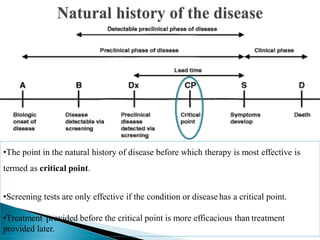
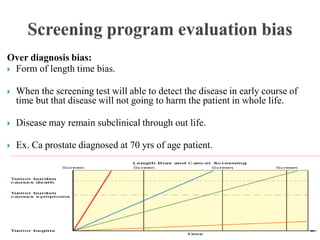
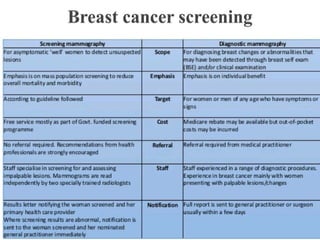
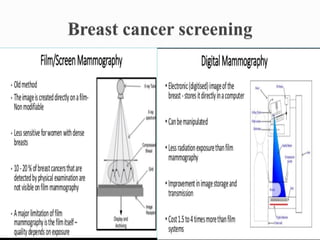
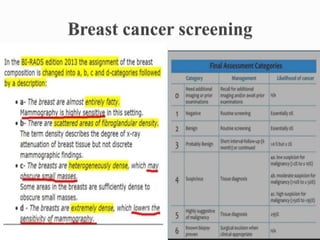
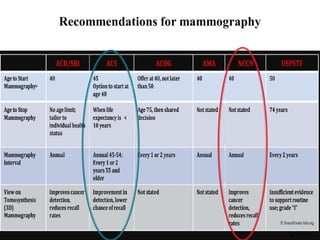
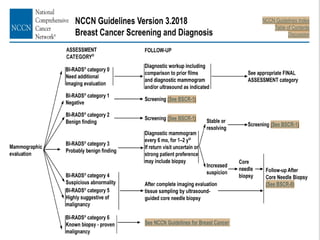
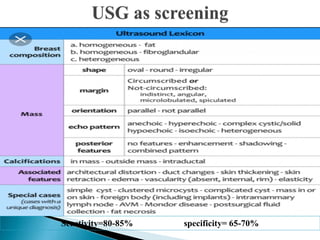
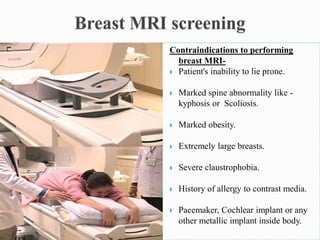
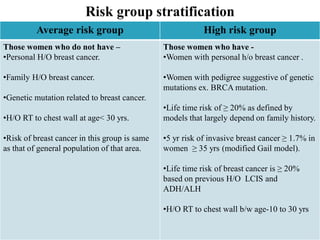
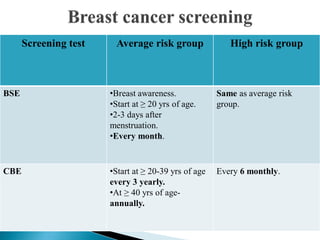
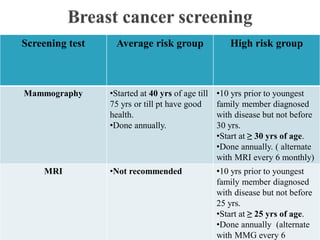
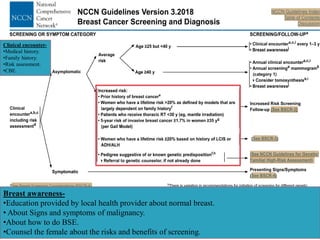
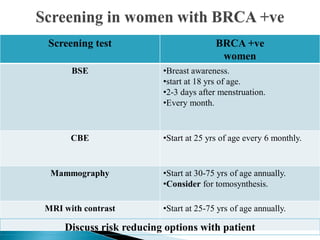
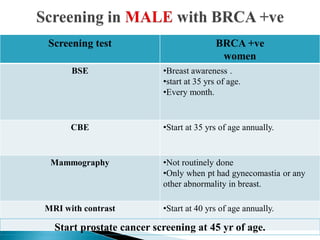
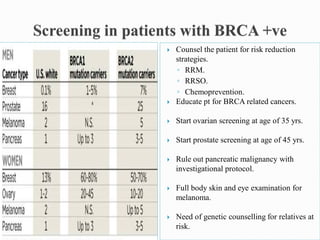
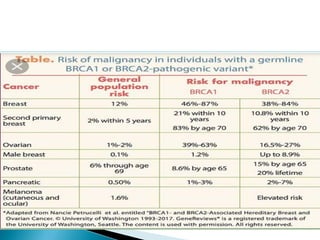
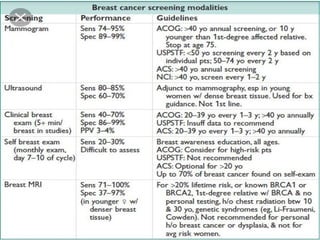
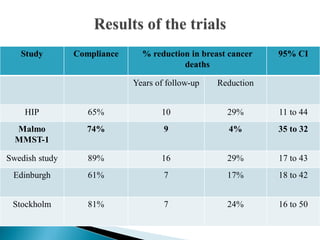
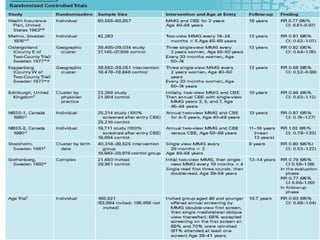
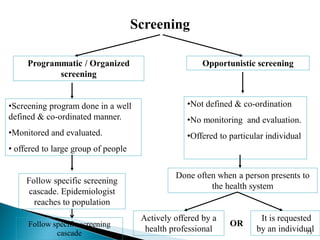
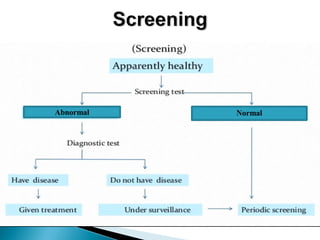
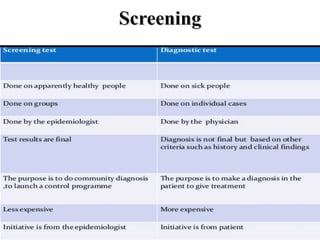
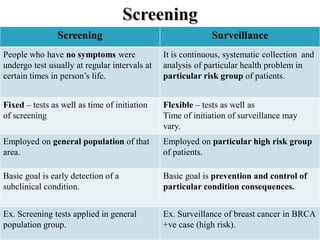
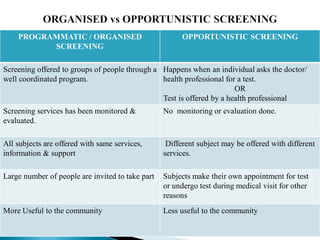
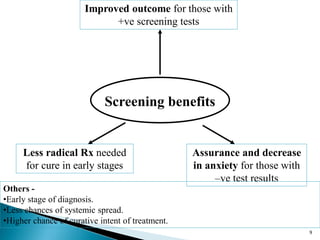
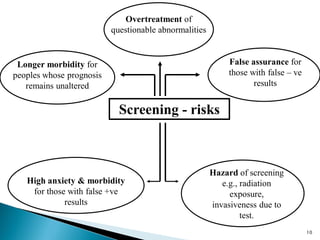
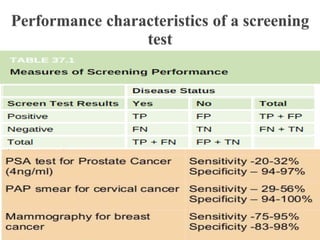
This document discusses cancer screening. It defines screening as testing asymptomatic individuals at regular intervals to detect cancer early. The goals of screening are to detect cancer early to prevent death and suffering while using minimal treatment. For screening to be effective, the disease must have a detectable preclinical phase and early treatment must improve outcomes. Screening can detect cancer earlier and allow less invasive treatment, but also has risks like overdiagnosis and false positives. Proper evaluation of screening programs is important to understand outcomes and costs. Guidelines recommend screening for breast, cervical and prostate cancers in average risk individuals.



