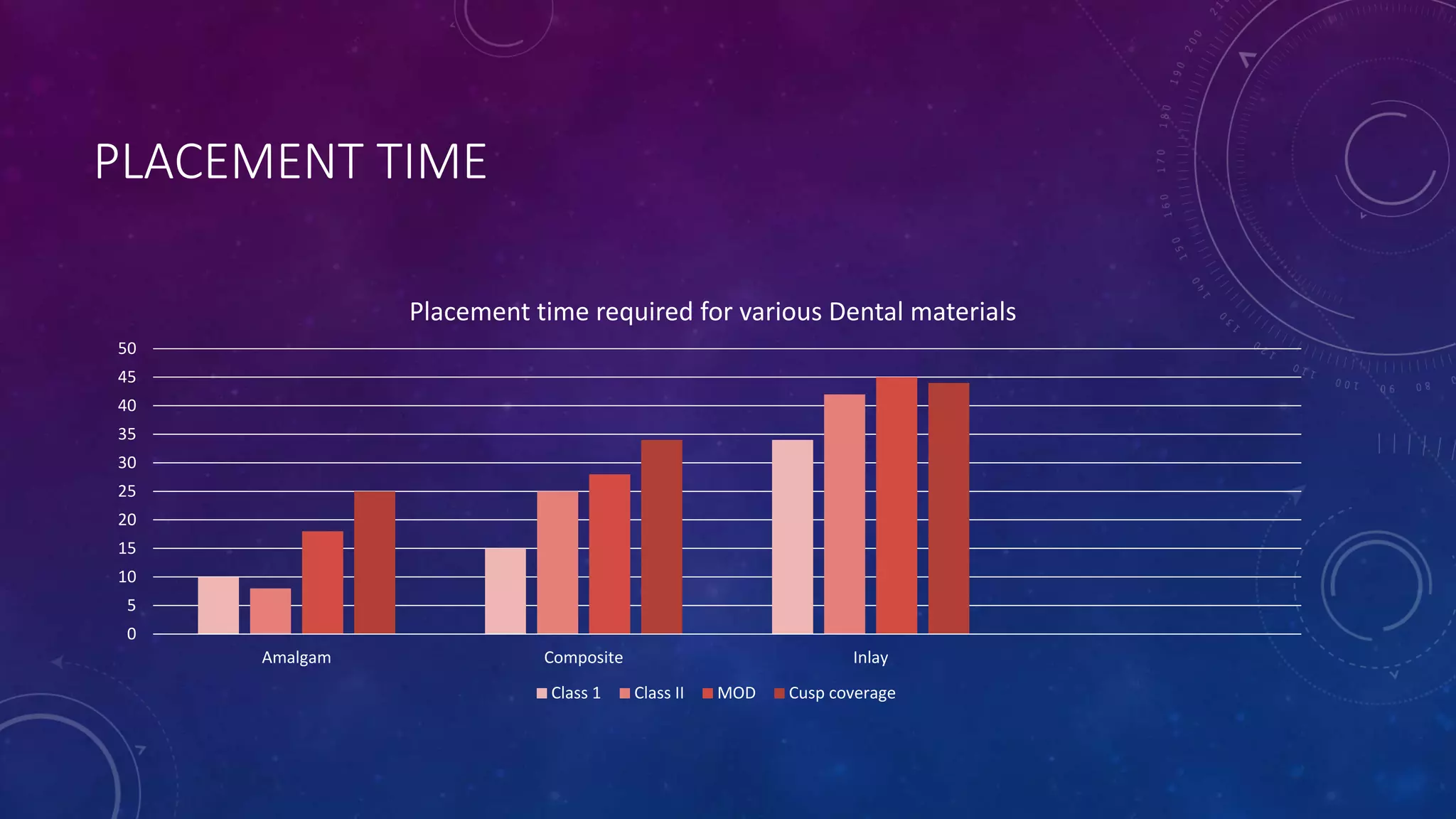
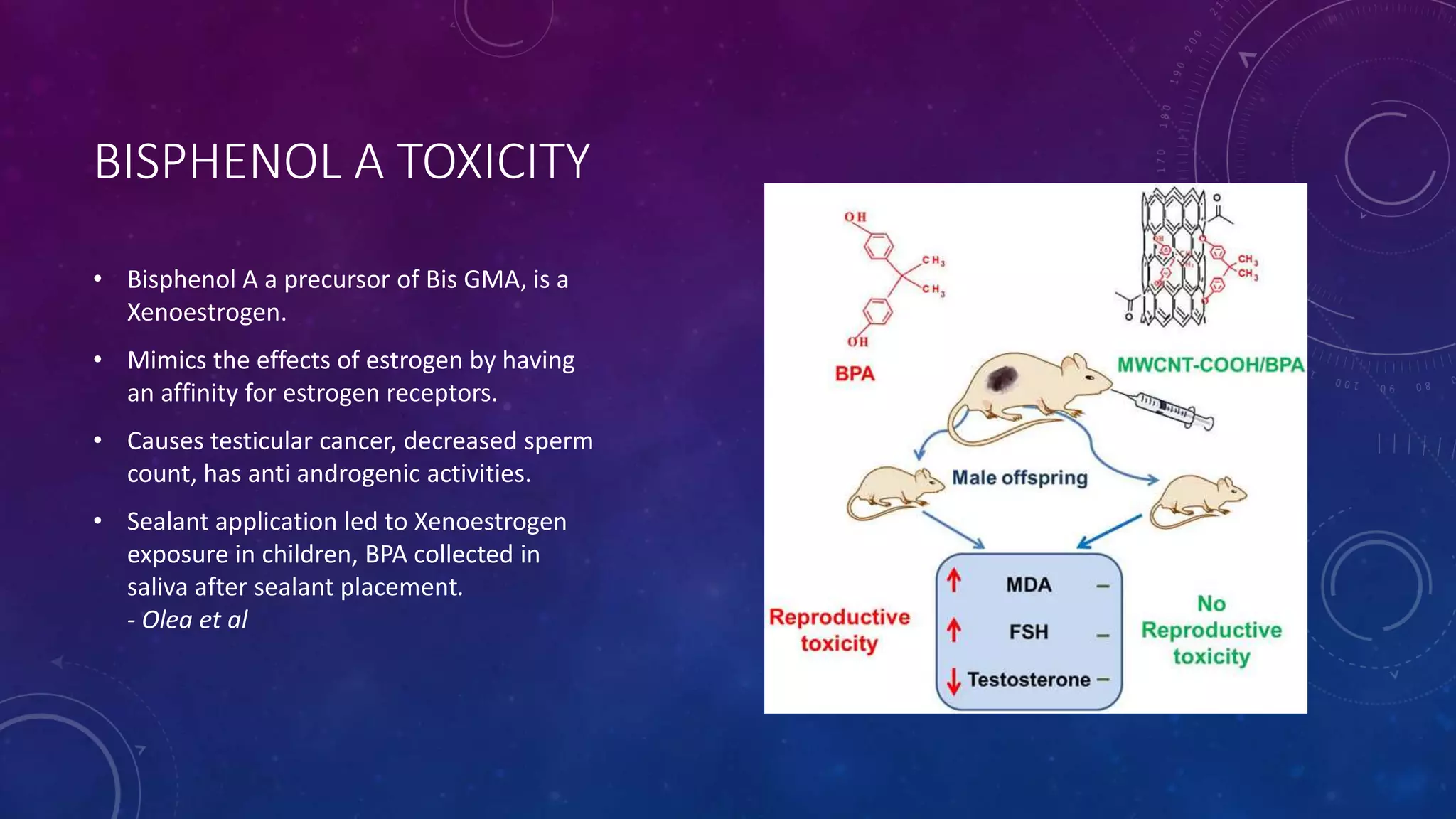
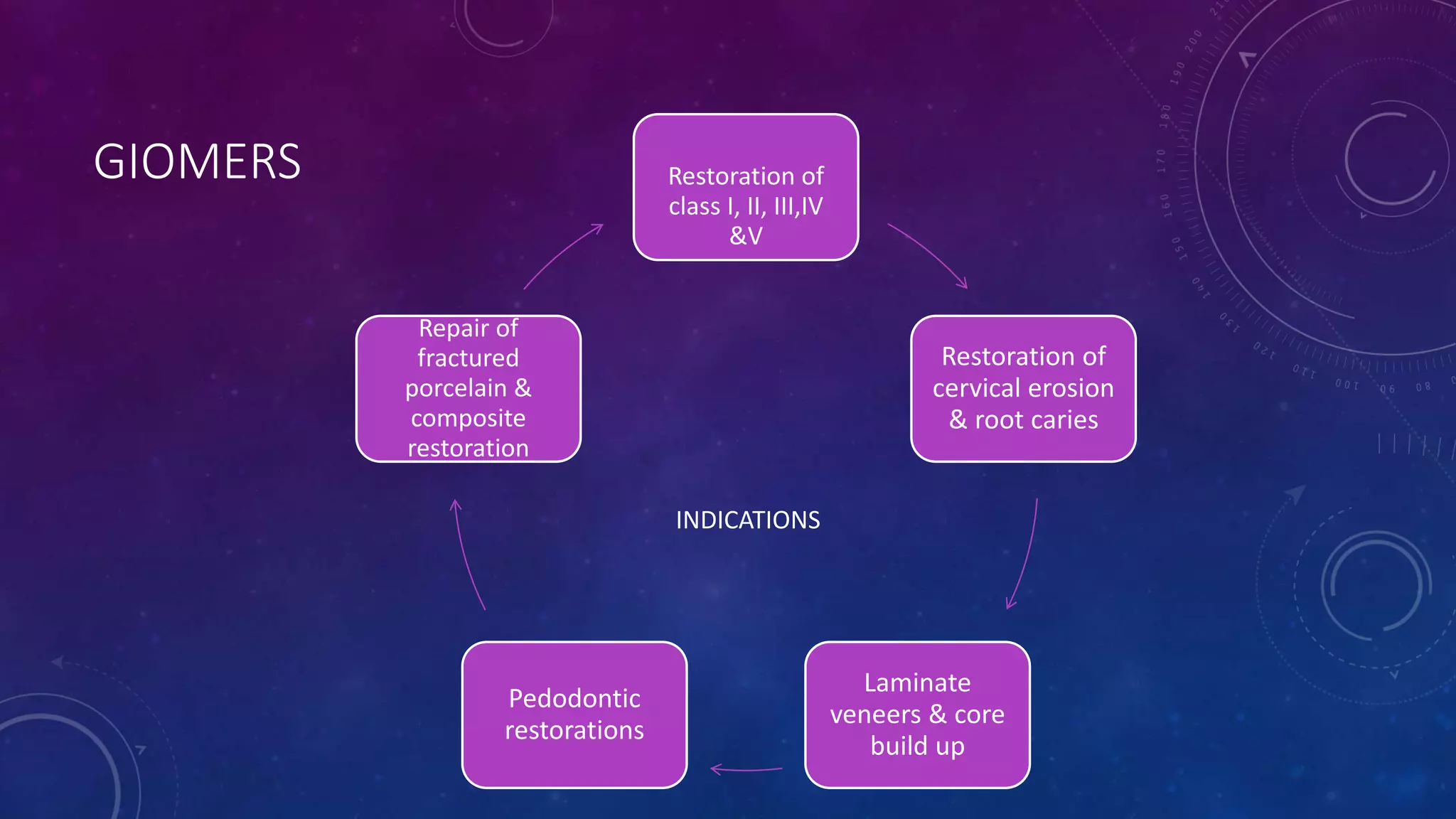
