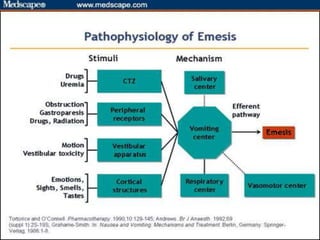
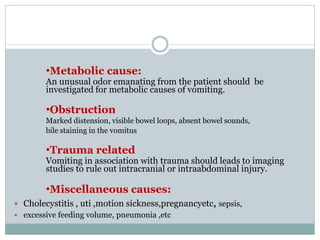
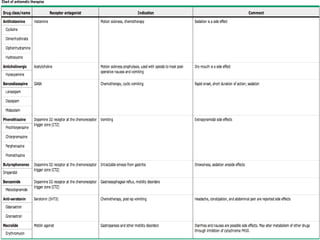
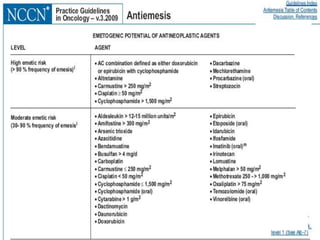
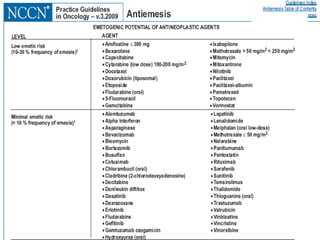
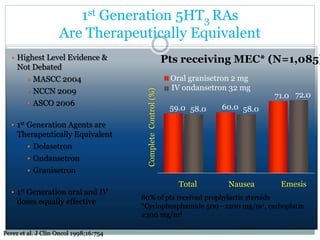
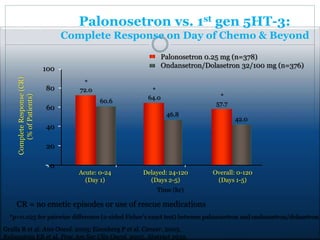
The document discusses the causes, diagnosis, and management of emesis (vomiting) in pediatric patients, highlighting differential diagnoses such as pyloric stenosis and acute appendicitis. It emphasizes the significance of recognizing gastrointestinal symptoms in children undergoing cancer treatment, with a focus on chemotherapy-induced nausea and vomiting (CINV). Additionally, it explores the efficacy of olanzapine as a preventive measure for CINV compared to traditional antiemetics, indicating its potential benefits for patient quality of life during treatment.