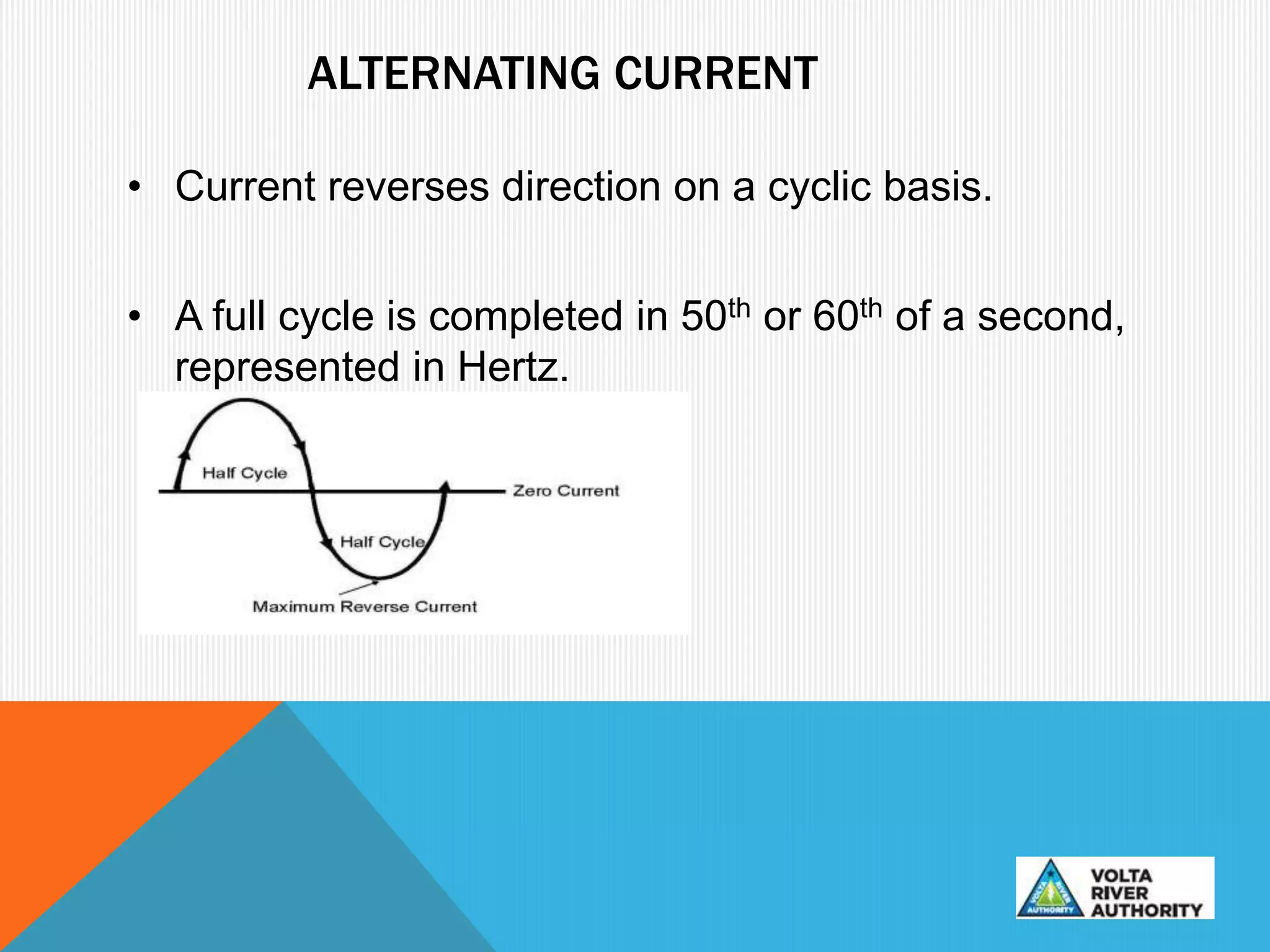
