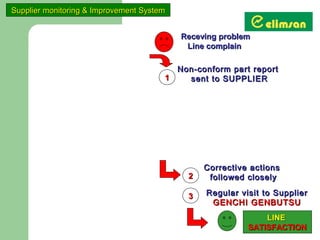
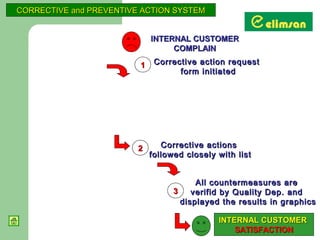
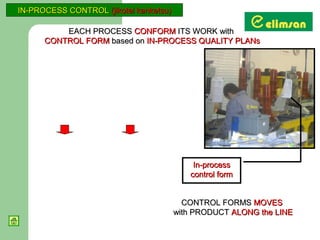
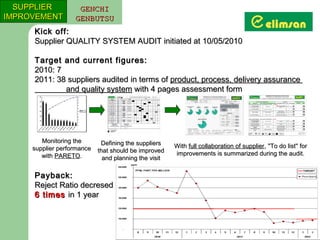
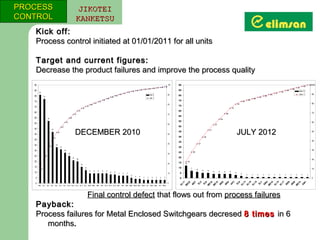
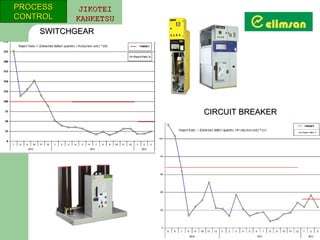
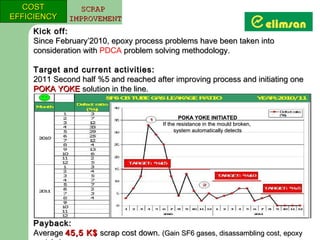
The document provides an overview of quality initiatives and performance metrics at Switchgear Equipments & Electromechanical Industry and Trade Inc. It discusses their quality core values and Elimsan Way approach, which focuses on standard work, parts, production, and process assurance. Metrics are provided tracking performance of their suggestion system, supplier improvement activities, process control, and other quality programs from 2010-2012, showing improvements in areas like cost reduction, scrap reduction, productivity increases, and more.