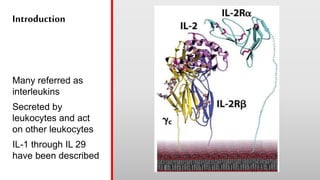
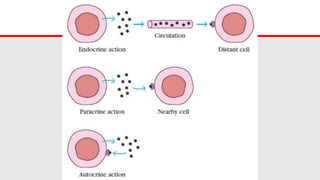
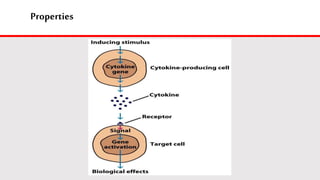
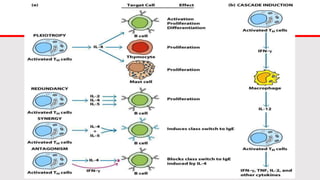
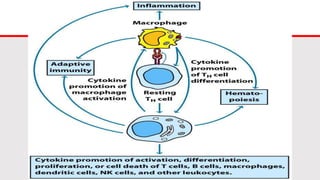
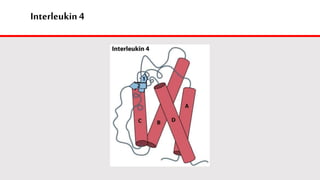
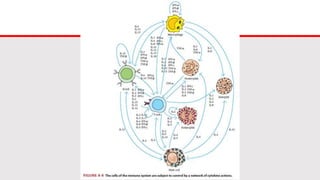
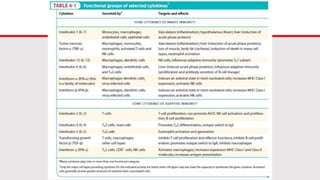
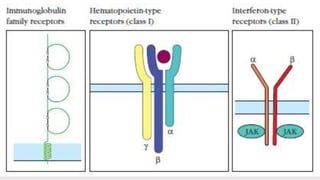
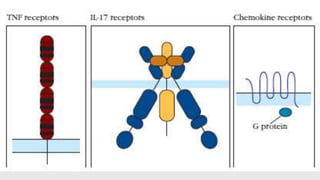
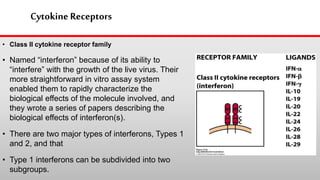
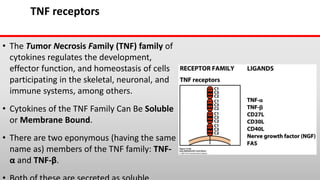
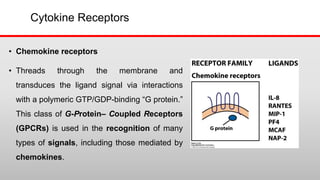
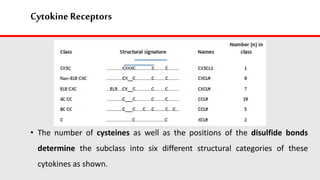
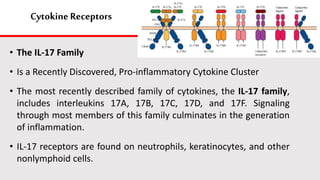
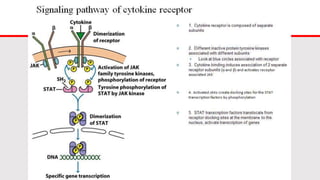
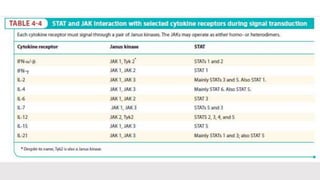
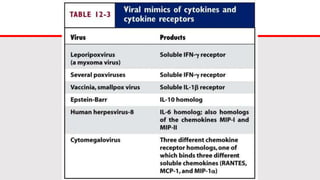
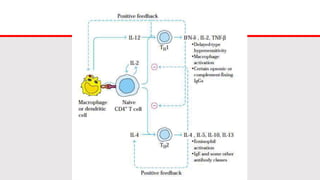
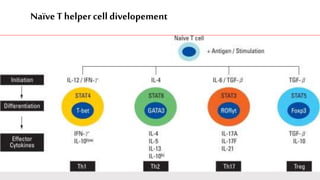
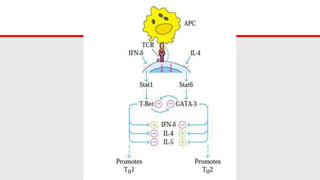
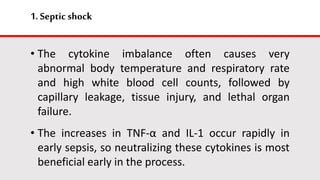
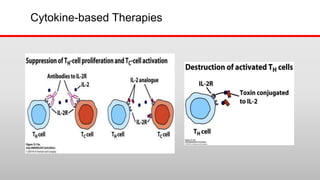
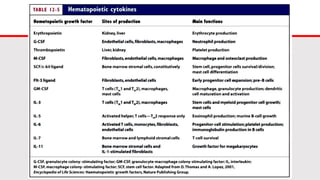
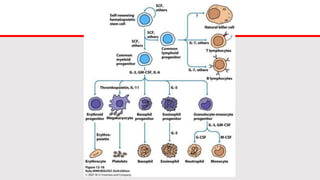
This document discusses cytokines, which are low molecular weight proteins that mediate communication between cells of the immune system. Cytokines are secreted by white blood cells and other cells to regulate immune responses. They belong to four main families and signal to target cells by binding specific receptors. Cytokines play roles in processes like immune cell development, inflammation, hematopoiesis, and wound healing. Their effects are targeted through selective receptor expression and localized secretion between interacting cells.