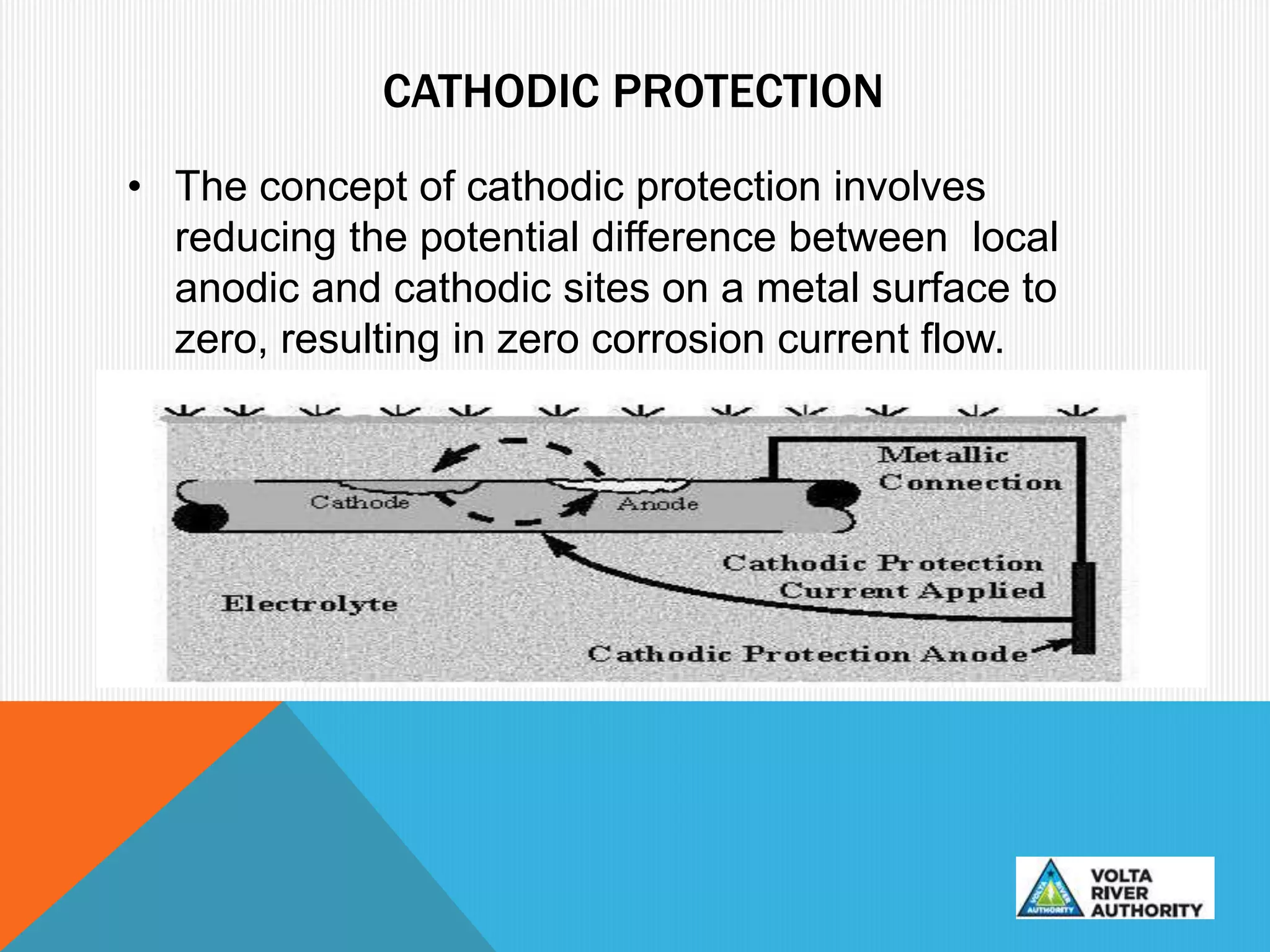
