This document discusses computer aided formulation development and optimization techniques. It provides an overview of optimization terminology, experimental design techniques like factorial designs and response surface methodology that can be used for optimization. It discusses how optimization is a regulatory requirement for formulation development and validation. It also mentions some commonly used computer software packages for experimental design like Design-Expert, Minitab, JMP etc.




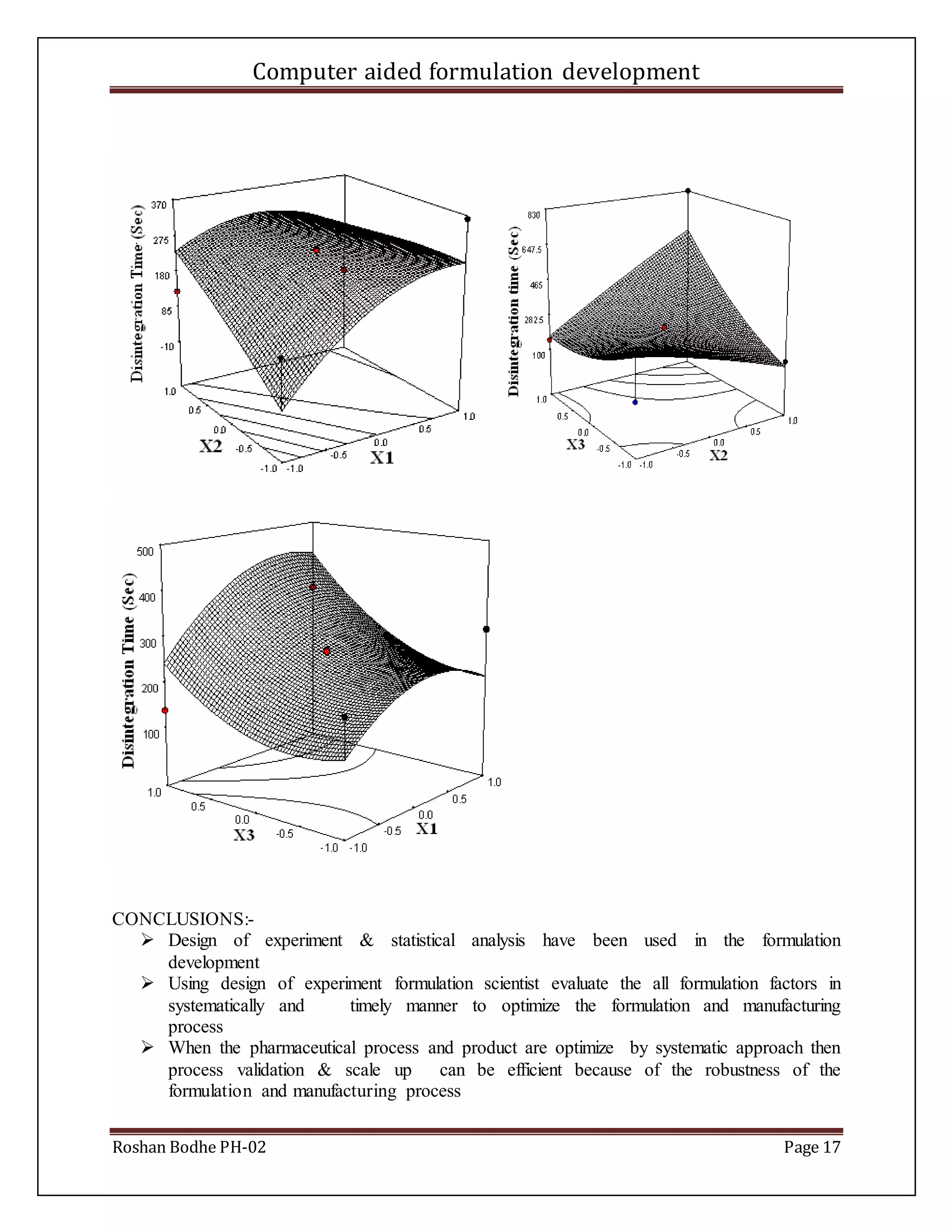
![Computer aided formulation development
Roshan Bodhe PH-02 Page 5
Besides RSM, the design is also used for screening of influential variables and factor influence
studies. Representsa 22 and 23 FD pictorially, where each point represents and individual
experiment.The mathematical model associated with the design consists of the main effects of
each variable plus all the possible interaction effects, i.e., interactions between the two variables,
and in fact, between as many factors as are there in the model.
The mathematical model generally postulated for FDs is given as
Y = b0 + b1X1... + b12X1X2... + b123X1X2X3... + e …
where,
bi, bij and e represent the coefficients of the variables and the interaction terms, and the random
experimental error, respectively. The effects (coefficients) in the model are estimated usually by
multiple linear regression analysis selection'. Their statistical significance is determined and then
a simplified model can be written
B Central composite design and its modifications:-
Also known as Box-Wilson design, it is the most often used design for second-order models ,
Central composite design (CCD) is comprised of the combination of a two-level factorial points
(2n),axial or star points (2n) and a central point.
Thus the total number of factor combinations in a CCD is given by 2n +2n + 1. The axial points
for a two-factor problem include, (± a, 0) and (0, ± a), where a is the distance of the axial points
from the center. A two factor CCD is identical to a 32 FD with square experimental domain .
a) face centered cube design (FCCD):-
Results when the same positive and negative distance is taken from the center in a CCD .
A rotatable is identical to FCCD except that the points defined for the star design are changed to
[± (2n )1/4,… 0] and those generated by the FD remain unchanged. In this way, the design
generates information equally well in all the directions.
e.g. the variance of the estimated response is same at all the points on a sphere centered at the
origin. The second-order polynomial for two factors, generally used for the composite designs.
b)Box-Behnken Design:-
Is a specially made design that requires only 3 levels (-1, 0, 1). It overcomes the inherent pitfalls
of CCD, where each factor has to be studied at 5 levels (except for 2 factors with a = ± 1, where
the number of levels per factor is 3), thus the number of experiments increases with rise in the
number of factors.
A BBD is an economical alternative to CCD. Also called as orthogonal balanced incomplete
block design, these are available for 3 to 10 factors. Because the design involves study at three
levels, the quadratic model is considered to be most appropriate.
C ) Mixture designs:-
In FDs and the CCDs, all the factors under consideration can simultaneously be varied and
evaluated at all the levels. This may not be possible under many situations. Particularly, in
pharmaceutical formulations with multiple excipients, the characteristics of the finished product
usually depend not so much on the quantity of each substance present but on their proportions.
Here, the sum total of the proportions of all excipients is unity and none of the fractions can be
negative.
Therefore, the levels of the various components can be varied with the restriction that the sum
total should not exceed one. Mixture designs are highly recommended in such cases. In a two-
component mixture, only one factor level can be independently varied, while in a three-
component mixture only two factor levels, and so on. The remaining factor level is chosen to](https://image.slidesharecdn.com/newmicrosoftassignment2-180413174853/75/COMPUTER-AIDED-FORMULATION-DESIGN-EXPERT-SOFTWARE-CASE-STUDY-5-2048.jpg)