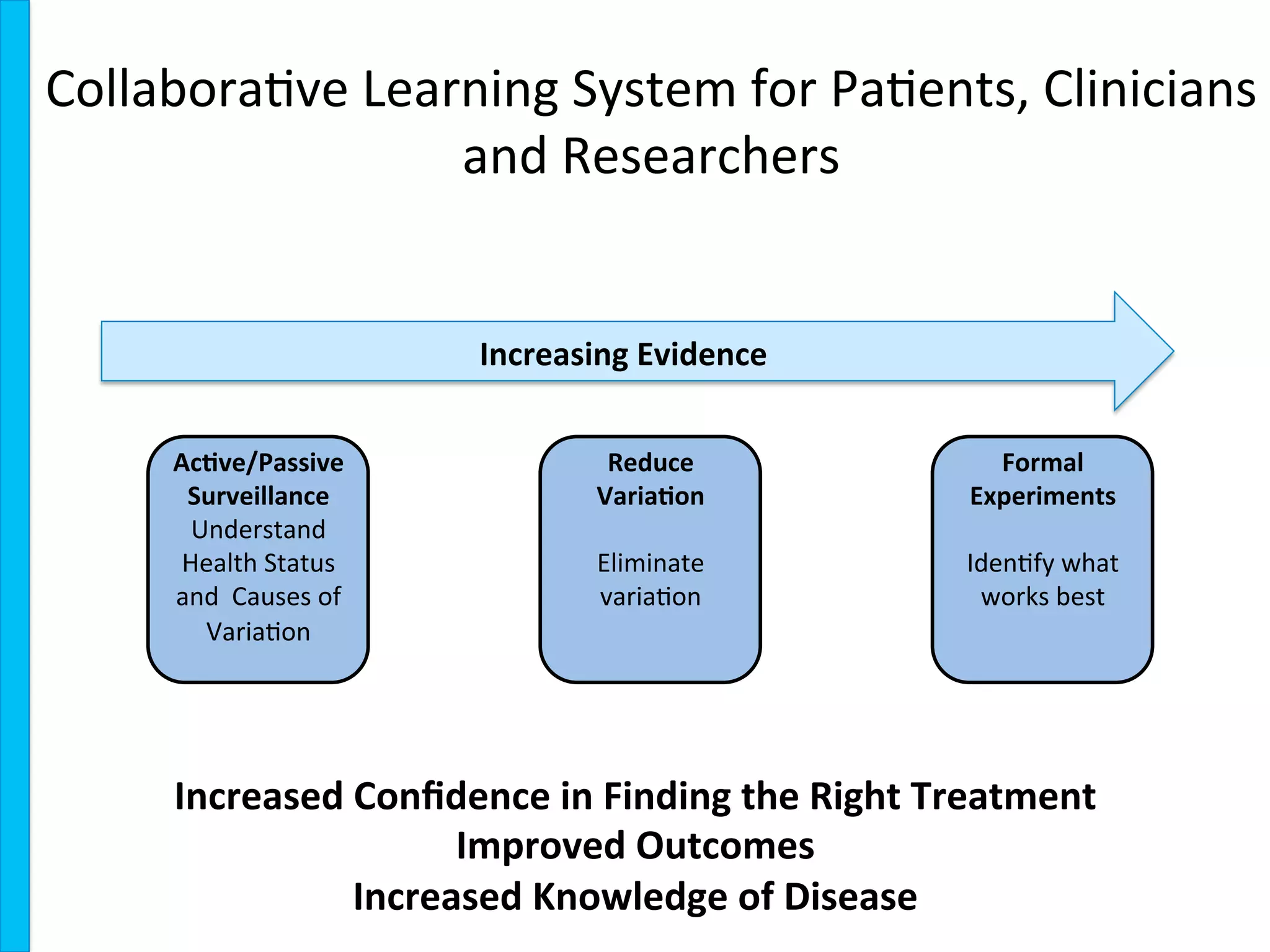
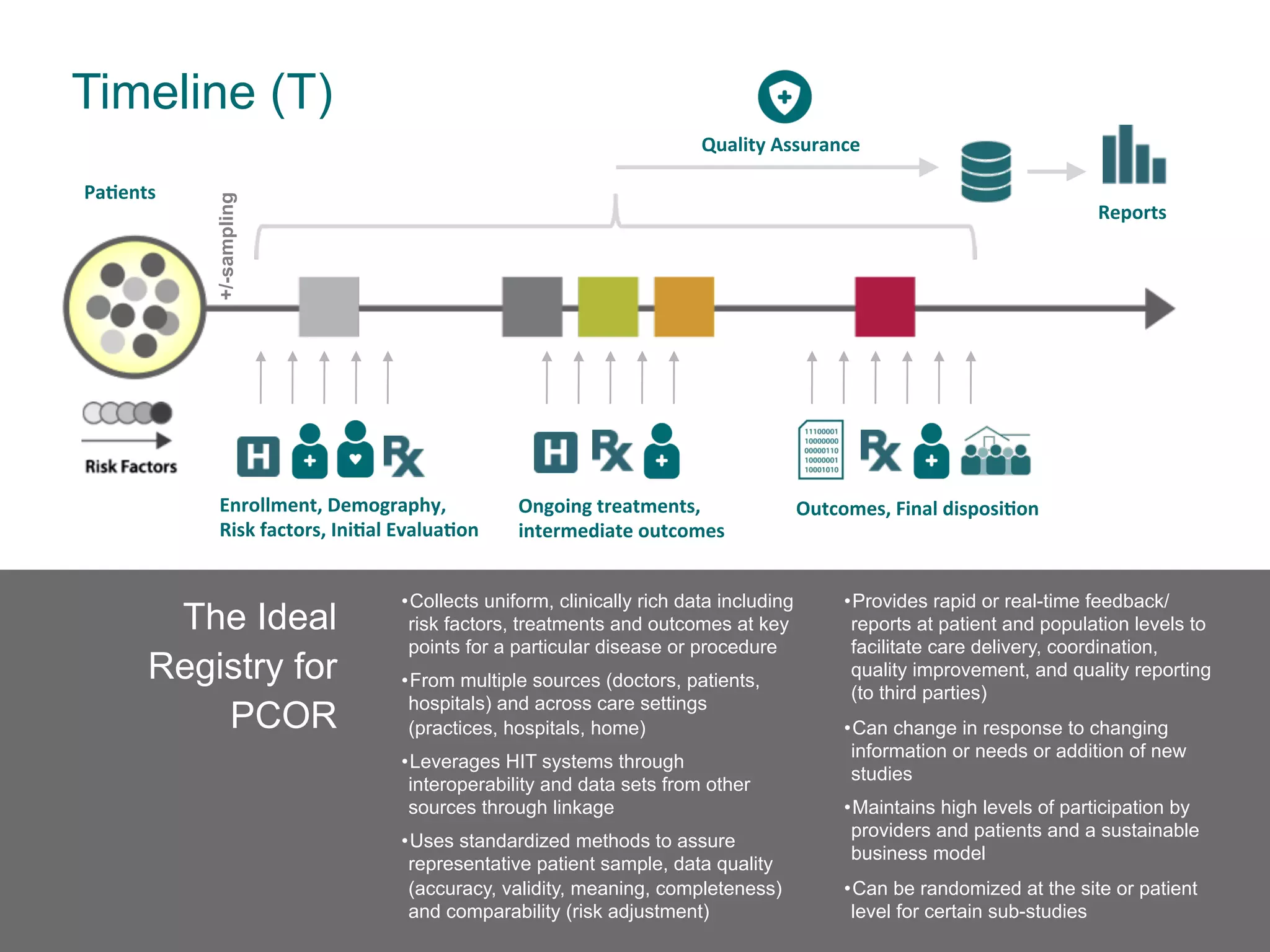
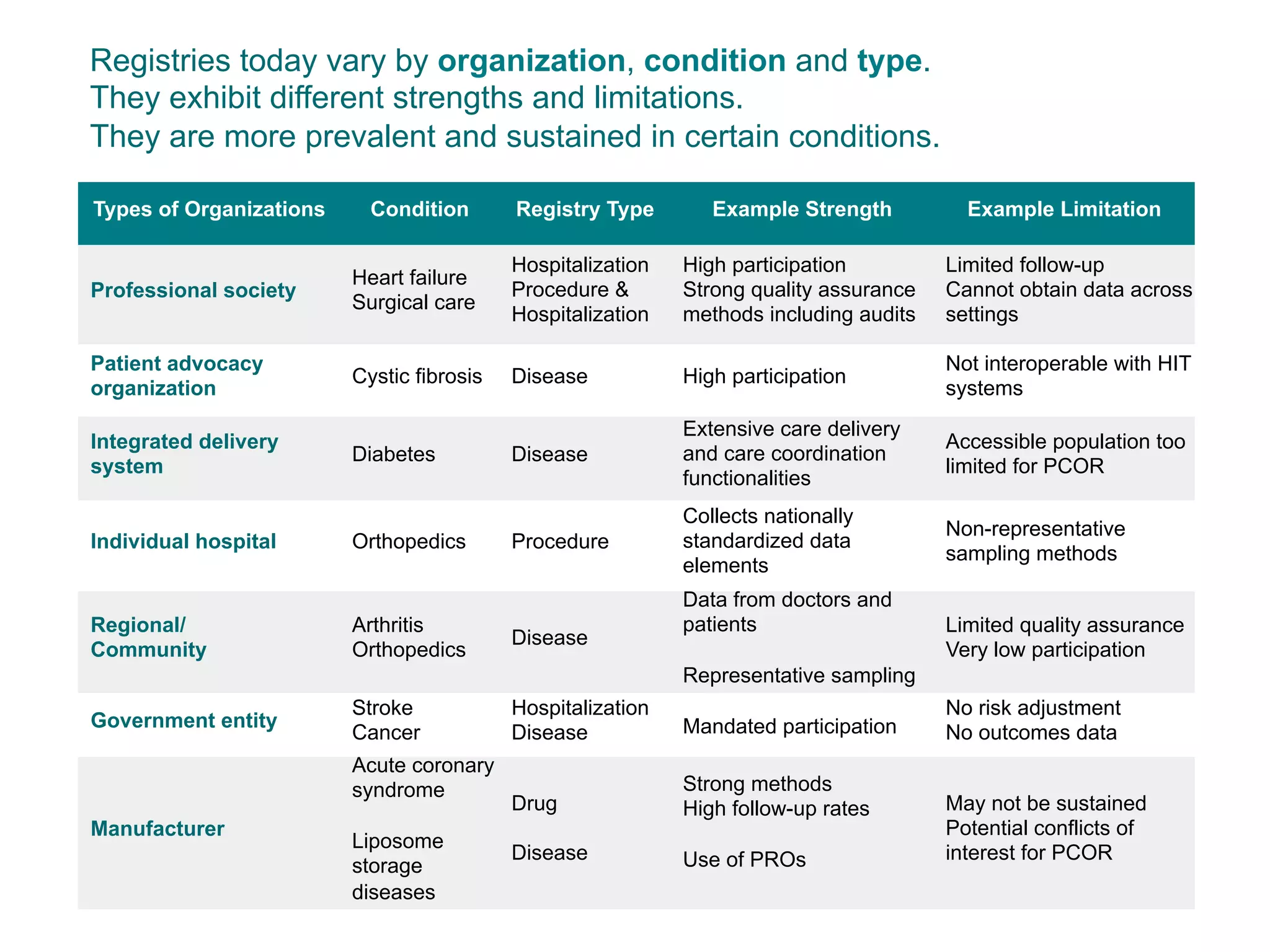
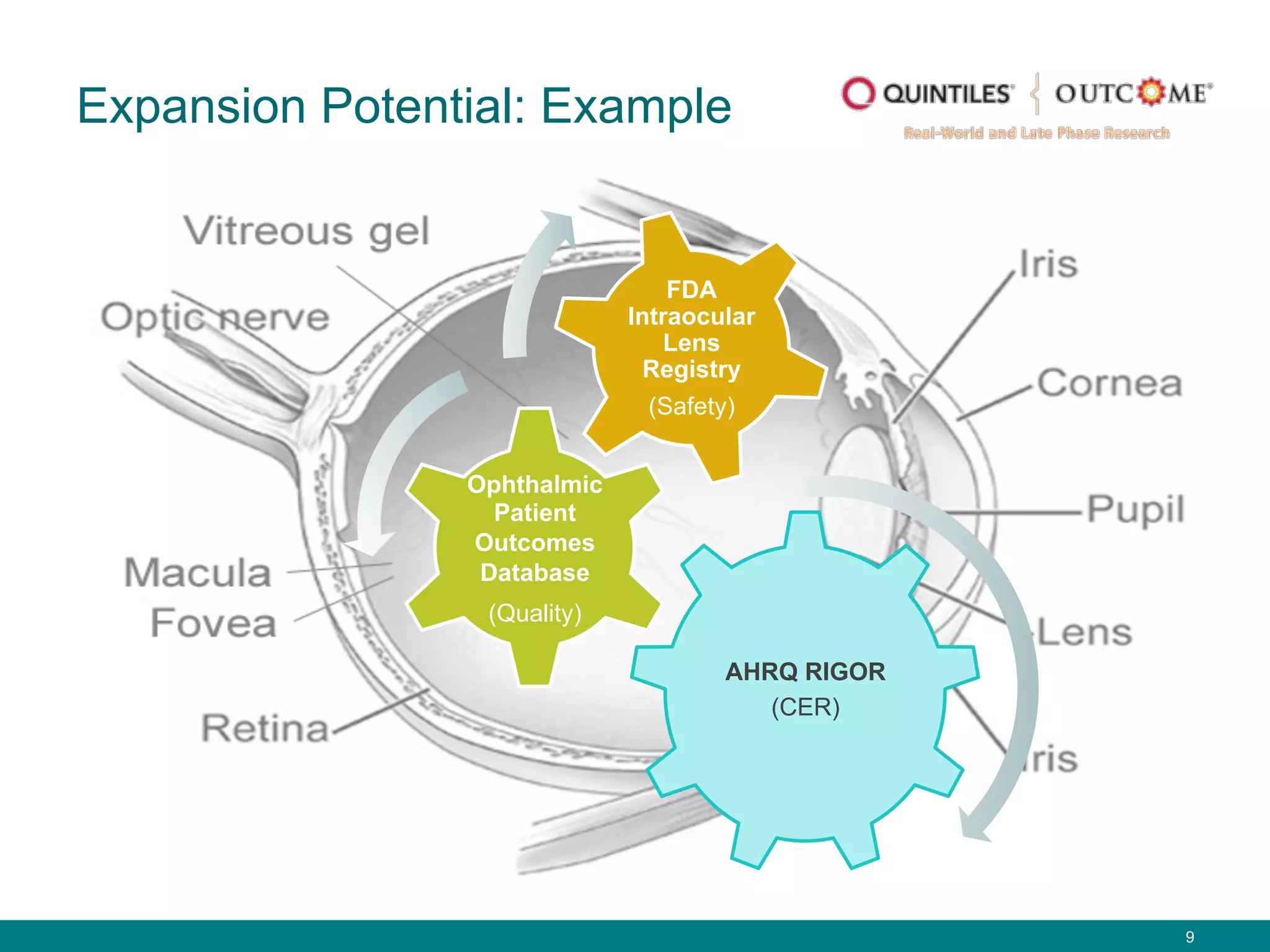
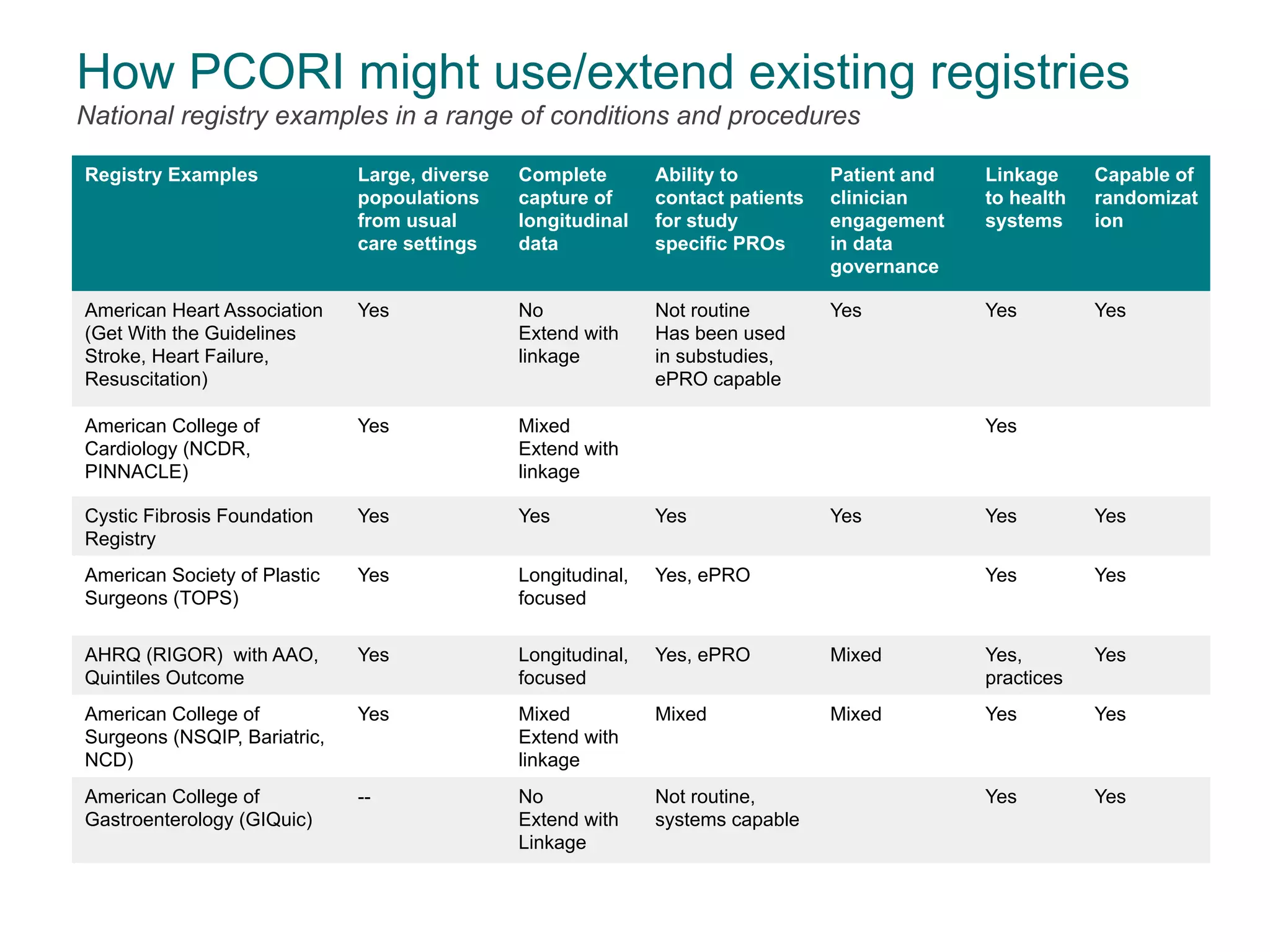
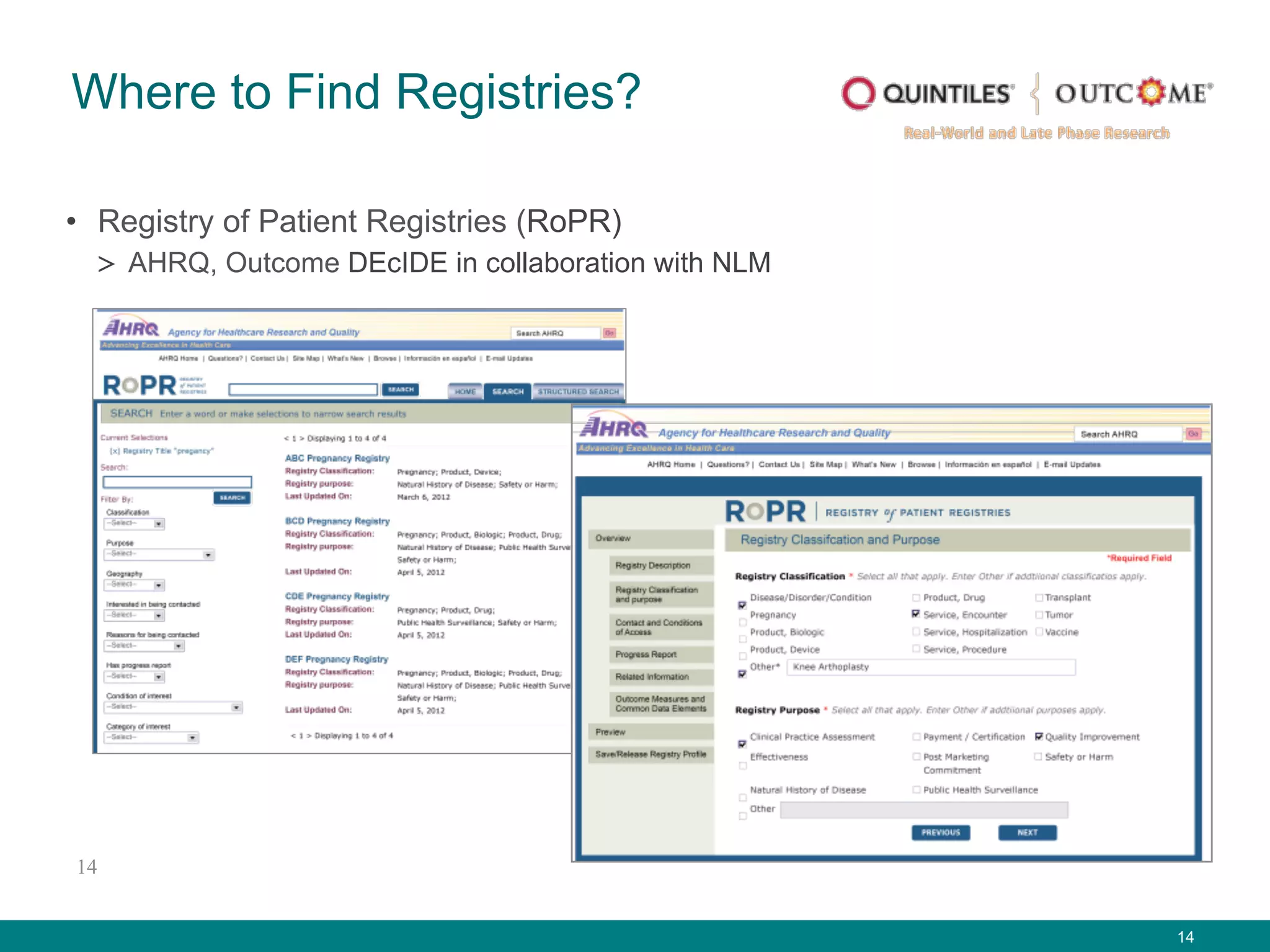
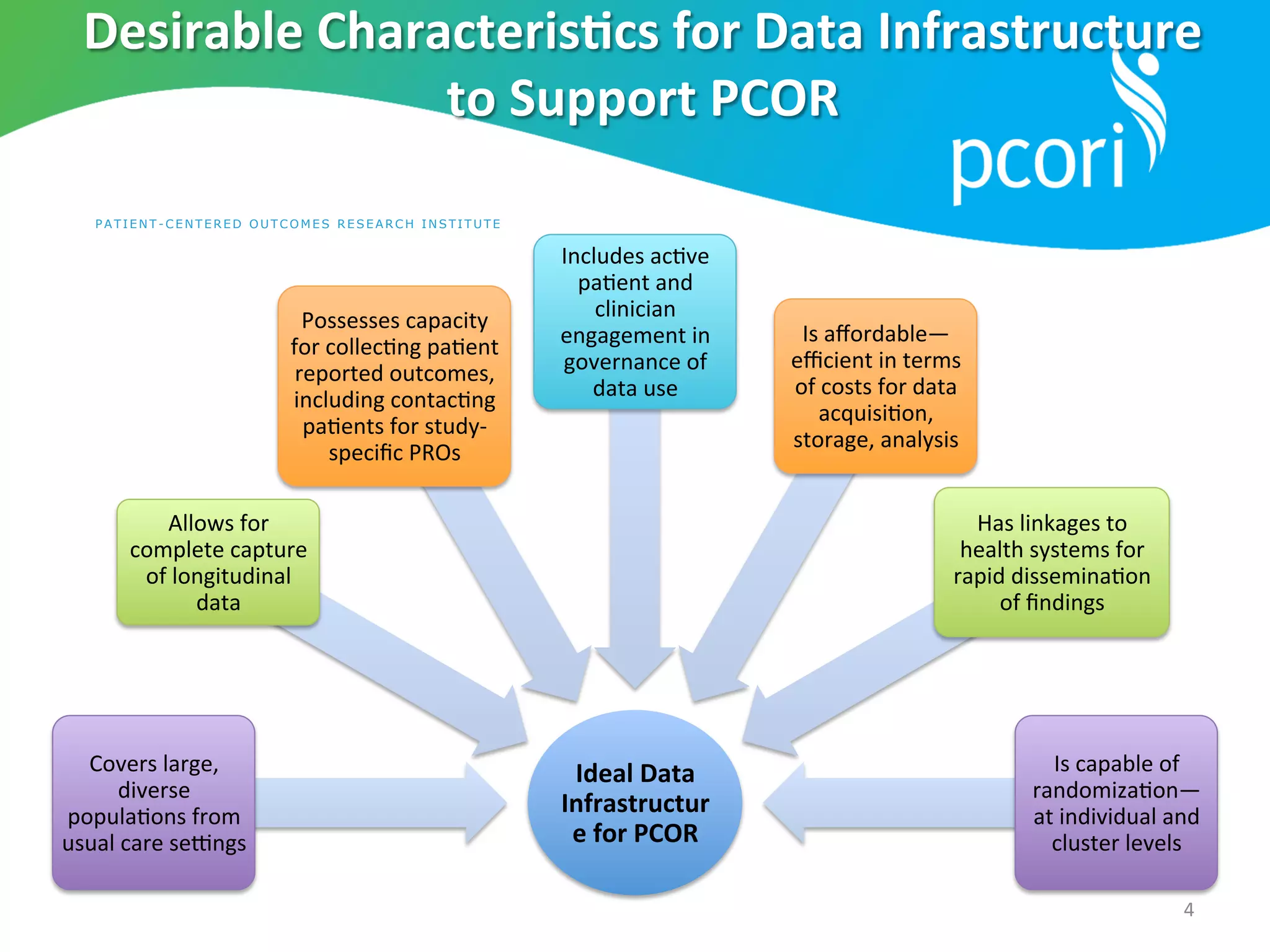
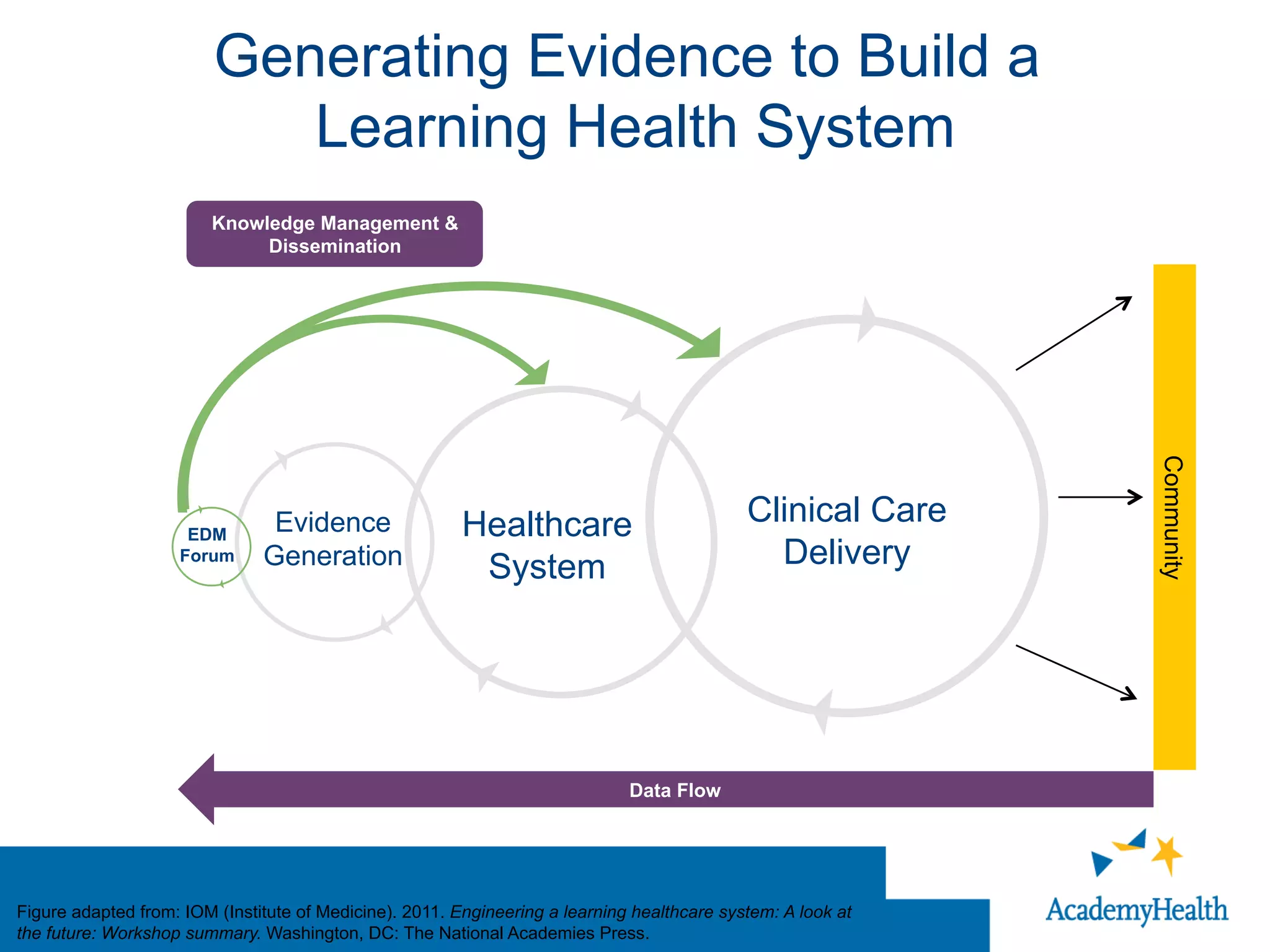
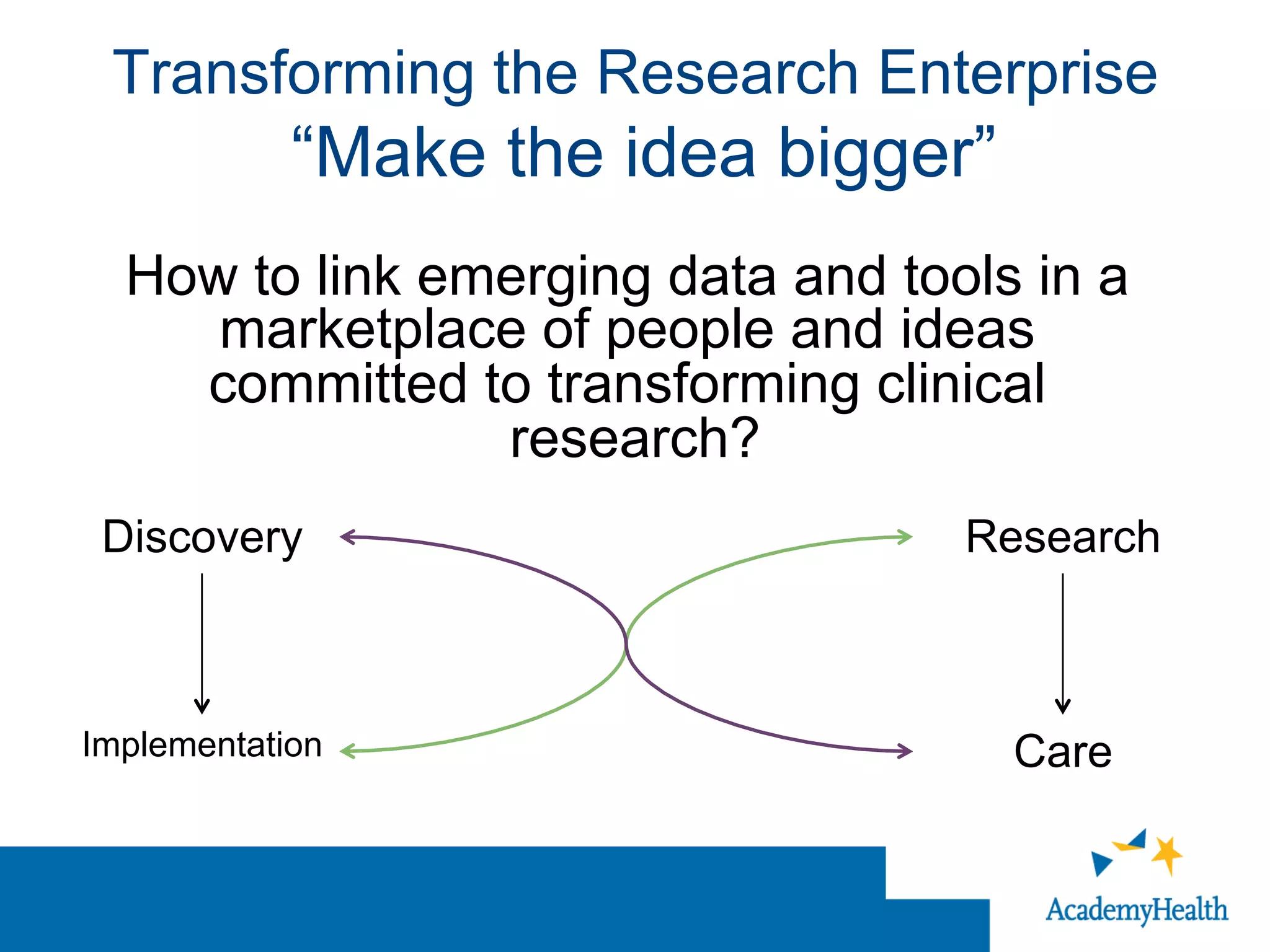
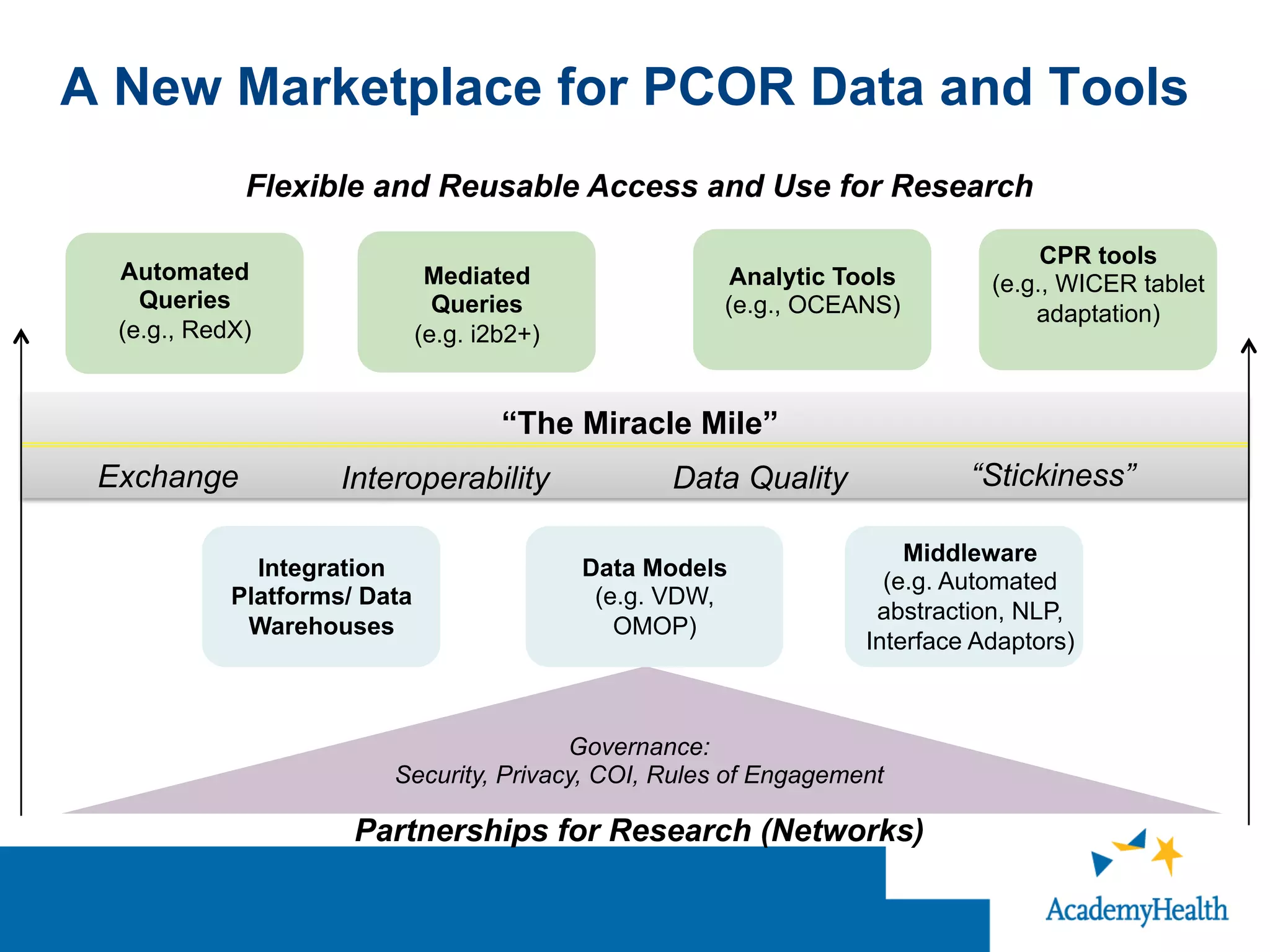