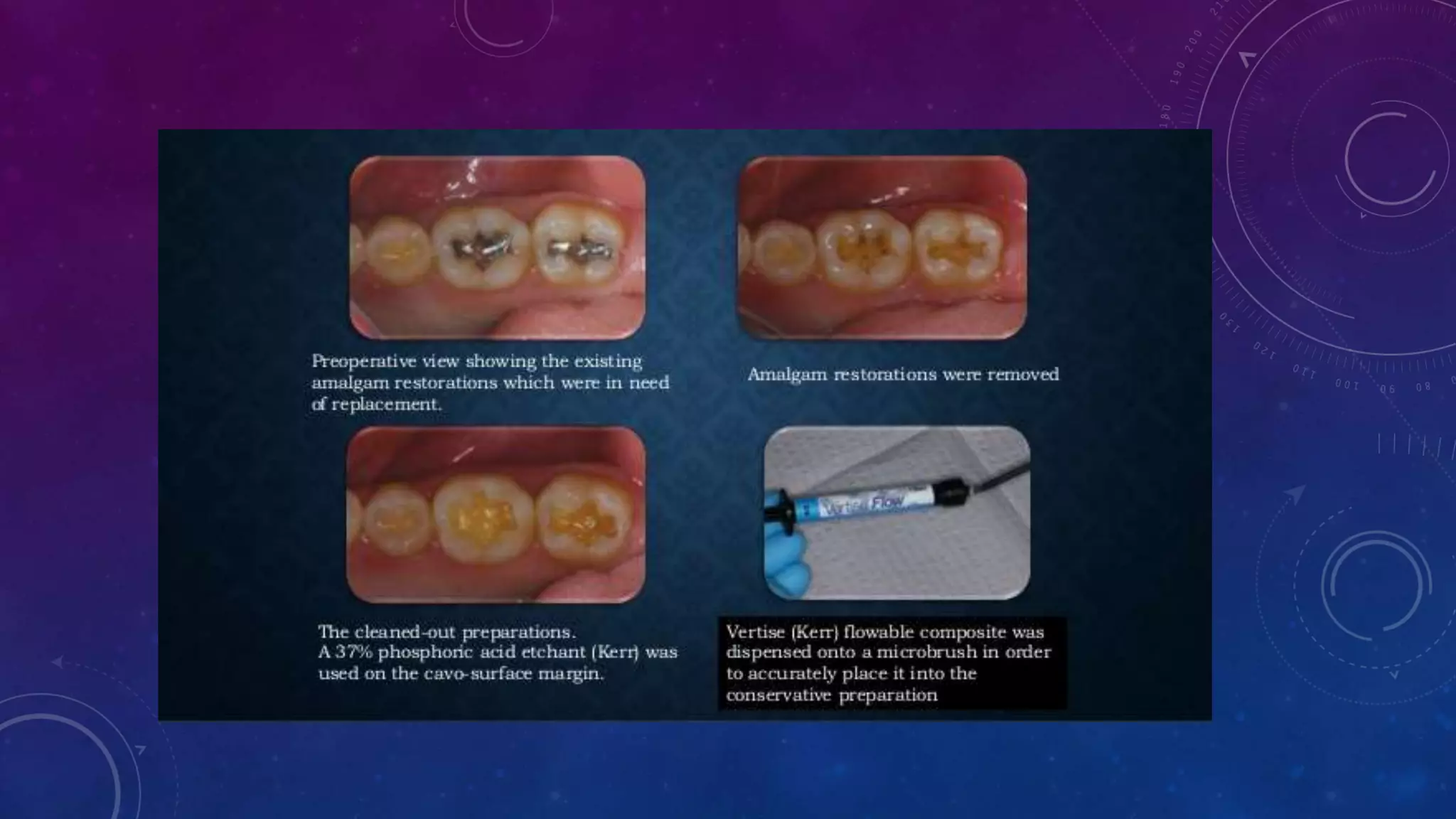
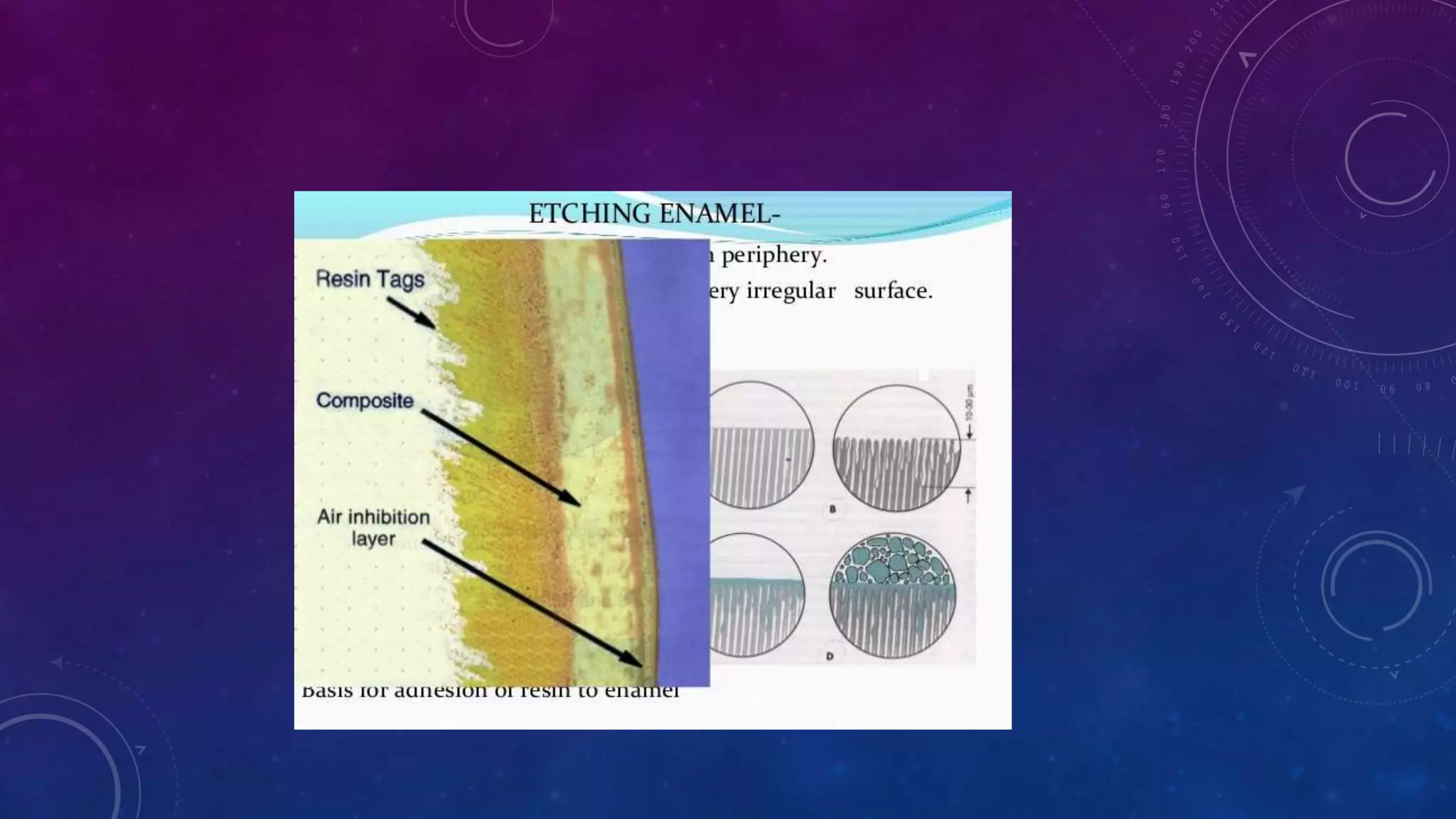
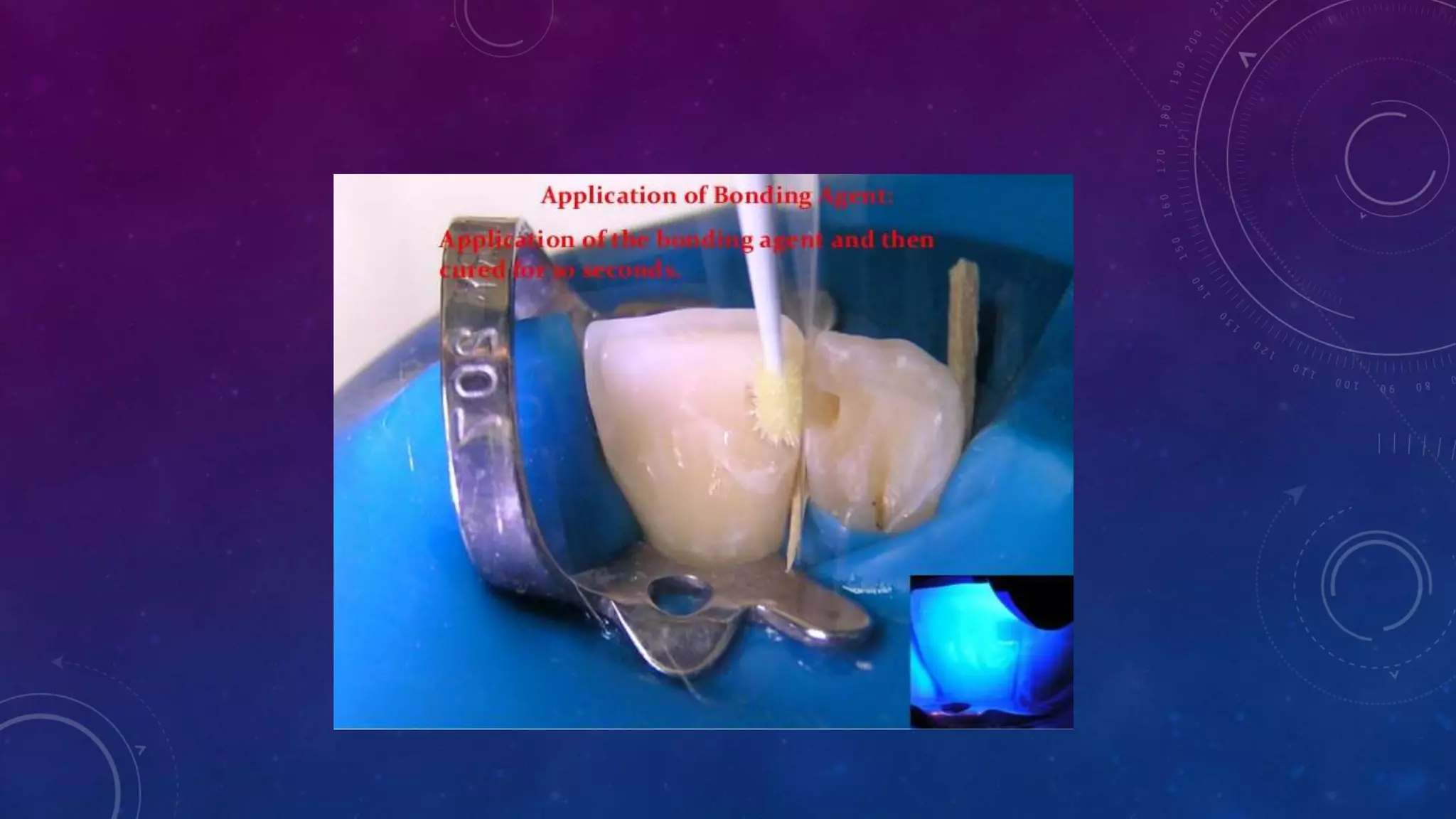
