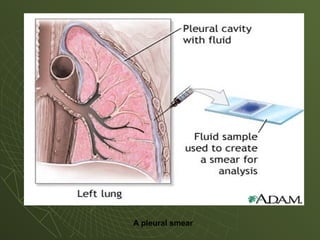
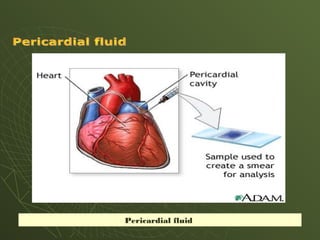
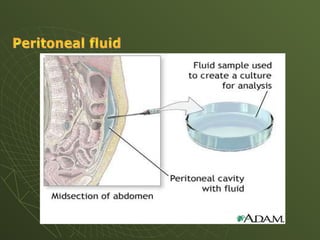
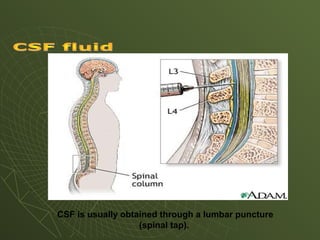
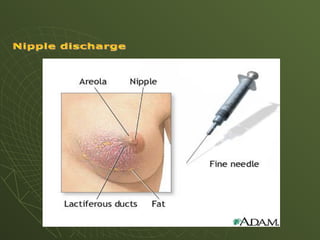
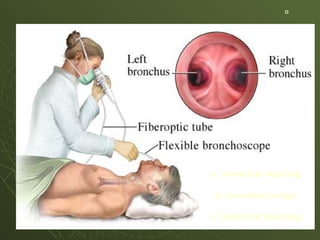
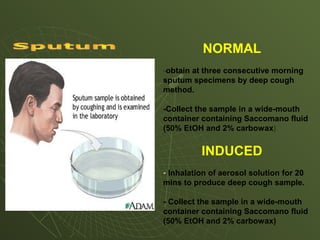
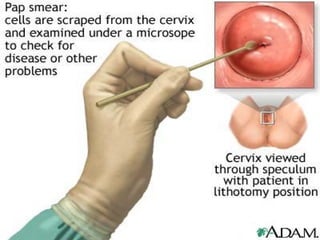
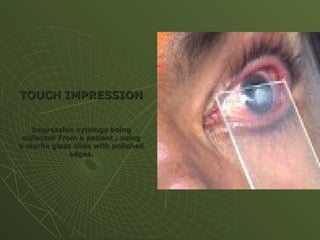
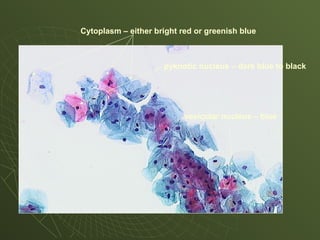
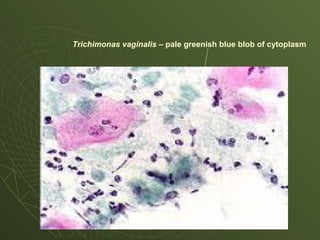
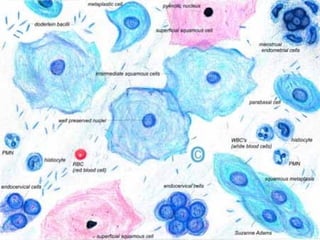
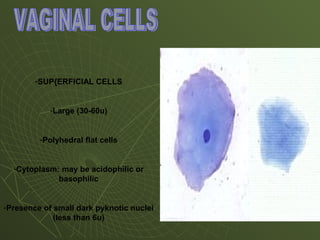
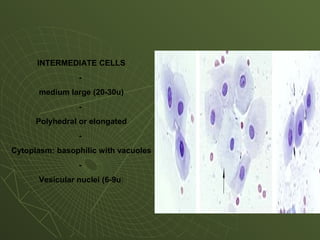
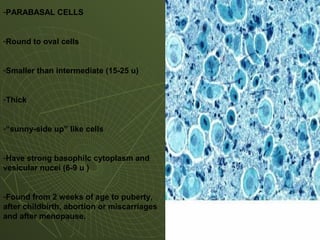
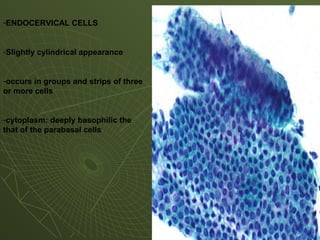
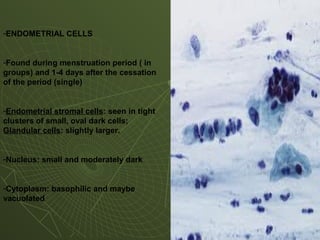
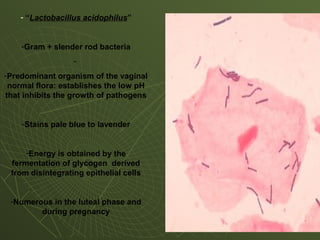
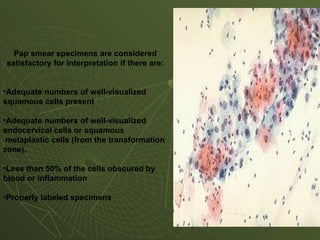
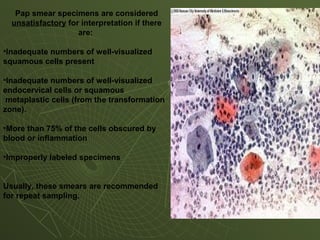
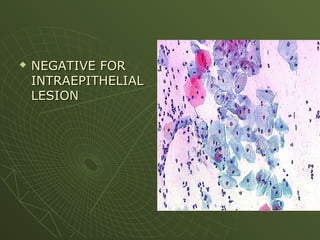
