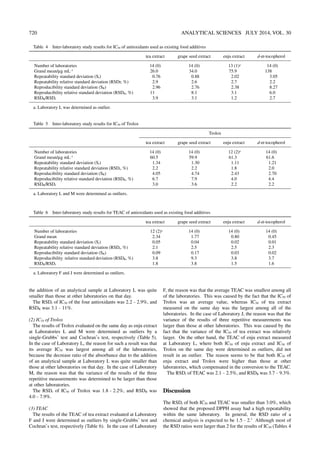
This document describes an inter-laboratory evaluation study conducted to validate the use of the 1,1-diphenyl-2-picrylhydrazyl (DPPH) assay as a standard method for evaluating the antioxidant capacity of food additives. Fourteen laboratories participated in the study. Five analytical samples - four existing food additives (tea extract, grape seed extract, enju extract, and d-α-tocopherol) and Trolox - were evaluated using the DPPH assay. The results showed good repeatability for measuring antioxidant capacity within laboratories, but greater variability between laboratories. However, expressing the results as Trolox equivalent antioxidant capacity reduced inter-laboratory variability, suggesting the DPPH assay could be