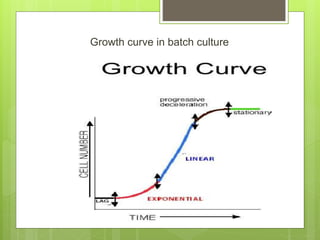
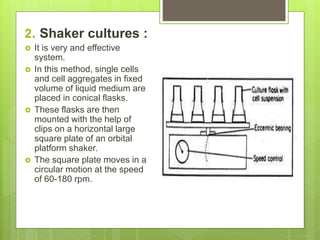
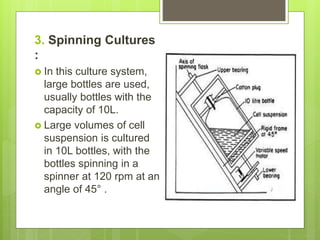
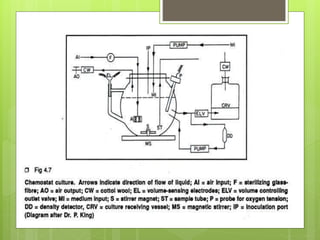
This document discusses cell suspension culture, which involves growing plant cells, tissues, or organs in liquid nutrient medium. There are two main types of cell suspension cultures: batch culture and continuous culture. Batch culture involves growing cells in a finite volume of agitated medium, while continuous culture replaces old medium continuously to maintain cells in steady growth. The document outlines different methods for each type of culture and their characteristics, advantages, and importance for studying plant cell physiology and biochemistry.