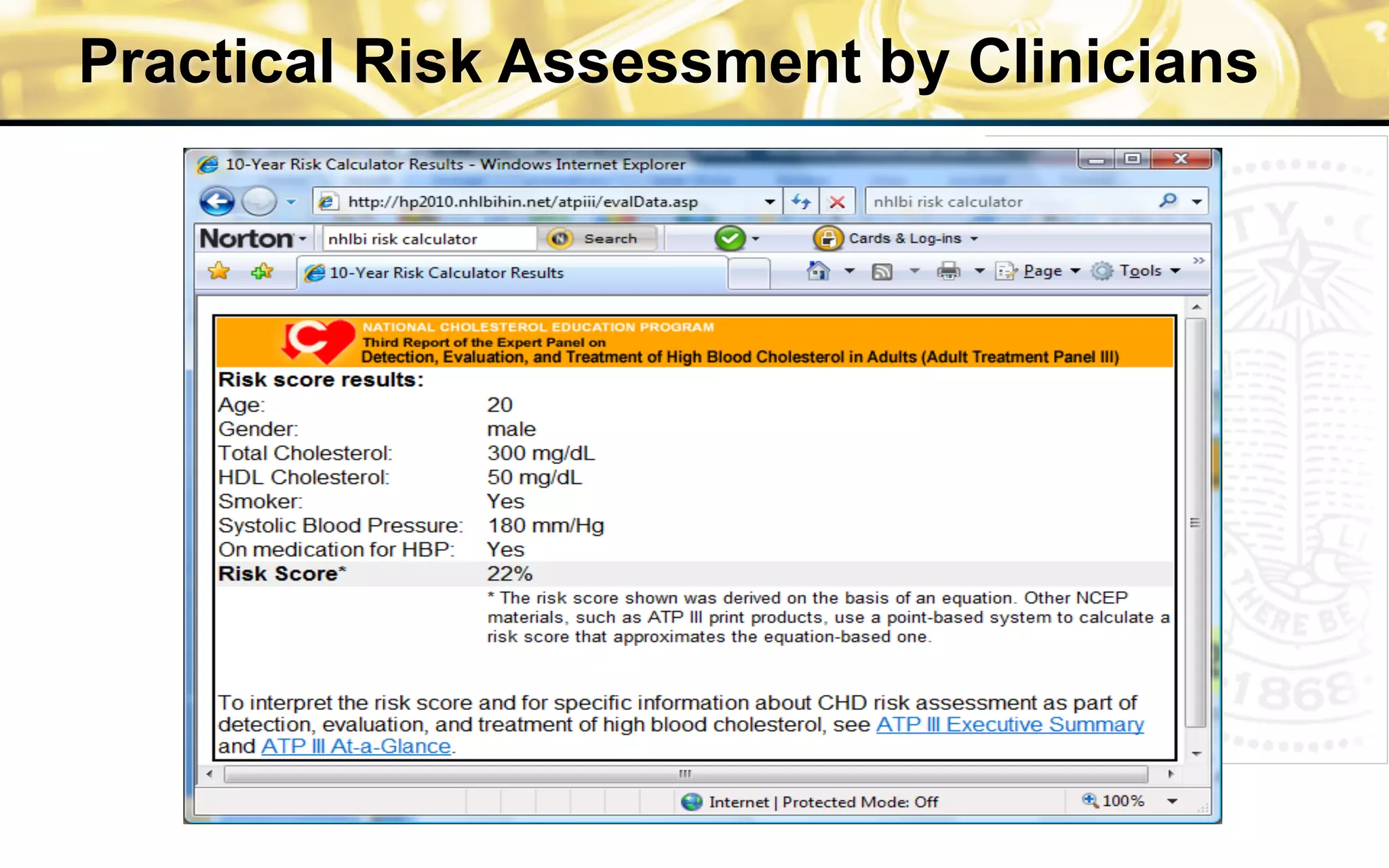
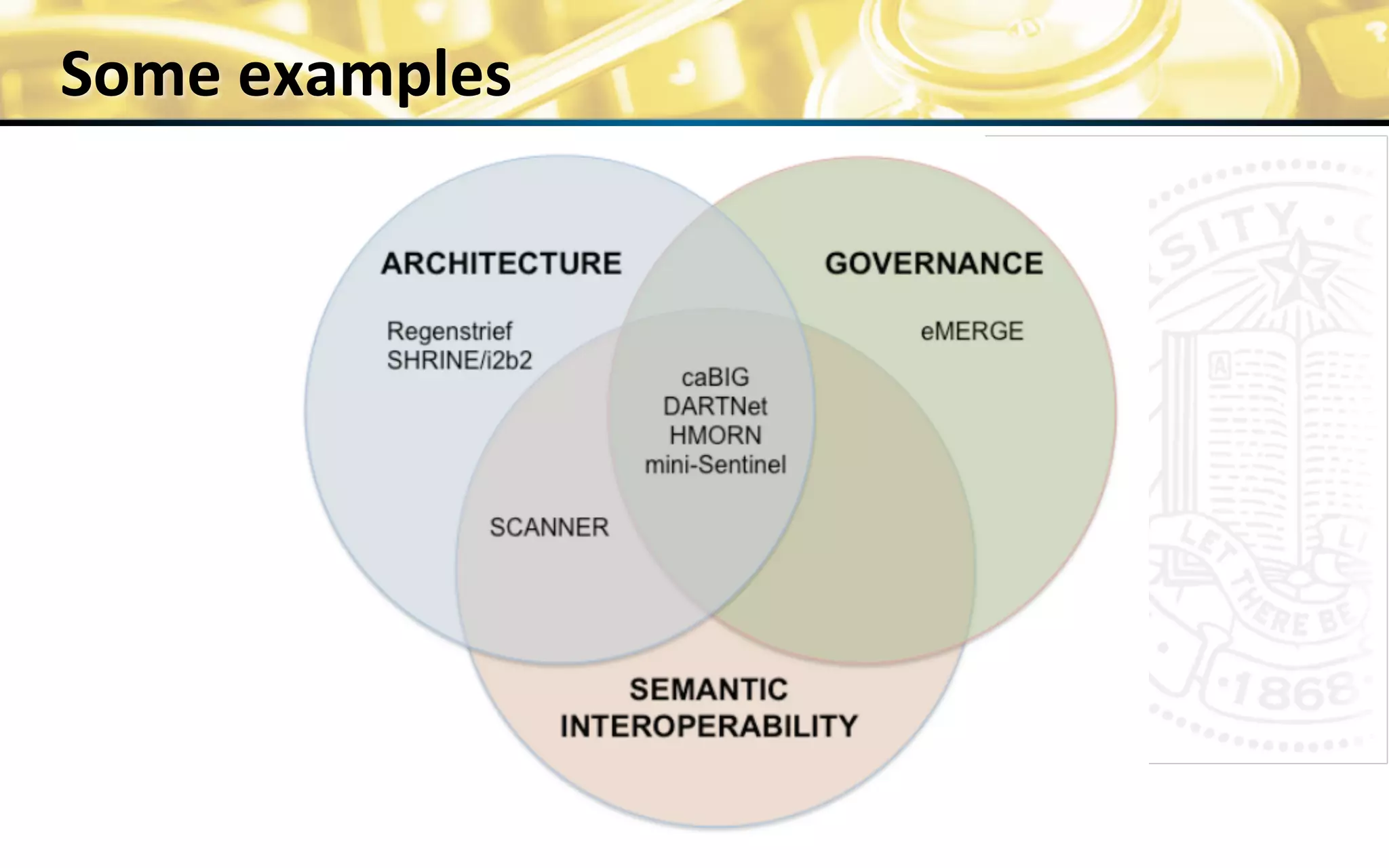
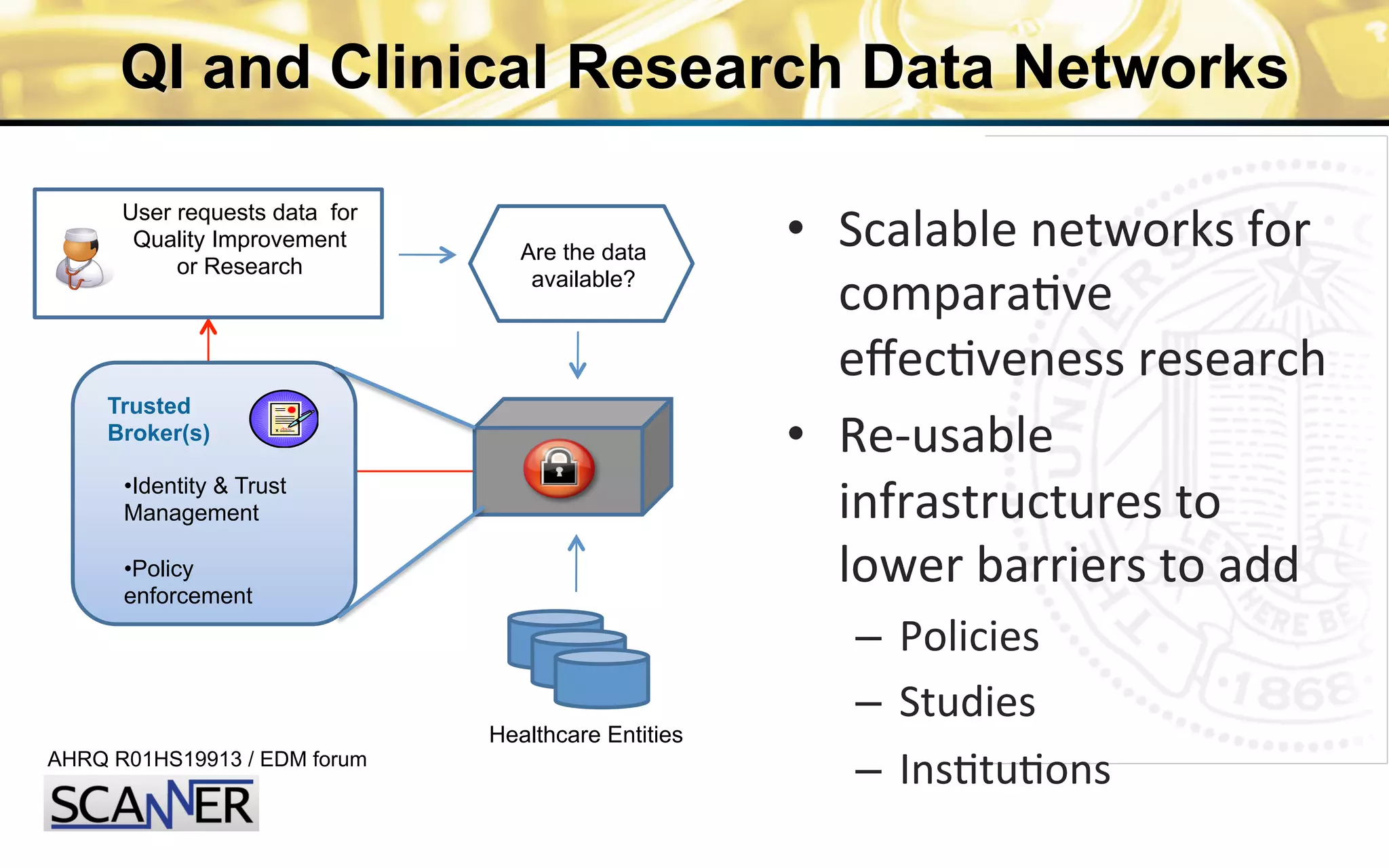
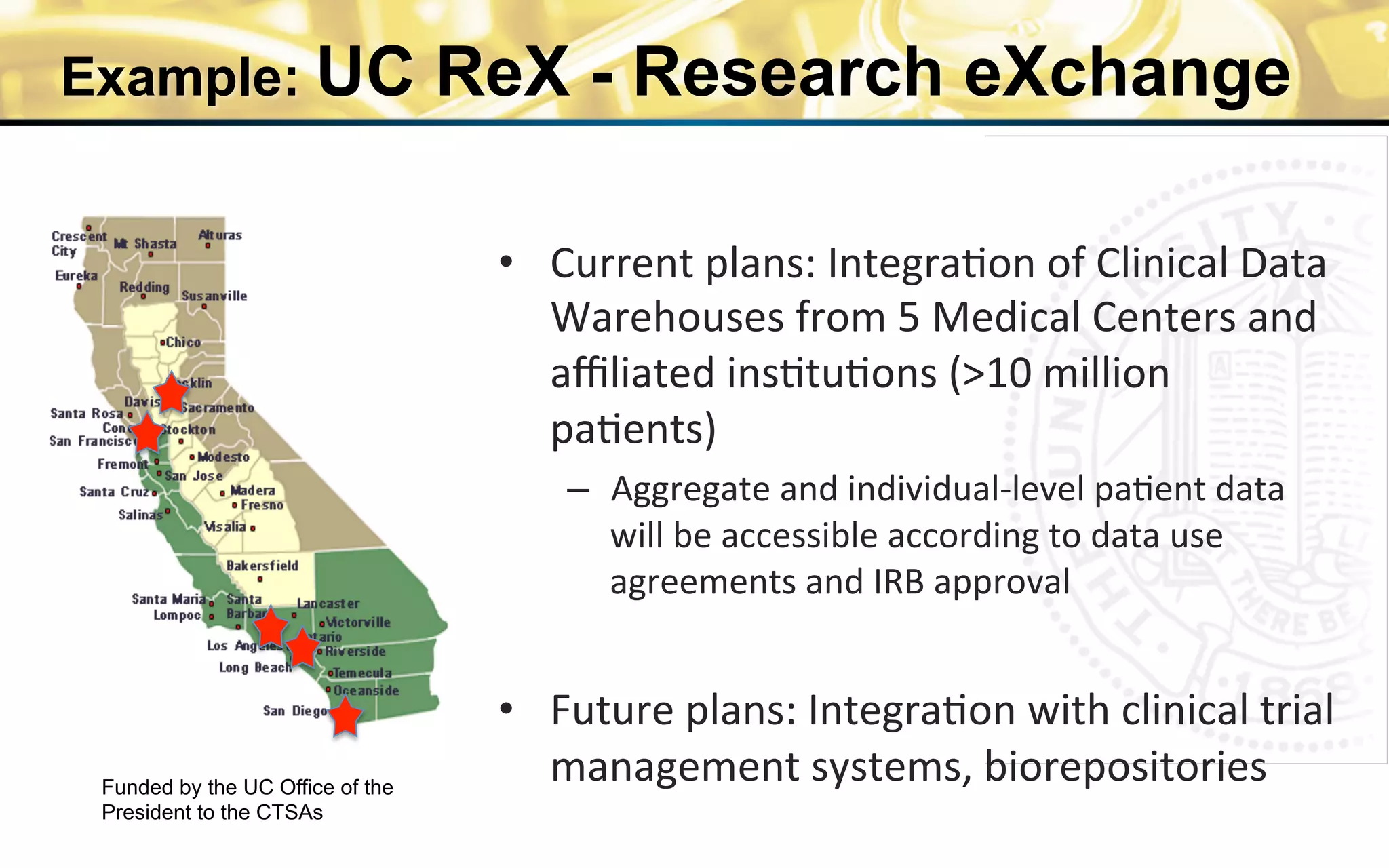
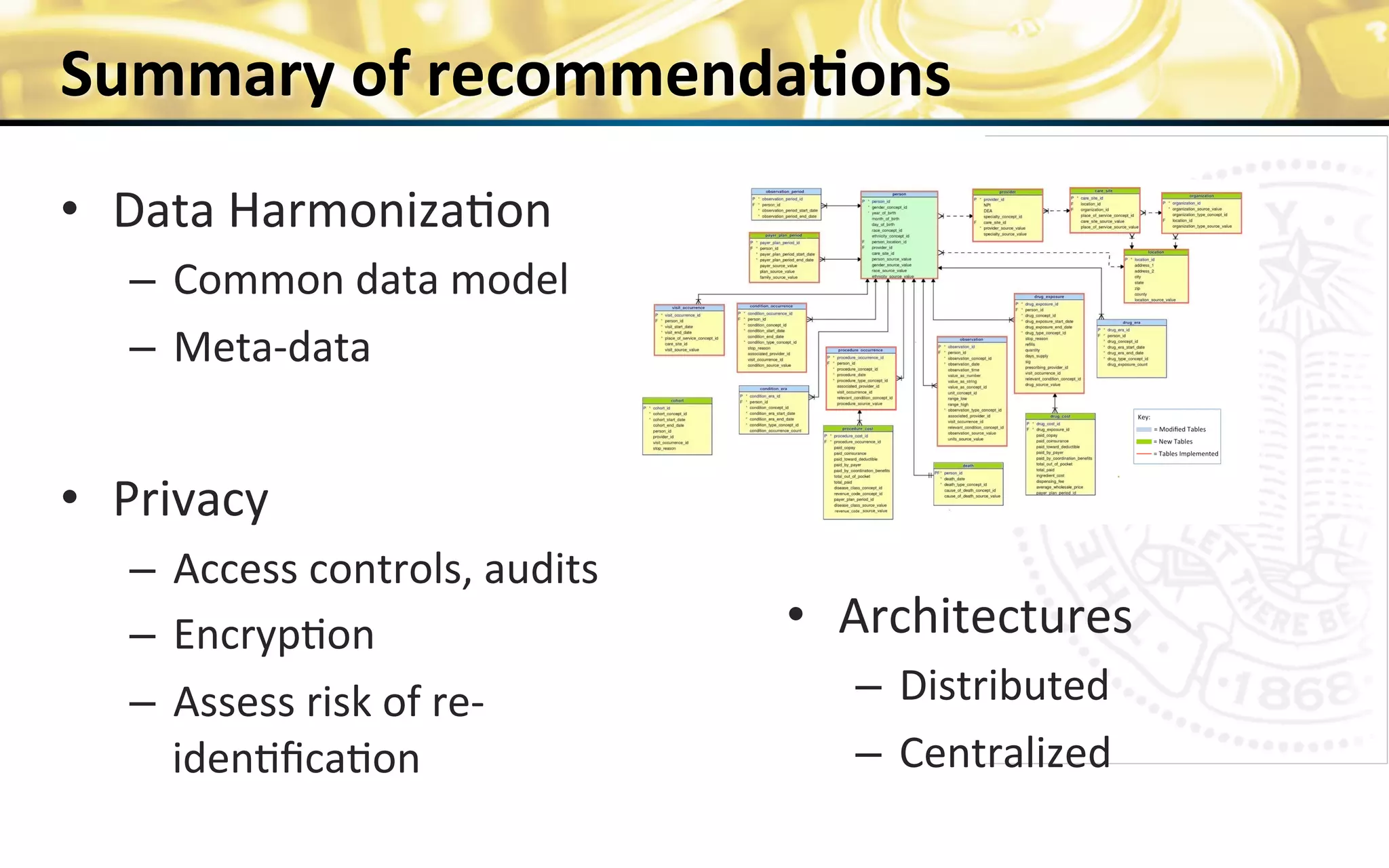
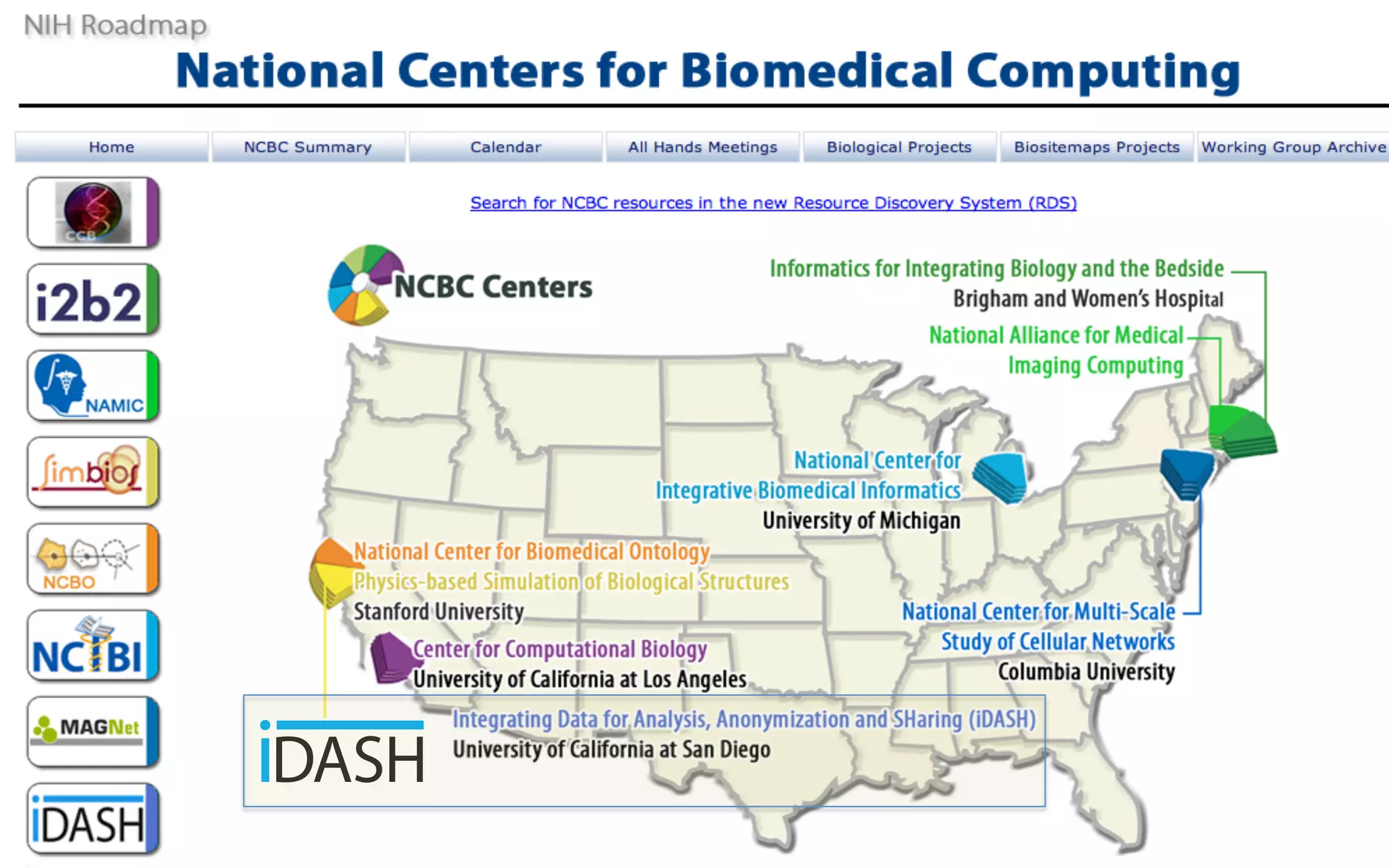
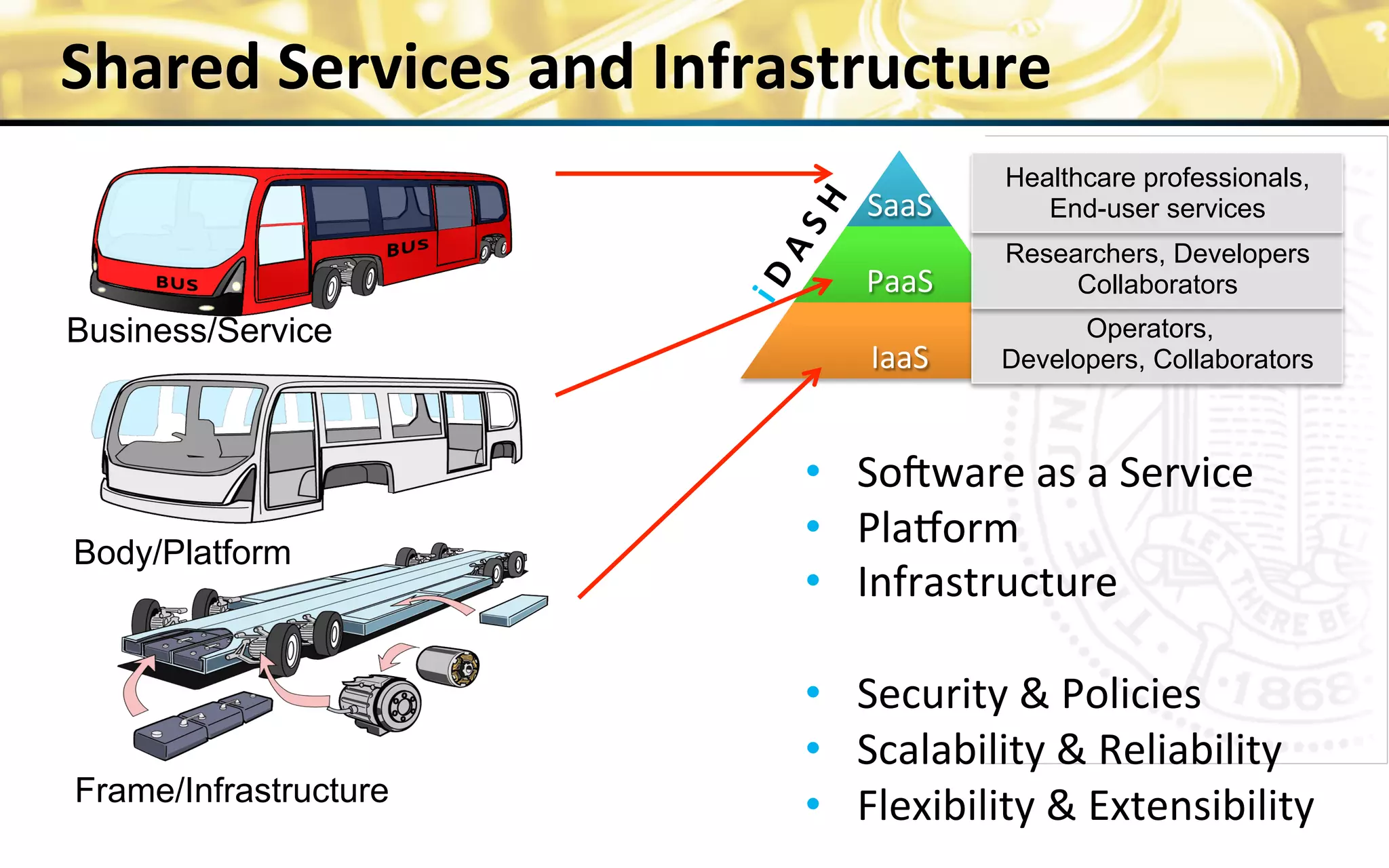
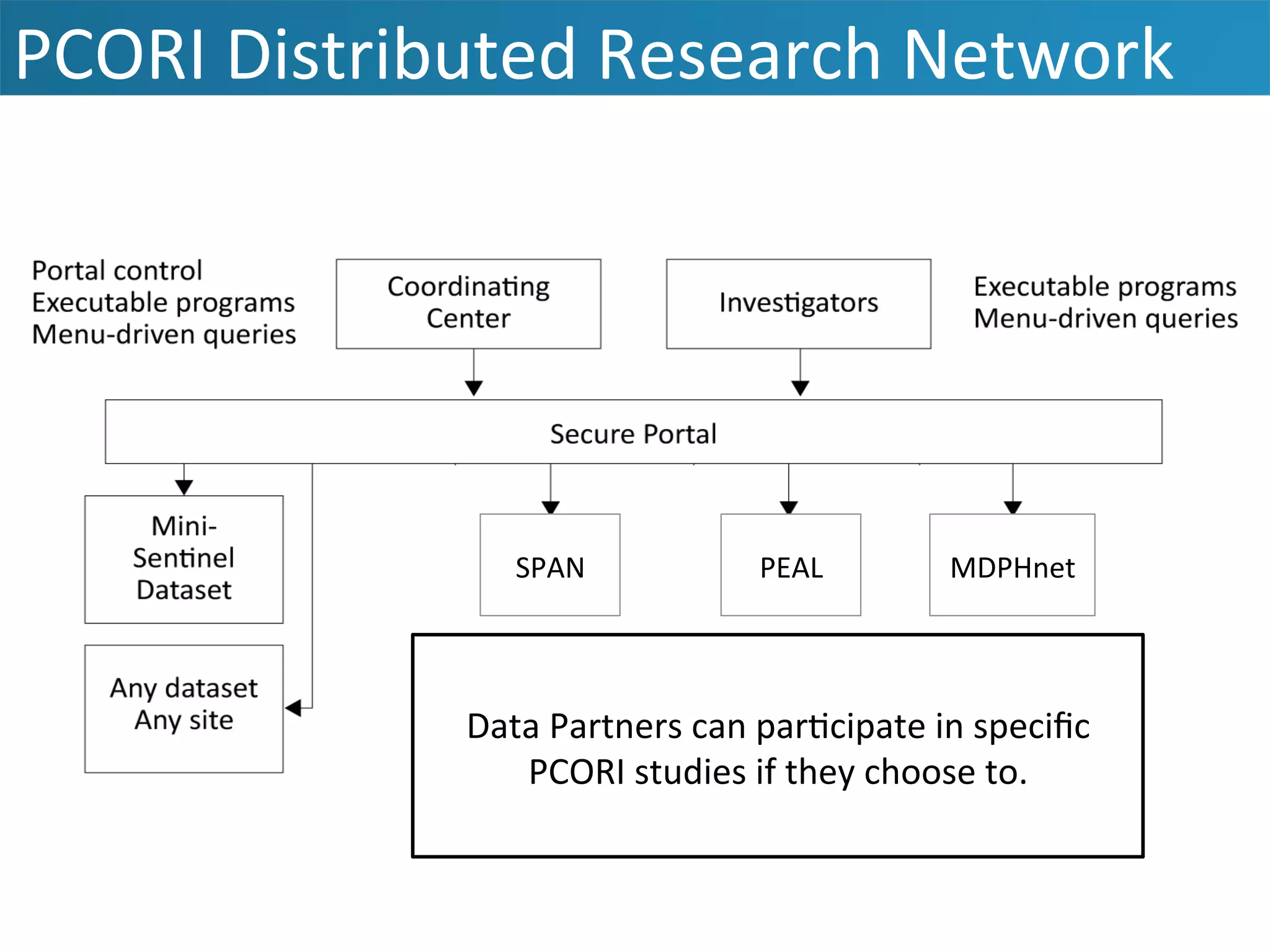
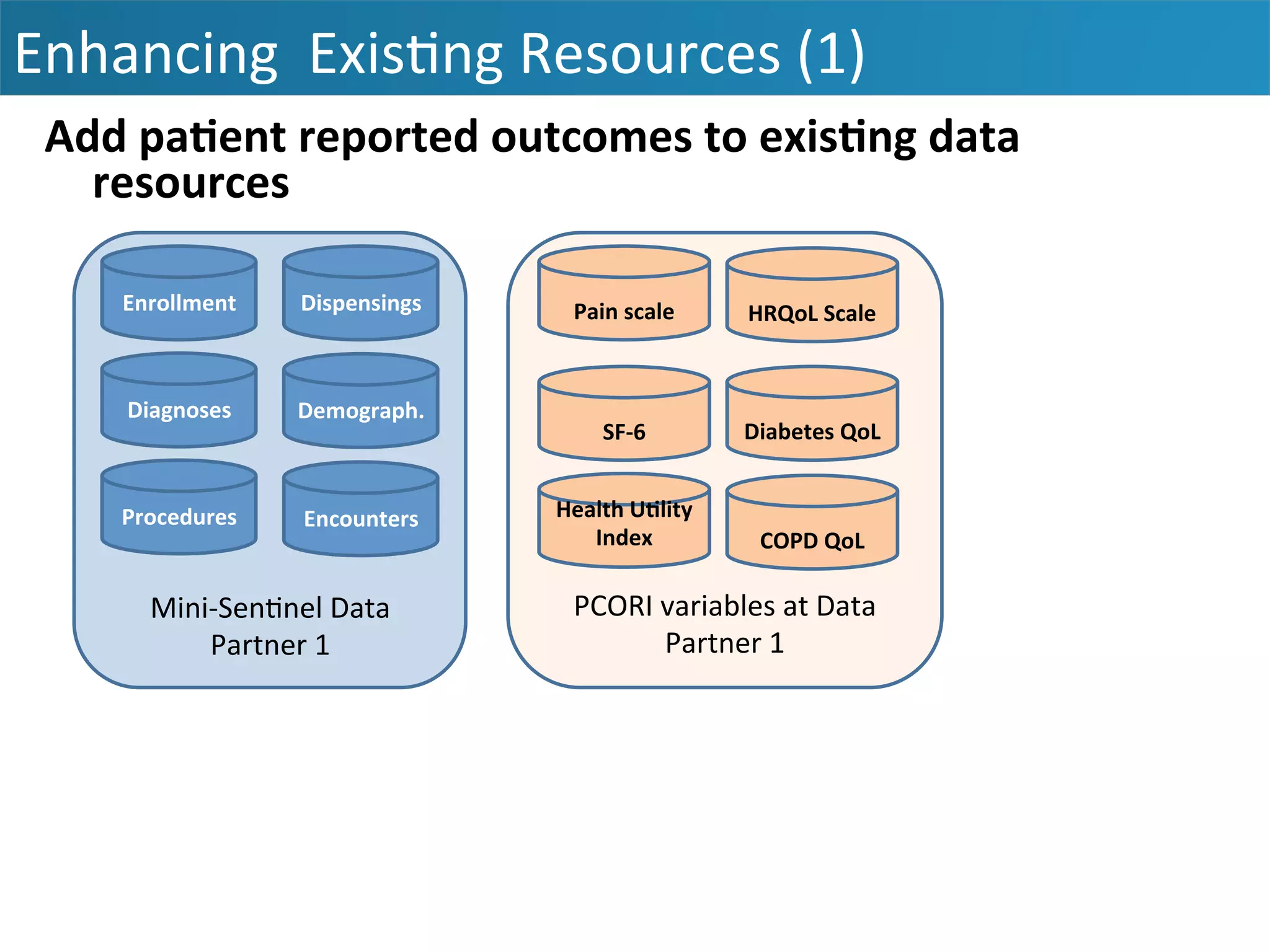
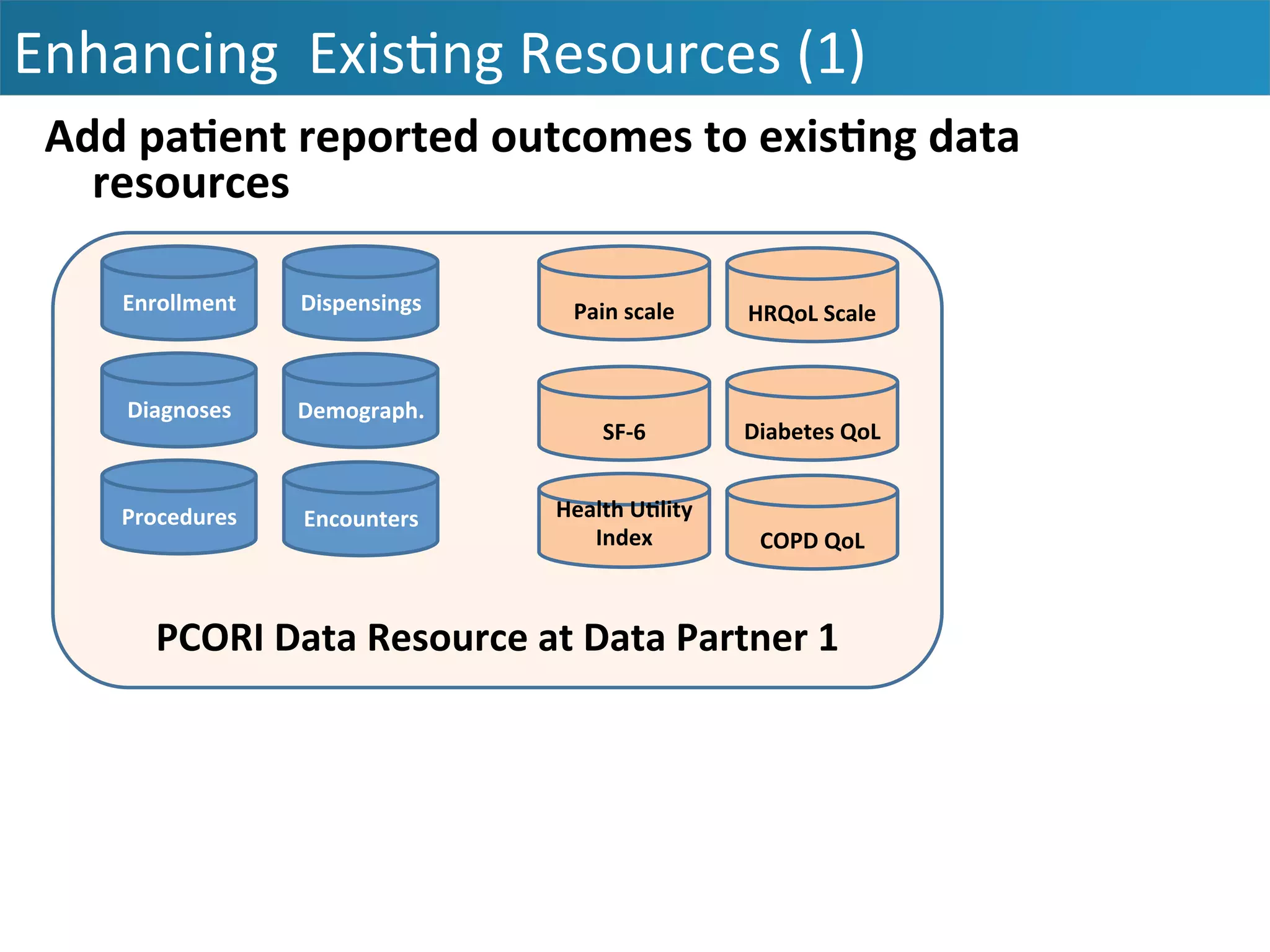
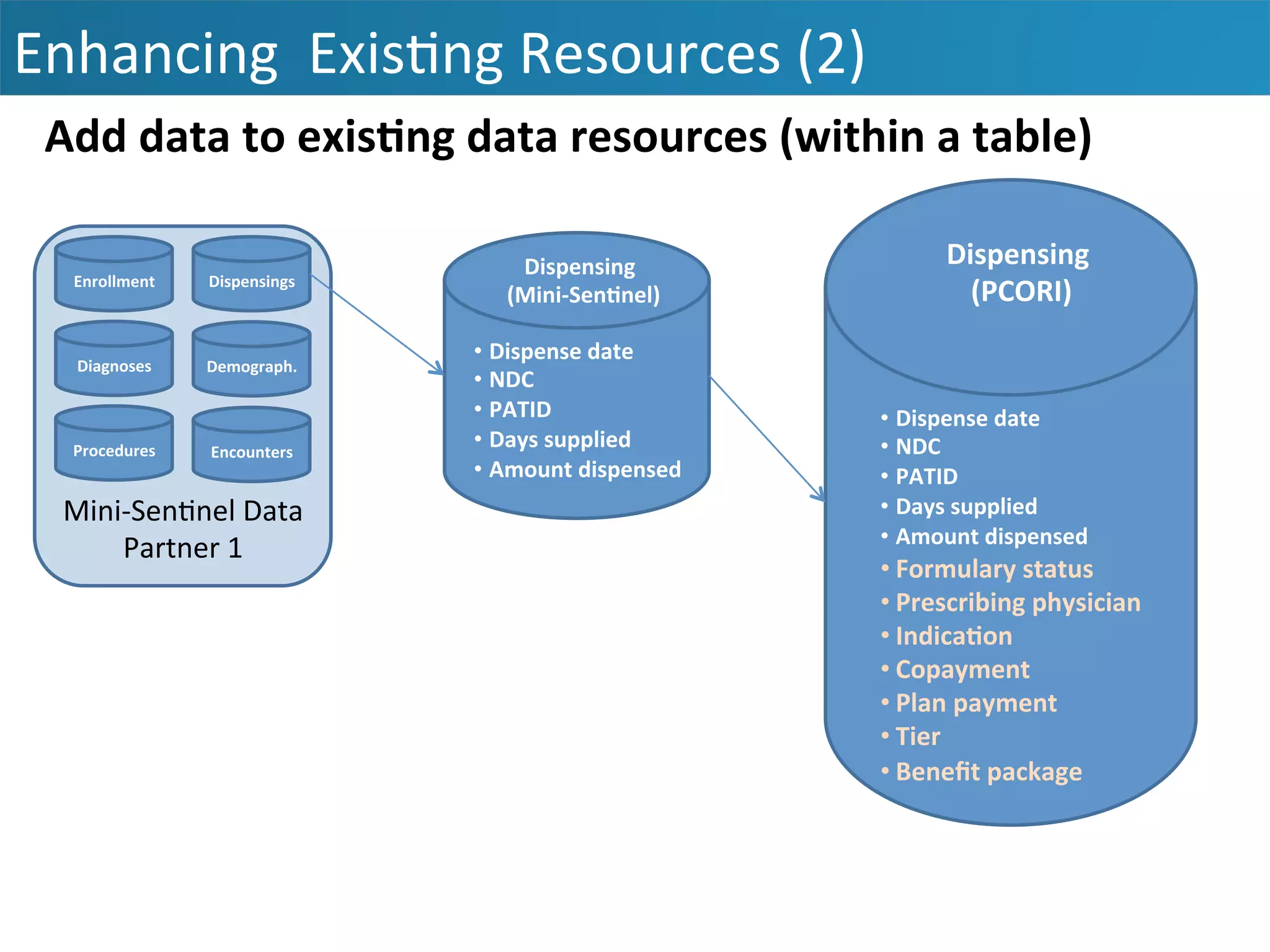
