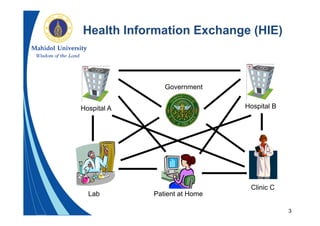
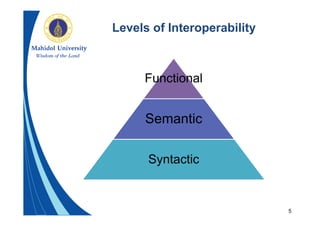
This document provides an overview of the HL7 Clinical Document Architecture (CDA) standard for exchanging clinical documents. It describes what CDA is, the components of a CDA document, how CDA documents can be rendered and exchanged in messages, and examples of use cases for CDA. The key points are that CDA specifies an XML format for clinical documents to enable their structured exchange between systems, CDA documents have human-readable and machine-processable parts, and CDA relies on other HL7 standards for semantics.















![18
What Is HL7 CDA?
• “A document markup standard that
specifies structure & semantics of “clinical
documents” for the purpose of exchange”
[Source: HL7 CDA Release 2]
• Focuses on document exchange, not
message exchange
• A document is packaged in a message
during exchange
• Note: CDA is not designed for document
storage. Only for exchange!!](https://image.slidesharecdn.com/exploringhl7cdaitsstructures-140505040219-phpapp01/85/Exploring-HL7-CDA-Its-Structures-18-320.jpg)
































