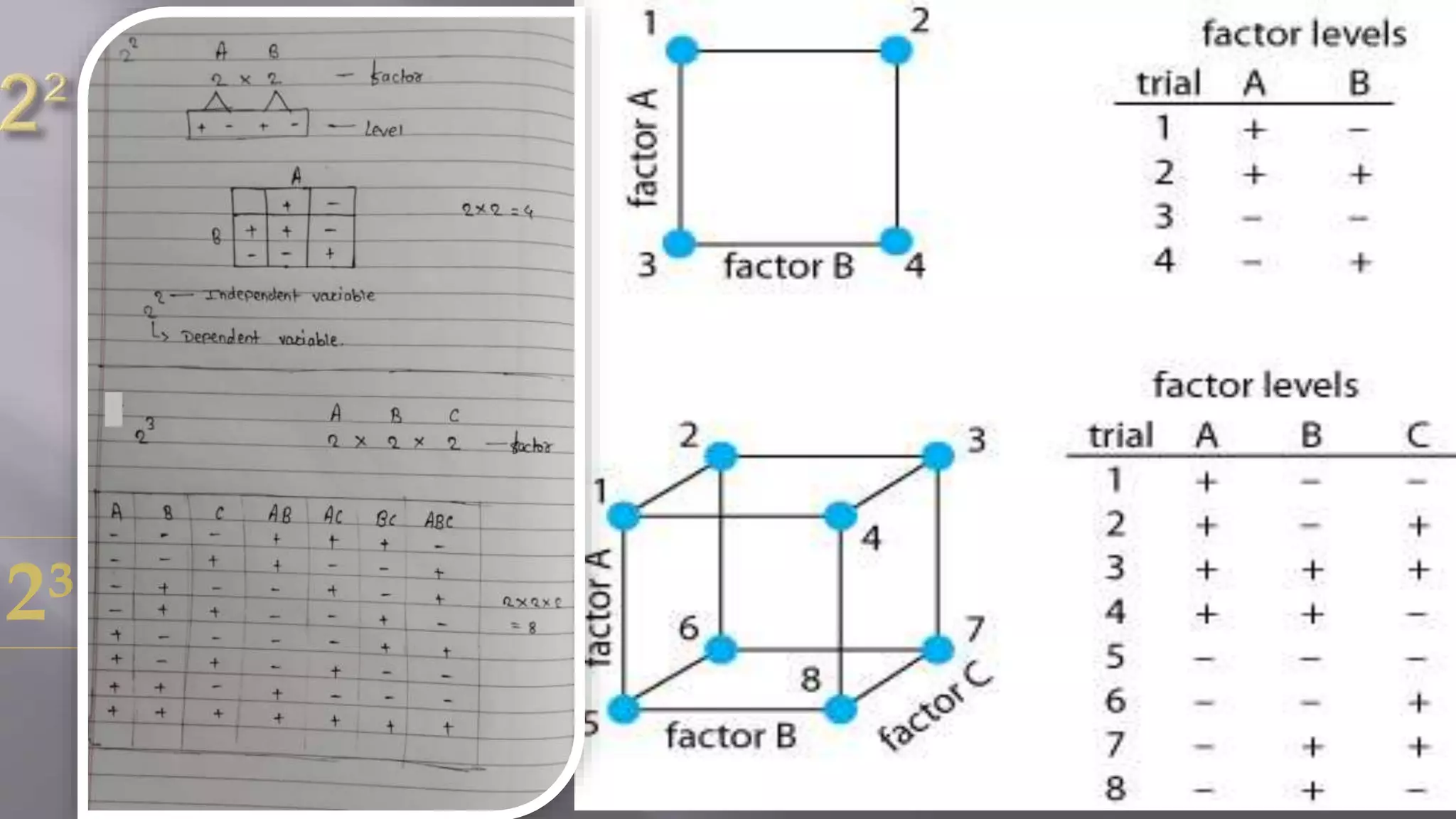
The document discusses factorial design in the context of pharmaceutical formulation, emphasizing its role in optimizing drug delivery systems through design of experiments (DOE). It outlines different types of factorial designs, their advantages and disadvantages, and their application in research. Additionally, it underscores the importance of patient safety and product quality linked to quality by design principles.