The document outlines the mission and vision of the Patient-Centered Outcomes Research Institute (PCORI), which aims to enable informed healthcare decisions by providing evidence-based information gathered from diverse patient populations. It discusses the necessity of developing a national infrastructure for patient-centered outcomes research (PCOR), emphasizing the importance of data interoperability and patient engagement in governance. Challenges and opportunities in building this infrastructure are explored, including the integration of electronic health records and the need for stakeholder collaboration to improve healthcare delivery and outcomes.

































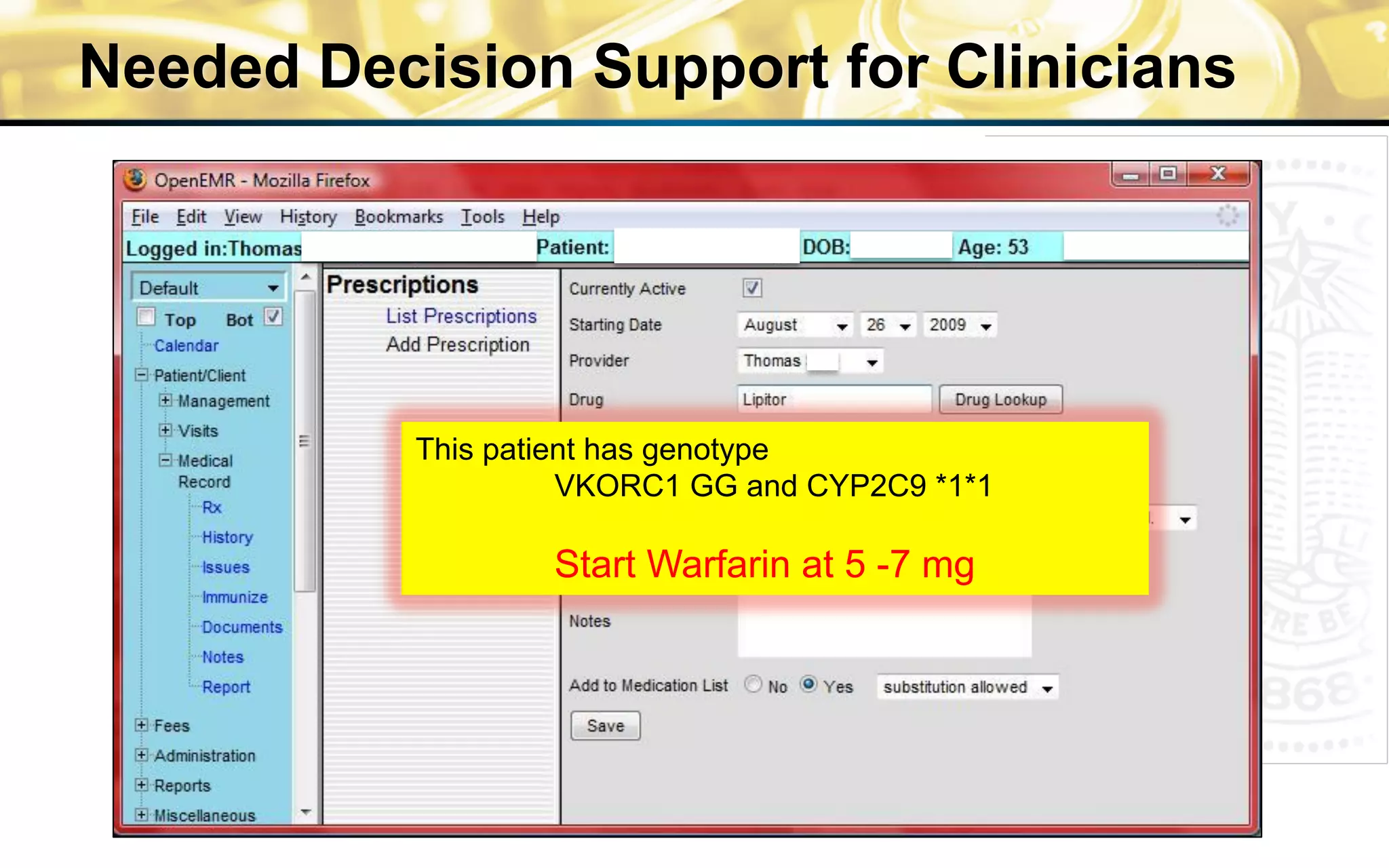



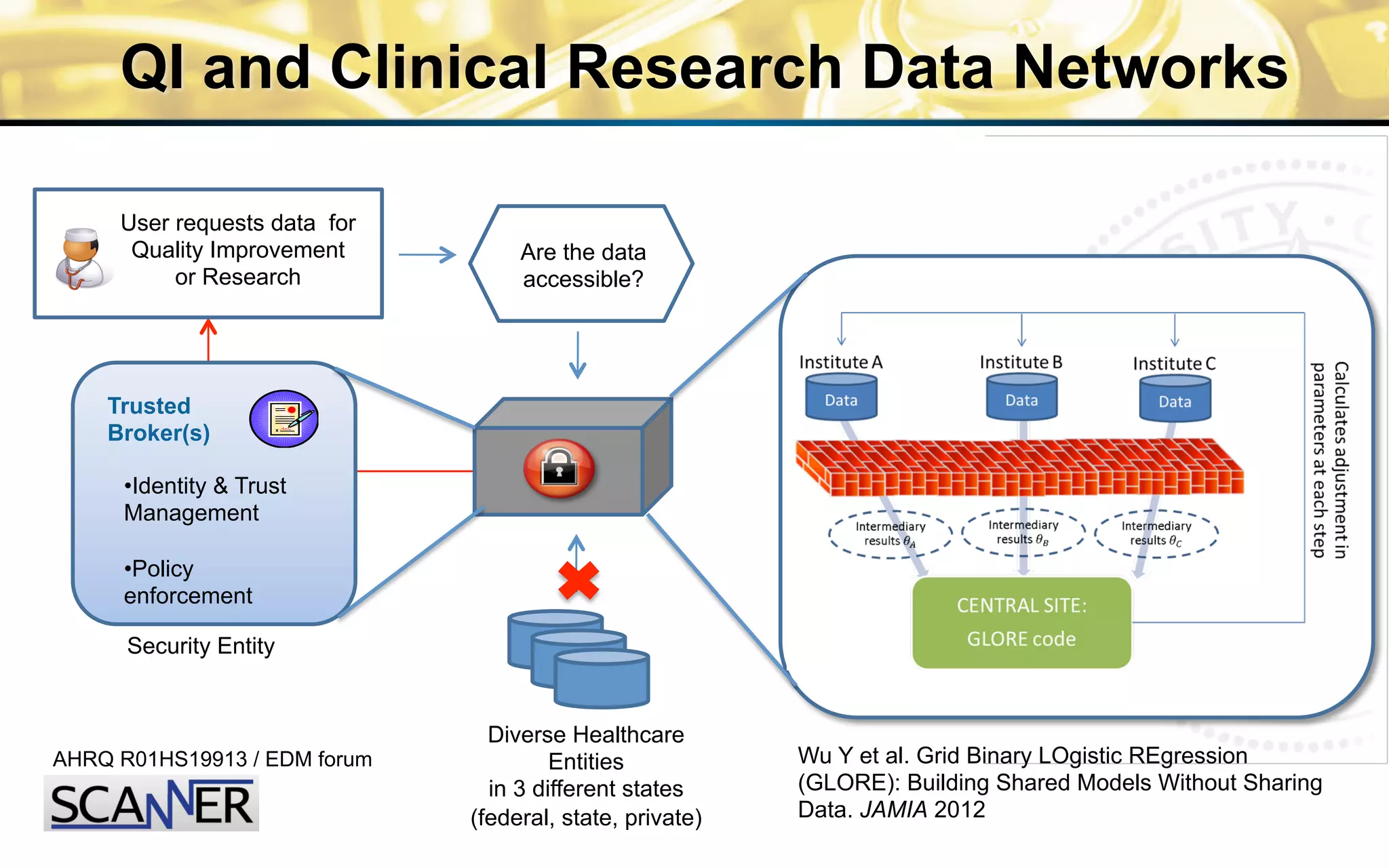






































