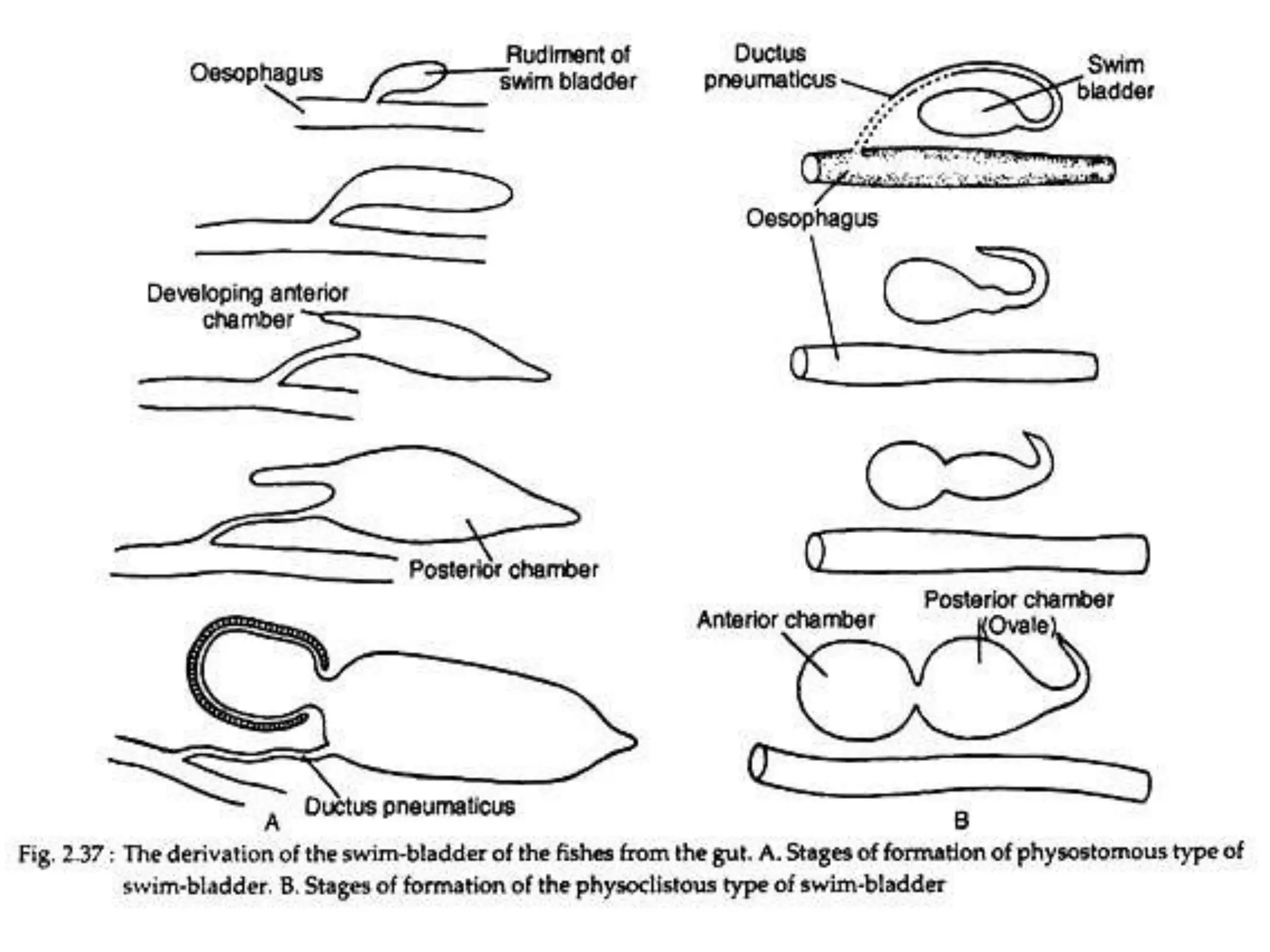
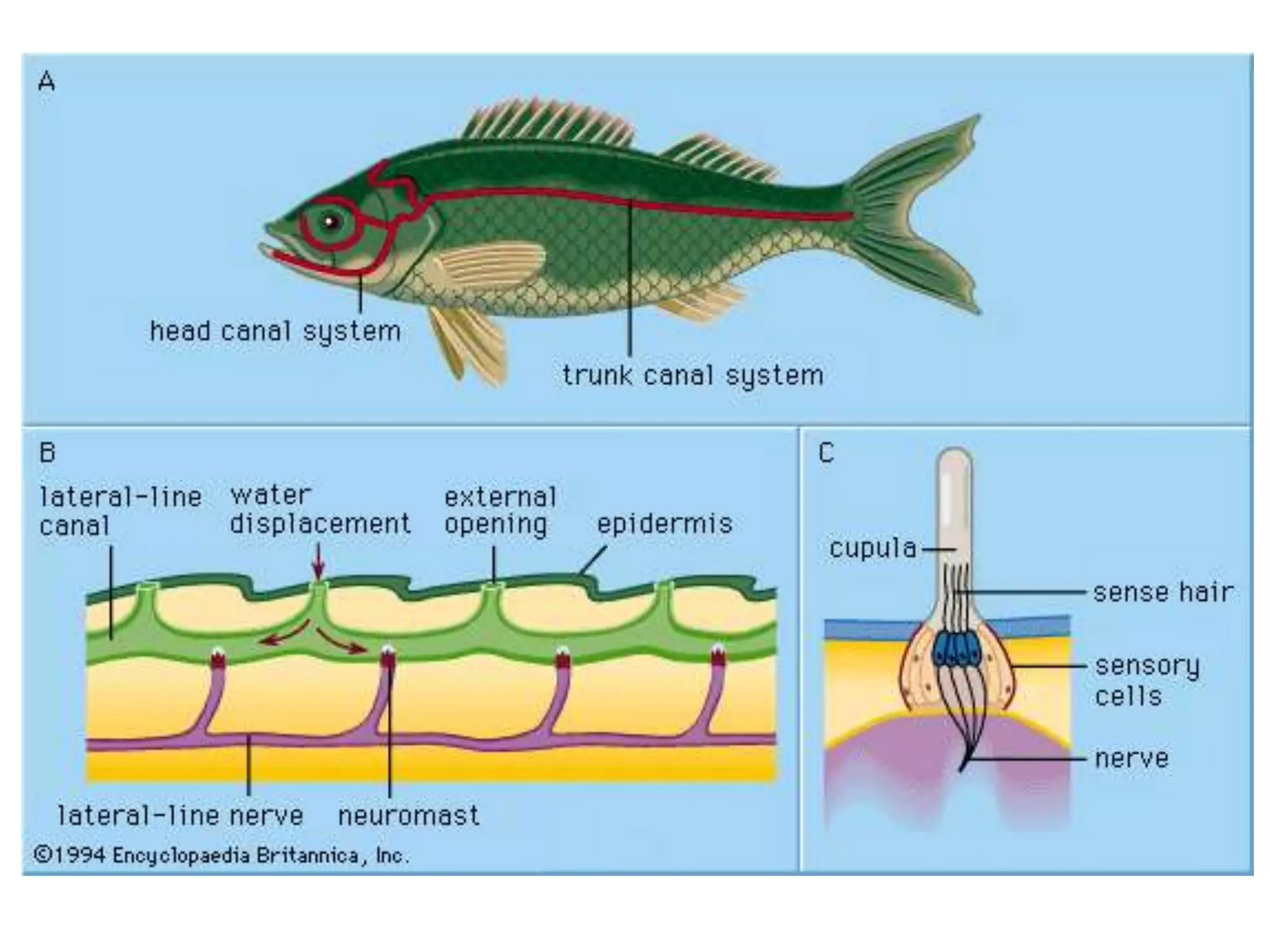
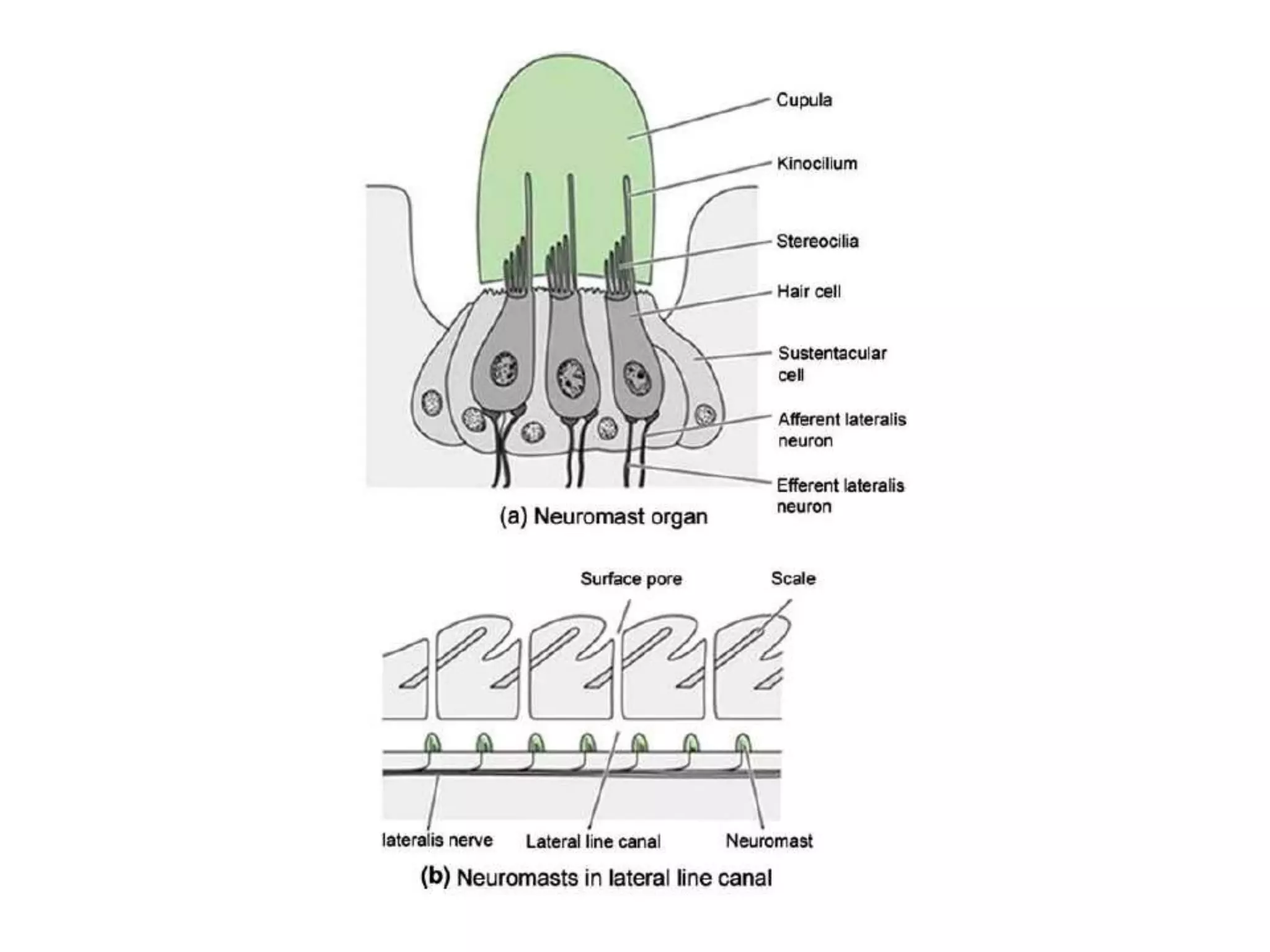
The gas bladder, or swim bladder, functions as a hydrostatic organ that helps regulate a fish's buoyancy. It allows fish to control their depth by increasing or decreasing the volume of gas inside the bladder. The bladder walls are made of elastic tissues and smooth muscles. Gases like nitrogen, oxygen, and carbon dioxide are stored inside. The lateral line system detects water movements and aids in tasks like navigation, prey detection, and schooling behavior. It consists of neuromasts, which are mechanoreceptors sensitive to water displacement. The neuromasts contain hair cells that detect water motions and transmit signals to the brain via afferent nerves. Color patterns in fish provide camouflage, communication, and other functions