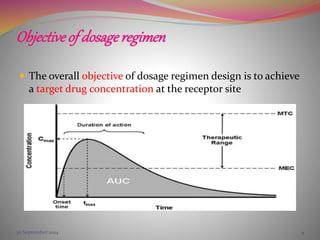
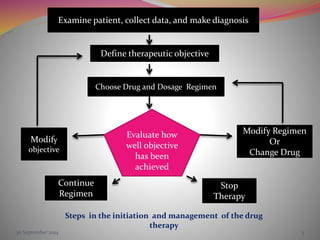
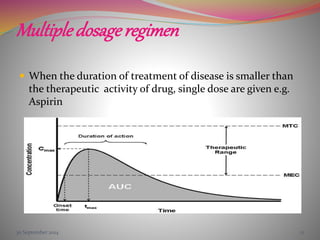
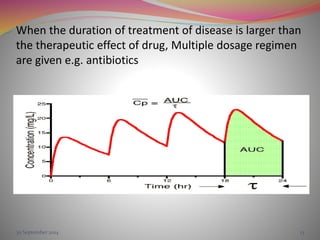
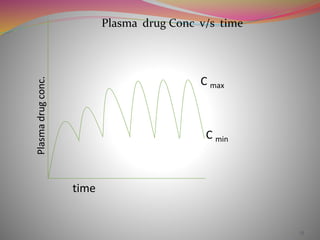
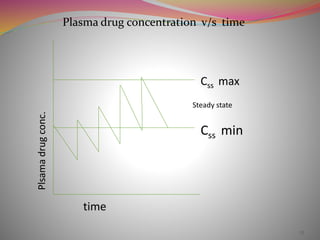
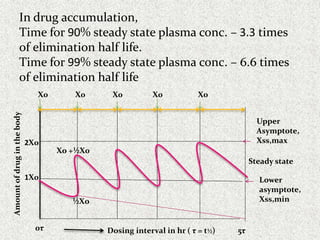
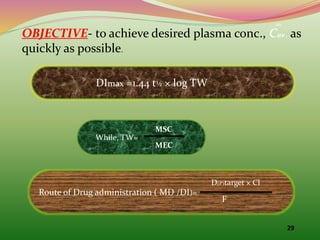
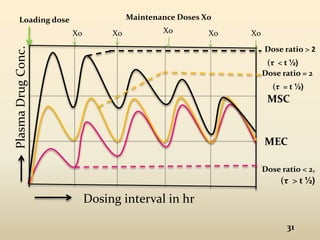
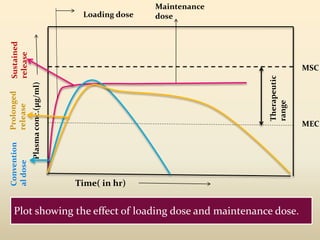
The document discusses pharmacokinetics of multiple dosing. It defines dosage regimen as how a drug is taken when treatment duration is longer than a single dose's therapeutic effect. The objectives of dosage regimen design are to achieve target drug concentrations. Multiple dosing is used when treatment duration is larger than a single dose's effect, leading to drug accumulation. Loading and maintenance doses are discussed to quickly reach the desired plasma concentration and maintain it.