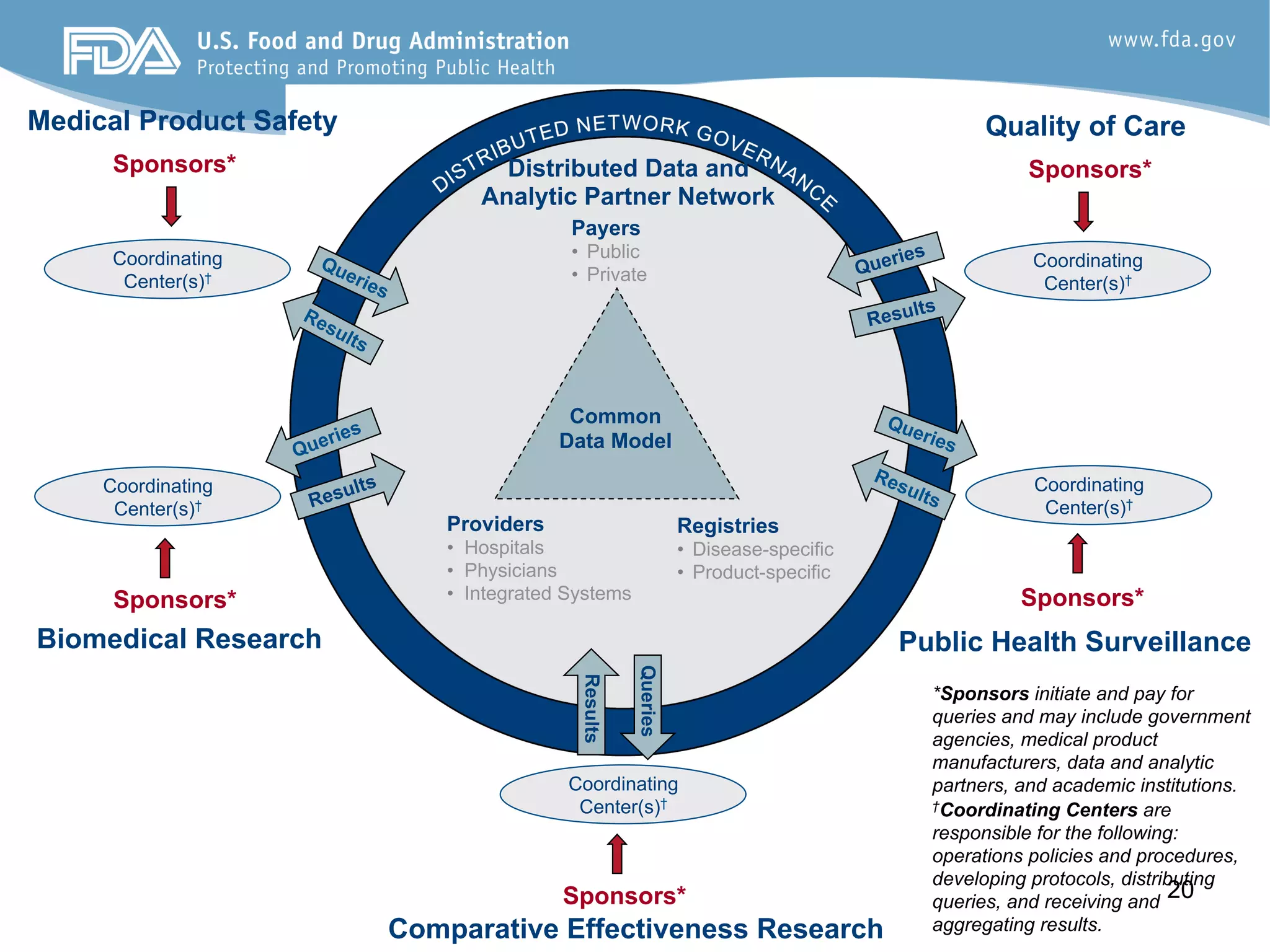





























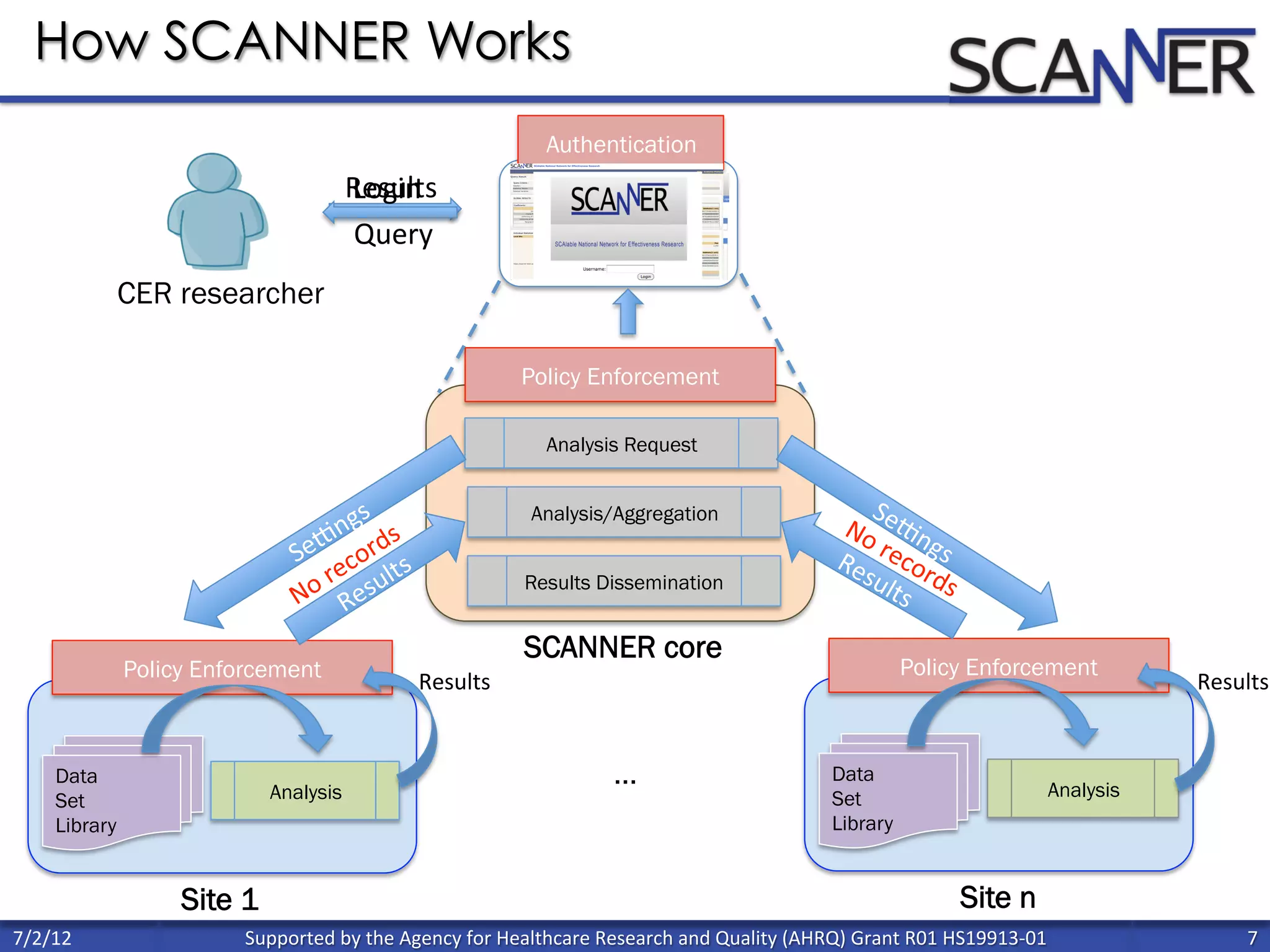
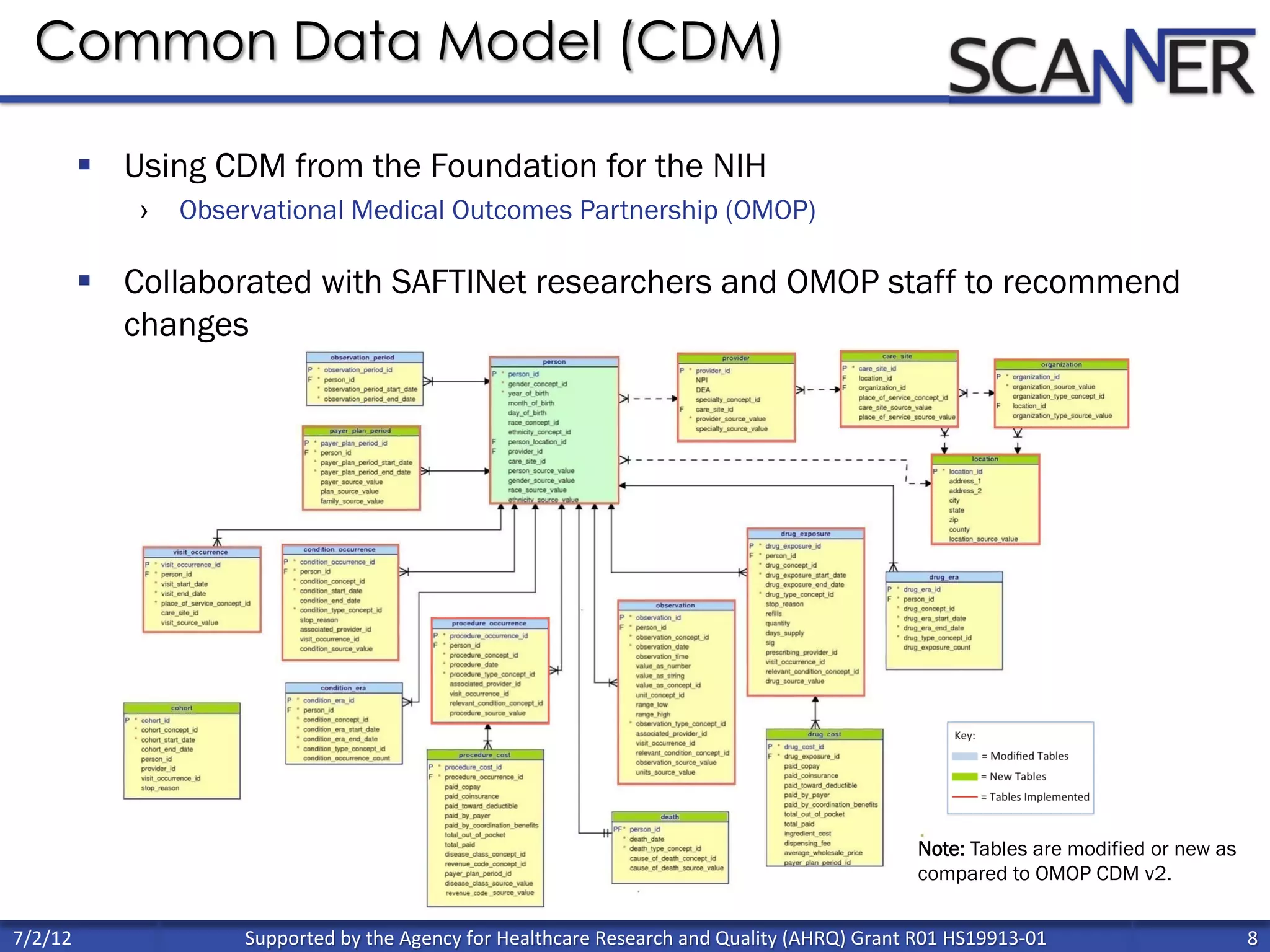

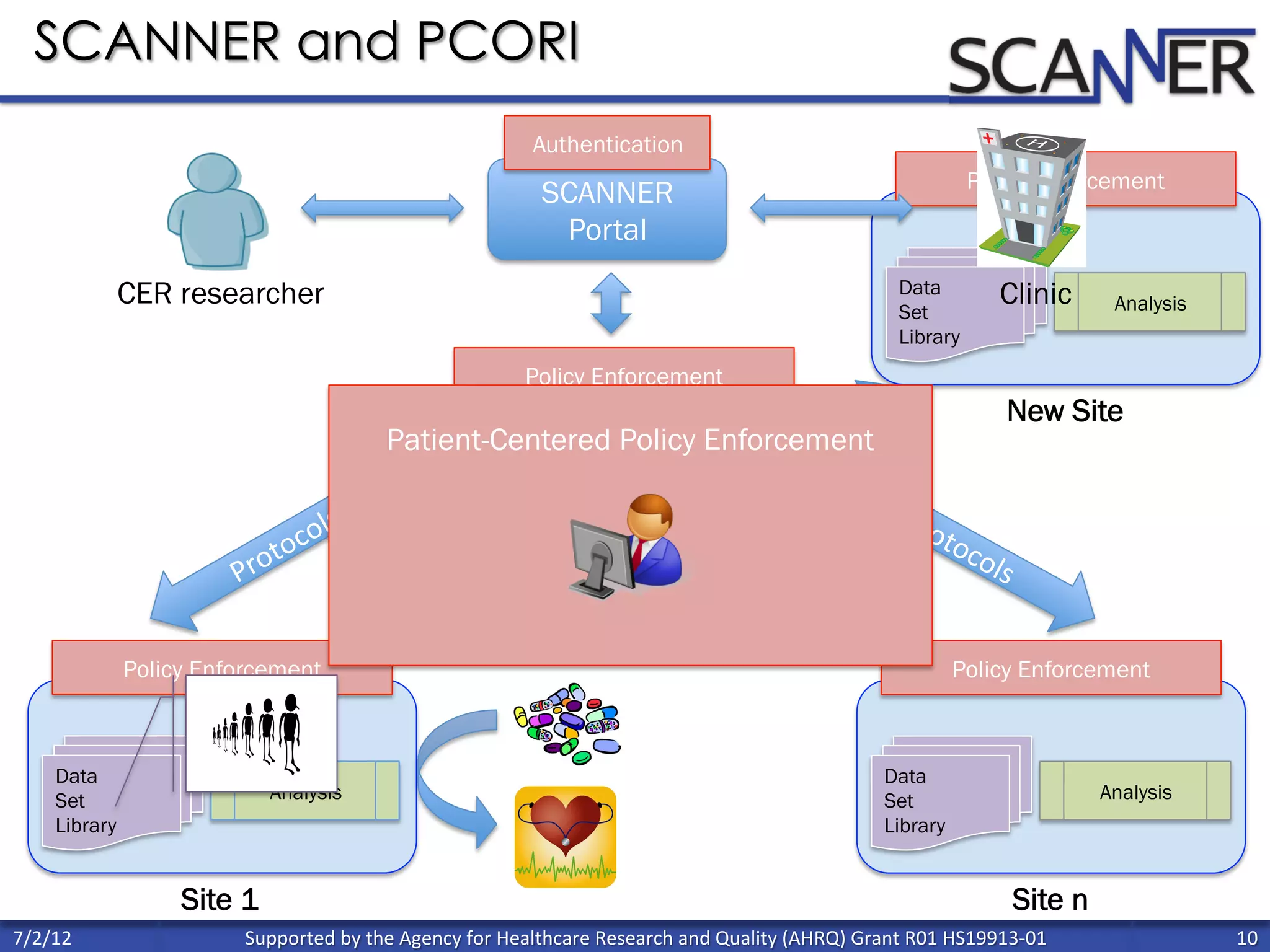

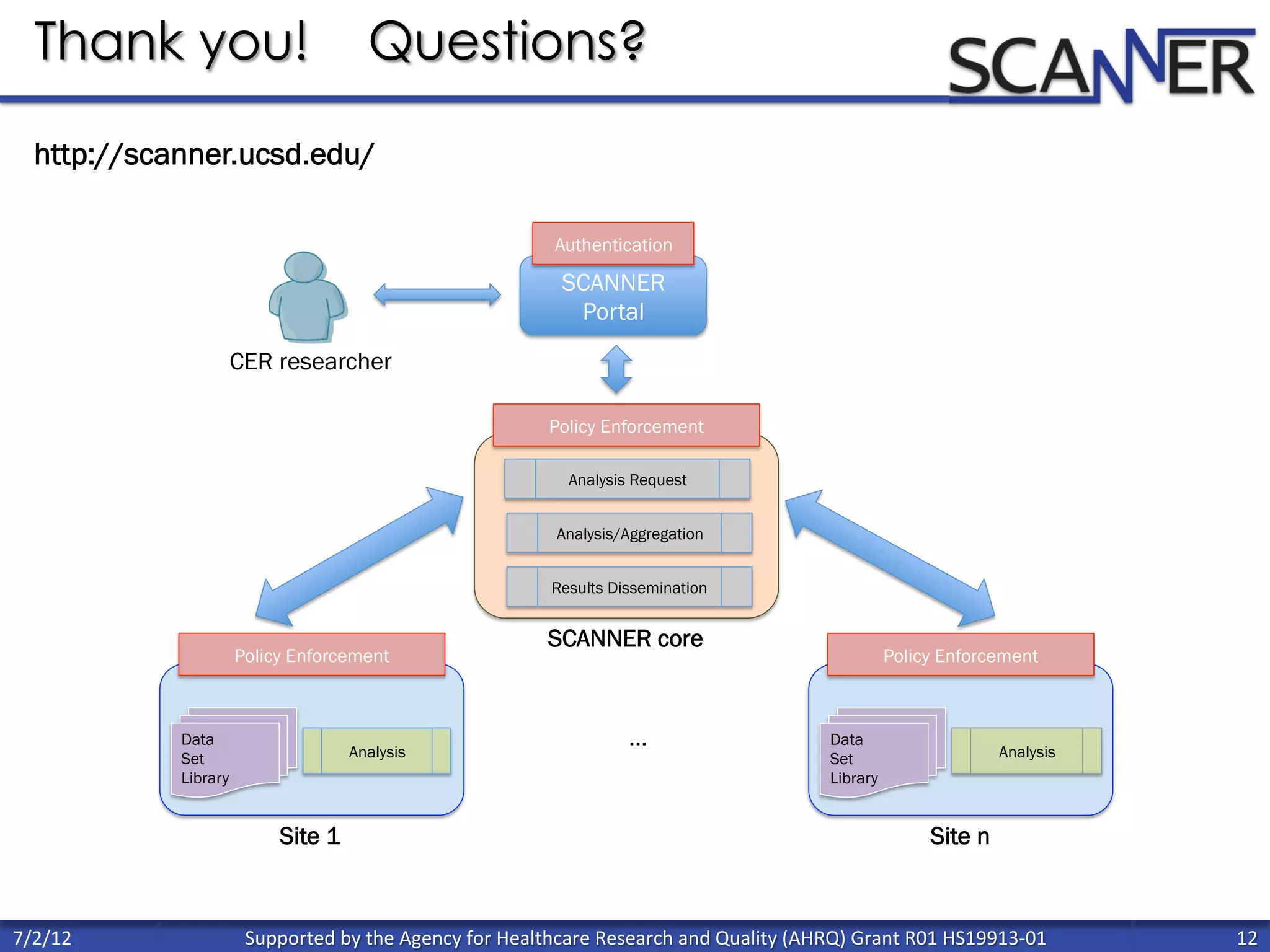





![Percent
of
Pa9ents
in
Remission
0%
10%
20%
30%
40%
50%
60%
70%
80%
90%
100%
Jul-2007N=338
Aug-2007N=396
Sep-2007N=428
Oct-2007N=479
Nov-2007N=508
Dec-2007N=531
Jan-2008N=570
Feb-2008N=607
Mar-2008N=643
