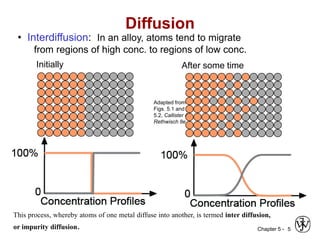
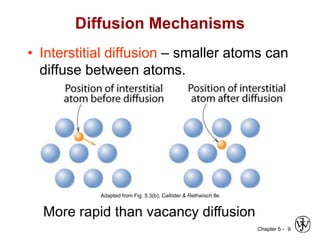
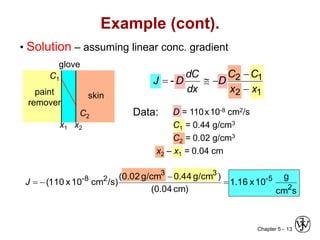
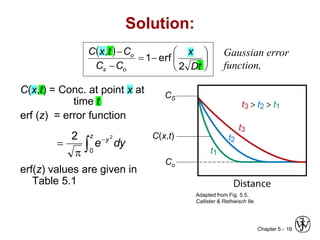
Diffusion is the mass transport of atoms through a solid by atomic motion. There are two main mechanisms: vacancy diffusion, where atoms exchange with vacancies in the lattice, and interstitial diffusion, where smaller atoms diffuse through spaces in the lattice. The rate of diffusion increases exponentially with temperature according to an Arrhenius relationship and is quantified by Fick's laws of diffusion. Diffusion plays an important role in many materials processes and semiconductor device fabrication.












![Chapter 5 - 19
Diffusion and Temperature
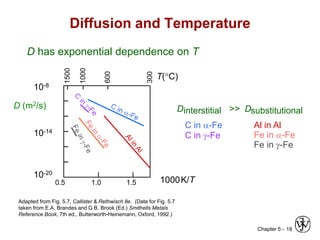
• Diffusion coefficient increases with increasing T.
D Do exp
Qd
RT
= pre-exponential [m2/s]
= diffusion coefficient [m2/s]
= activation energy [J/mol or eV/atom]
= gas constant [8.314 J/mol-K]
= absolute temperature [K]
D
Do
Qd
R
T](https://image.slidesharecdn.com/diffusionfinalppt-220828085320-a7b03c5e/85/diffusion-final-PPT-ppt-19-320.jpg)



