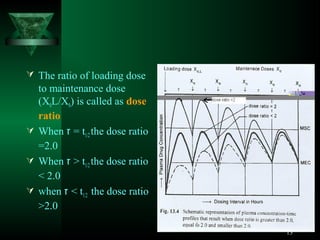
This document discusses dosage regimens for drugs administered through multiple doses. It defines key concepts like maintenance dose, loading dose, and accumulation index. The loading dose is given to quickly reach the steady state concentration, while the maintenance dose is used to maintain that concentration by replacing eliminated drug. The accumulation index describes the extent of drug accumulation in the body during multiple dosing as a function of dosing interval and elimination half-life. Designing optimal dosage regimens requires considering factors like dose size, dosing frequency, and pharmacokinetic parameters.