Apr-2008N=654
May-2008N=667
Jun-2008N=671
Jul-2008N=686
Aug-2008N=731
Sep-2008N=754
Oct-2008N=801
Nov-2008N=832
Dec-2008N=901
Jan-2009N=973
Feb-2009N=995
Mar-2009N=1021
Apr-2009N=1070
May-2009N=1112
Jun-2009N=1194
Jul-2009N=1240
Aug-2009N=1277
Sep-2009N=1314
Oct-2009N=1344
Nov-2009N=1366
Dec-2009N=1400
Jan-2010N=1421
Feb-2010N=1410
Mar-2010N=1440
Apr-2010N=1455
May-2010N=1461
Jun-2010N=1471
Jul-2010N=1489
Aug-2010N=1518
Sep-2010N=1547
Oct-2010N=1576
Nov-2010N=1985
Dec-2010N=2032
Jan-2011N=2043
Feb-2011N=2065
Mar-2011N=2124
Apr-2011N=2191
May-2011N=2206
Jun-2011N=2272
Jul-2011N=2301
Aug-2011N=2335
Percent
of
Pa8ents
Month
Percent
of
IBD
Pa8ents
in
Remission
(PGA)
Crandall,
Margolis,
Colle]
et
al
Pediatrics
2012;129:1030
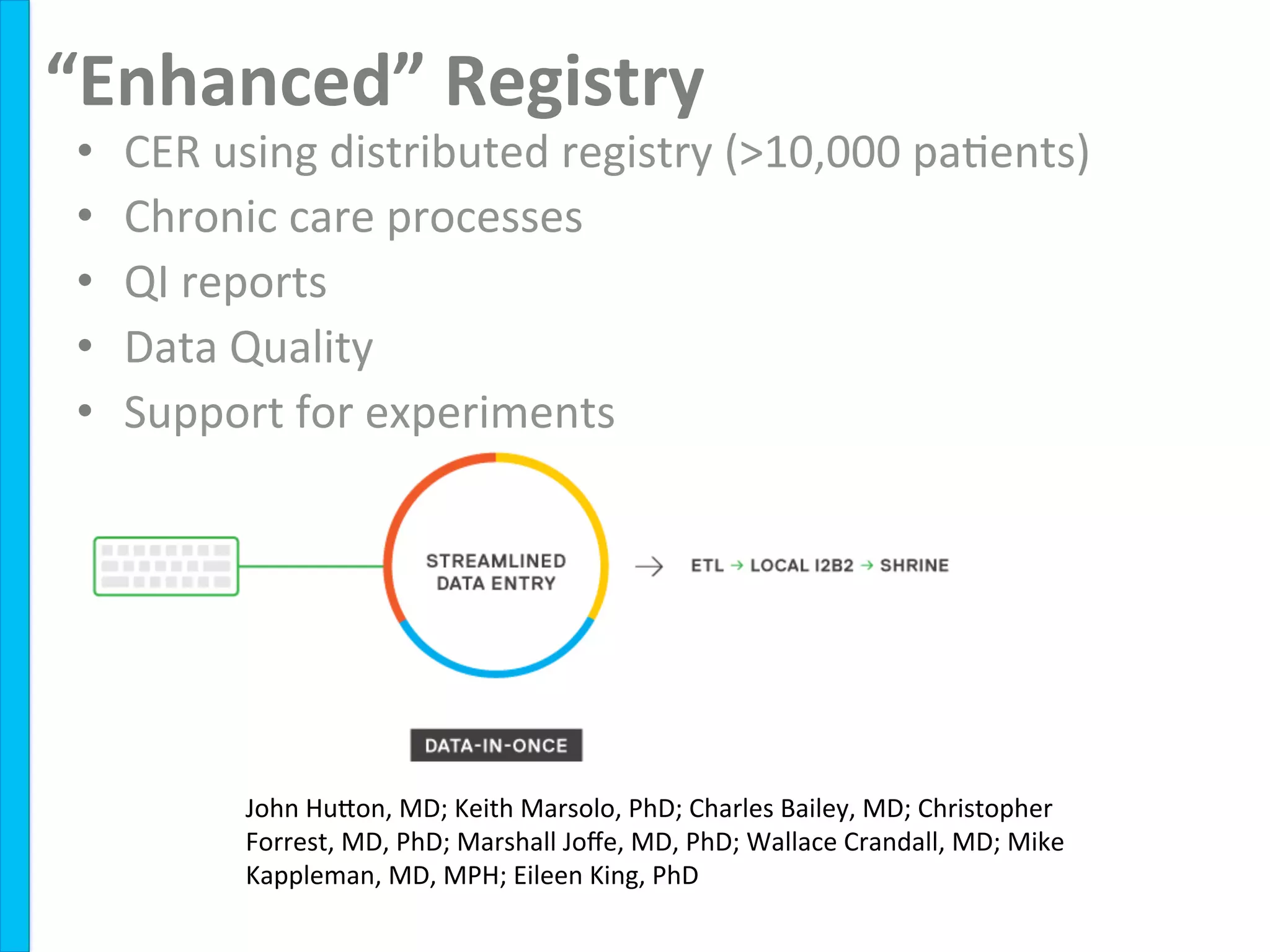
Remission
rate:
55%
to
75%
36
Care
Sites
310
physicians
>10,000
pa8ents
Standardized
