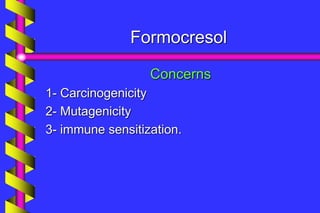
This document discusses the use of ferric sulfate as a medicament in pulpotomy procedures in primary teeth. It begins with an overview of pulpotomy techniques and medicaments used, including ferric sulfate, formocresol, mineral trioxide aggregate, calcium hydroxide, and glutaraldehyde. It then presents a clinical case where ferric sulfate was used successfully in a pulpotomy. The document discusses debates around the continued use of formocresol due to concerns about carcinogenicity and immune sensitization, though some research finds the dose from pulpotomies to be very small and its use safe. Overall, the document examines ferric sulfate and formocresol as options for pulpot

































![Ferric Sulphate
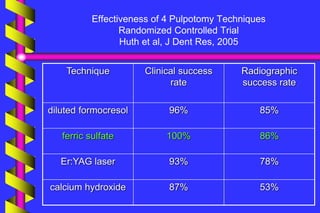
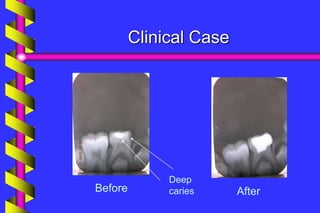
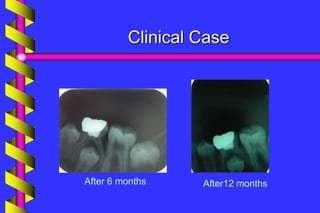
Clinical outcome
A meta-analysis of formocresol versus ferric
sulphate primary molar pulpotomies included
94 studies found both approaches to have a
similar rate of clinical and radiographic
success
[Loh et al. Pediatric Dentistry 2004]](https://image.slidesharecdn.com/ferricsulfate2-200803190936/85/Ferric-sulfate-2-40-320.jpg)