![Principles of screening [Wilson & Junger, WHO Report 1968]-
The condition must be an important health problem.
There should be existence of a suitable diagnostic test.
The test should be acceptable by general population.
Facilities for that acceptable diagnostic test should be available.
The natural history of disease or time line of disease must be clearly understood.
There should be a recognized latent or pre clinical phase.
There should be acceptable and recommended treatment guidelines for that
particular disease.
The cost of case finding should be economically balanced.](https://image.slidesharecdn.com/chapter2-191105204624/85/Chapter-2-3-cancer-screening-7-320.jpg)



![There must be balance between cost & benefits of screening test.
Basic models for the evaluation of costs & outcomes:
COST BENEFIT ANALYSIS-
◦ Expressed in terms of monetary benefit.
◦ Difficult & arbitrary.
COST EFFECTIVENESS ANALYSIS-
◦ Expressed in terms of health outcomes benefit.
◦ Cost to detect one cancer case, prevent one cancer related mortality, addition of a
year of life , improvement in QOL etc.
MARGINAL COST PER YEAR OF LIFE SAVED[MCYLS]-
◦ Most appropriate estimate of cost effectiveness of cancer screening at the most
basic level.
◦ Marginal costs= Cost of screening program- Cost of case detection &
management without screening
◦ Marginal effectiveness= Years gained by screening- years expected without
◦ MCYLS= Marginal cost/Marginal effectiveness](https://image.slidesharecdn.com/chapter2-191105204624/85/Chapter-2-3-cancer-screening-15-320.jpg)