care](https://image.slidesharecdn.com/combinedpowerpoints-130805112828-phpapp02/75/National-Workshop-to-Advance-Use-of-Electronic-Data-151-2048.jpg)



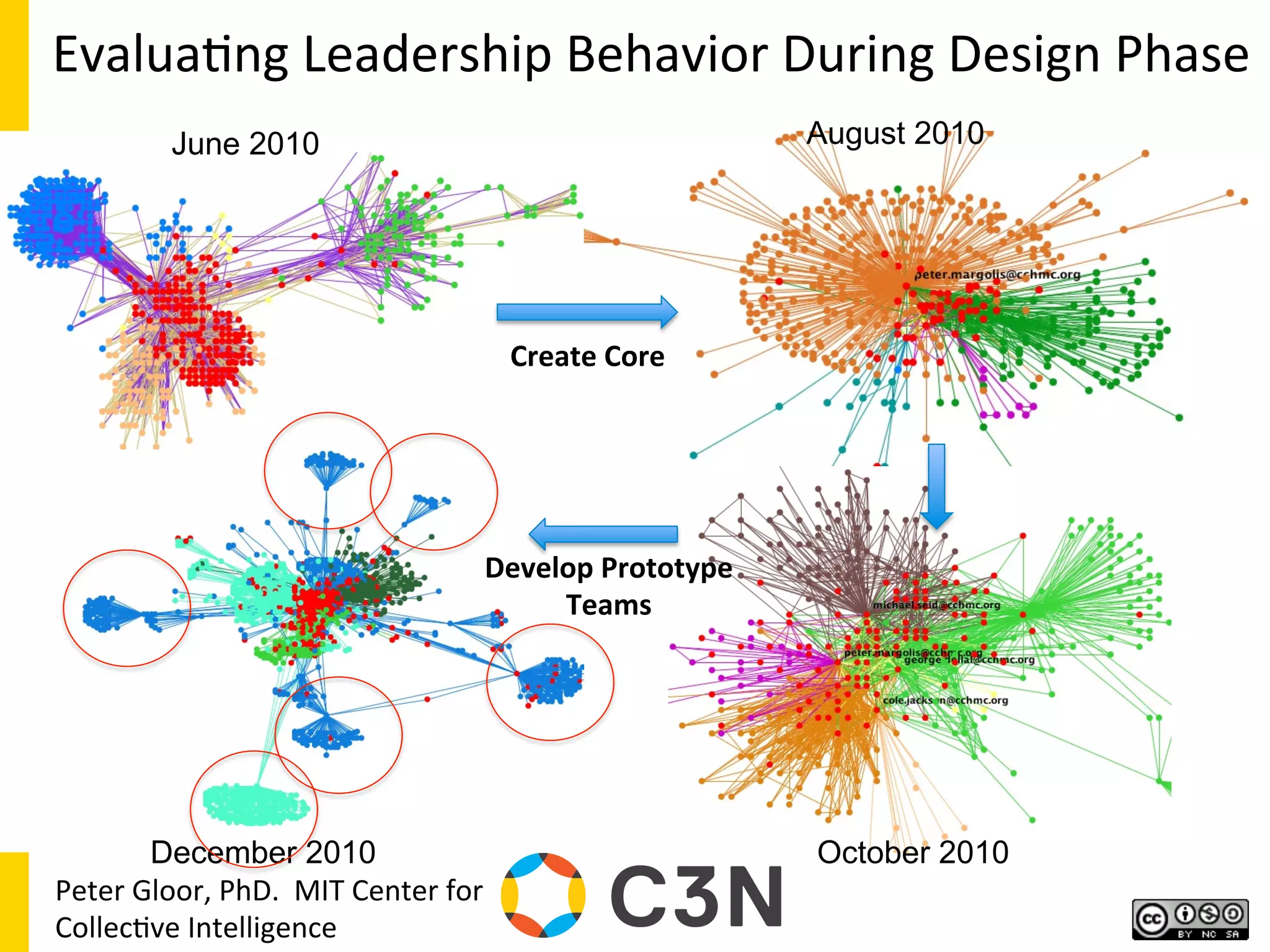
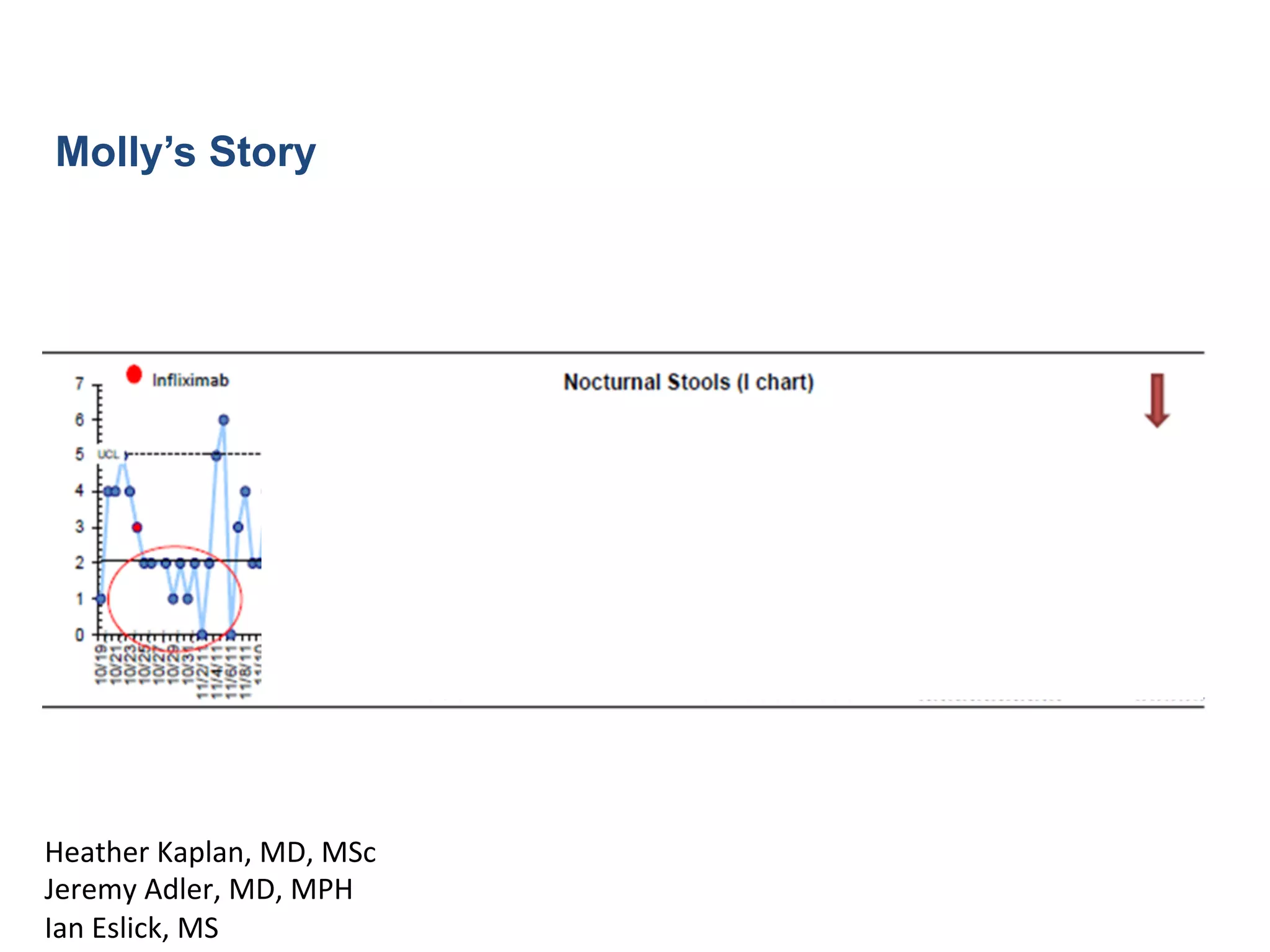
![13
Courtesy
Richard
Colle],
MD
Keith
Marsolo,
PhD](https://image.slidesharecdn.com/combinedpowerpoints-130805112828-phpapp02/75/National-Workshop-to-Advance-Use-of-Electronic-Data-157-2048.jpg)