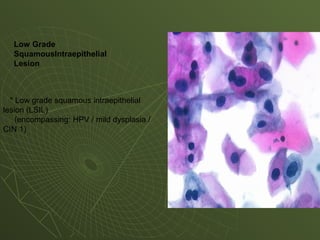
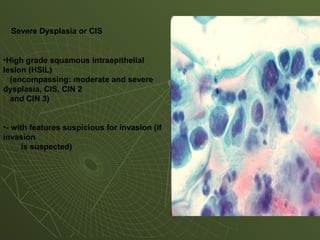
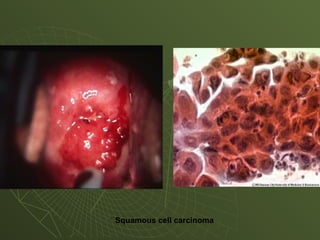
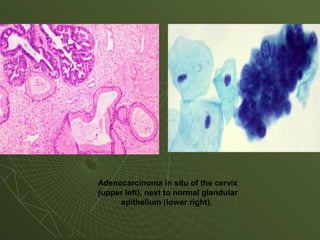
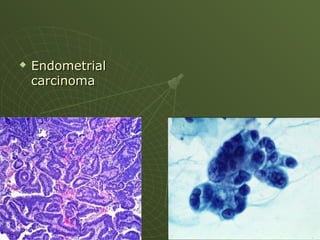
The document discusses cytology and exfoliative cytology. It describes how exfoliative cytology involves the microscopic study of cells that have desquamated from epithelial surfaces. Some applications of exfoliative cytology include detection of malignant cells, precancerous lesions, and infectious microorganisms. The document also outlines procedures for collecting and preparing different cytology specimens, such as body fluids, nipple discharge, bronchial brushings, sputum, gastric washings, and cervicovaginal smears. It provides details on fixation, staining techniques including Pap's stain, and morphological changes that indicate malignancy in cytology samples.