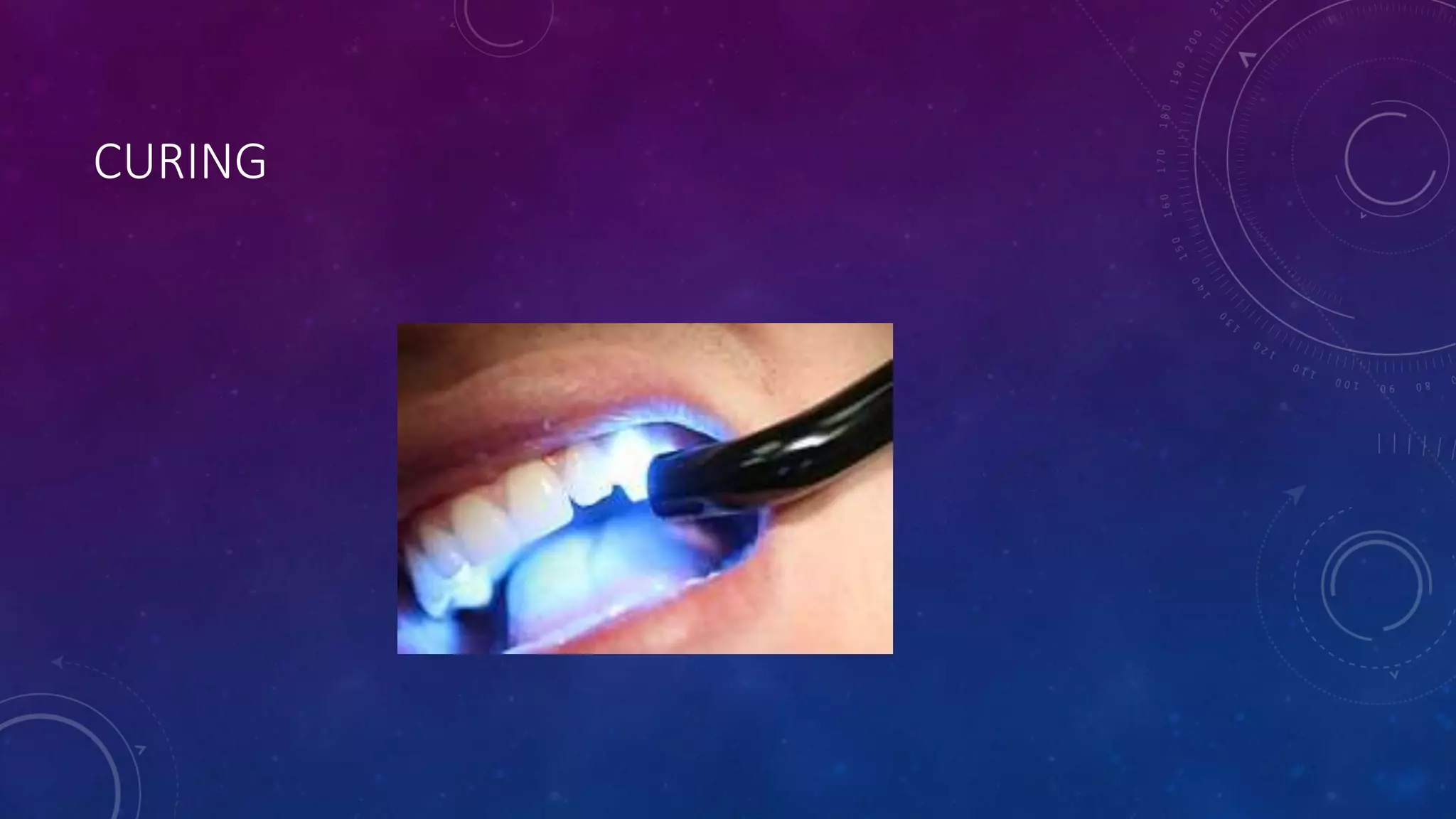
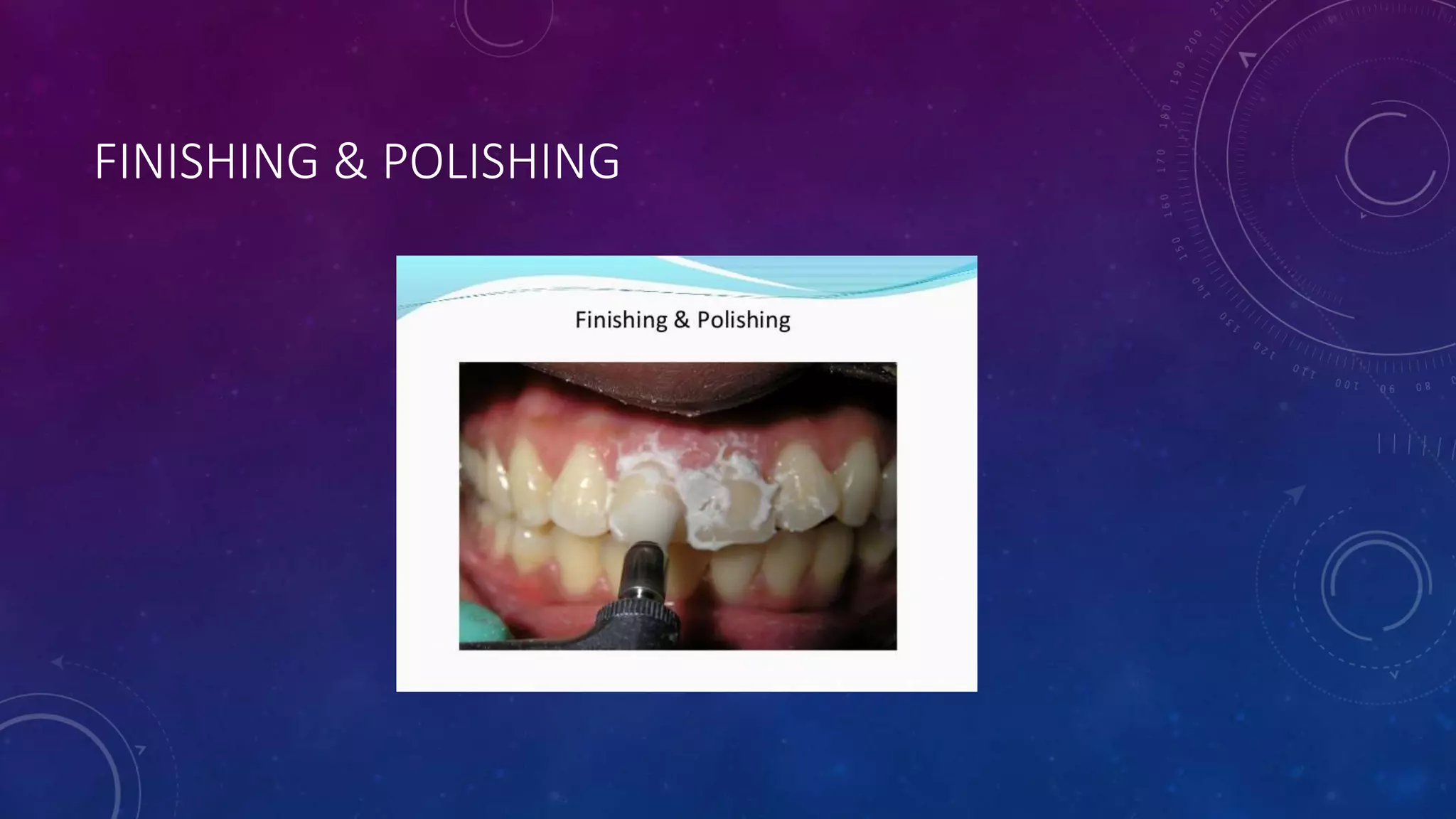
Composite resins are composed of three main components: a cross-linked polymeric matrix, coupling agents, and glass/resin fillers. They have evolved from macrofilled composites in the 1960s to modern nanofilled and self-adhesive composites. Composites are classified based on filler size and type, as well as their intended application area. Photocuring units such as LED lamps are used to activate the initiator-accelerator system and initiate polymerization. Properties such as degree of conversion, matrix constraint, and wear resistance are influenced by factors like filler loading and size. Recent advancements continue to improve longevity, placement time, and biocompatibility of these resin-based materials.