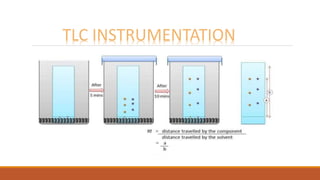
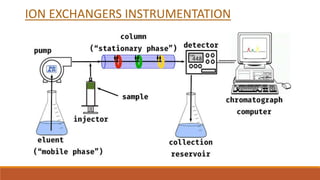
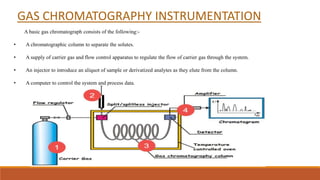
This document provides an overview of chromatography, including the different types. It begins by defining chromatography as a process that separates components of a sample mixture based on their differential distribution between a stationary and mobile phase. It then describes the principal, noting that chromatography usually consists of a mobile phase and stationary phase. It provides details on thin layer chromatography (TLC), ion exchange chromatography, and gas chromatography. For each type, it discusses the stationary and mobile phases, instrumentation, and advantages. The document is intended to introduce the topic of chromatography and key aspects of different chromatographic methods.