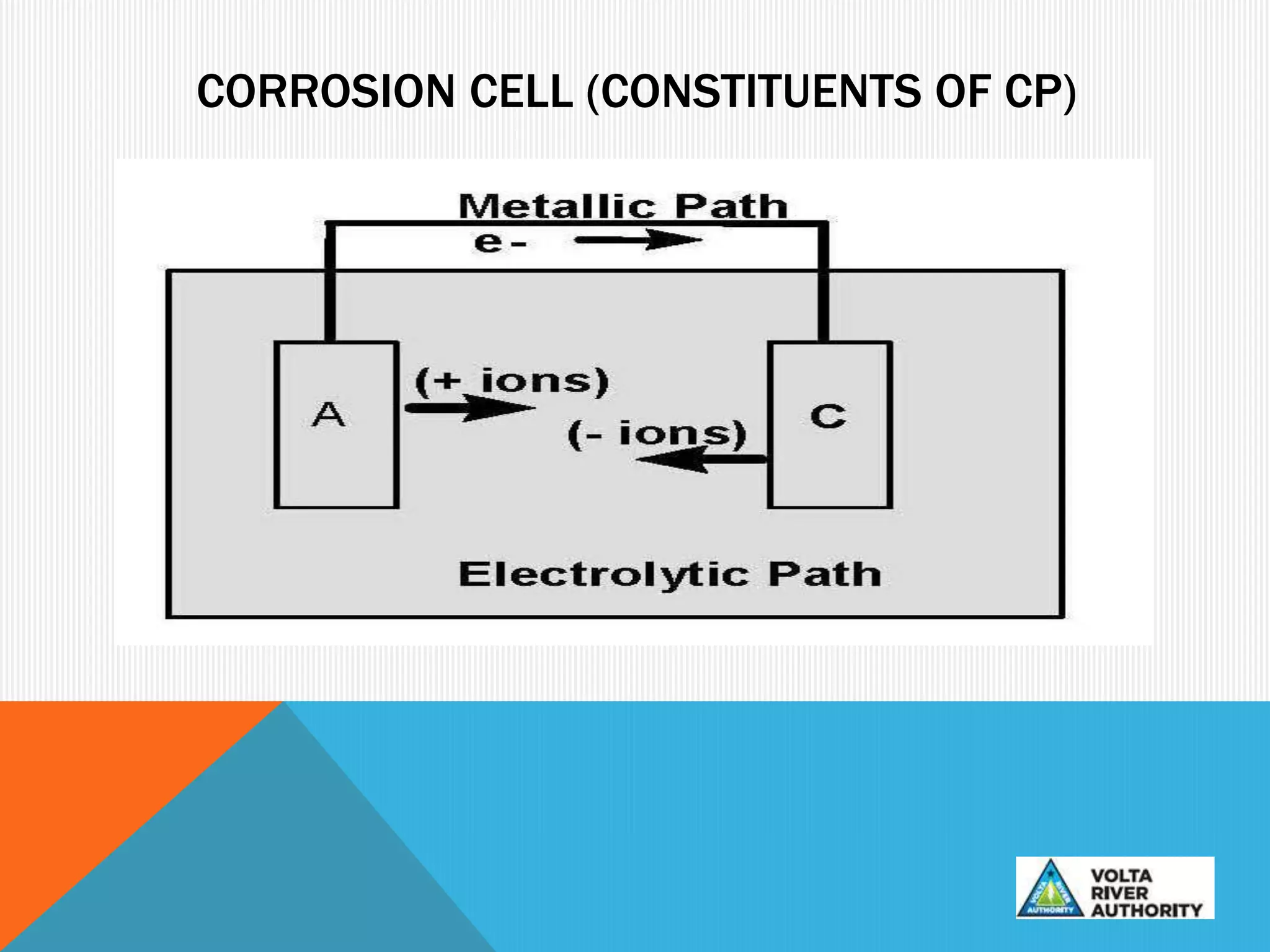
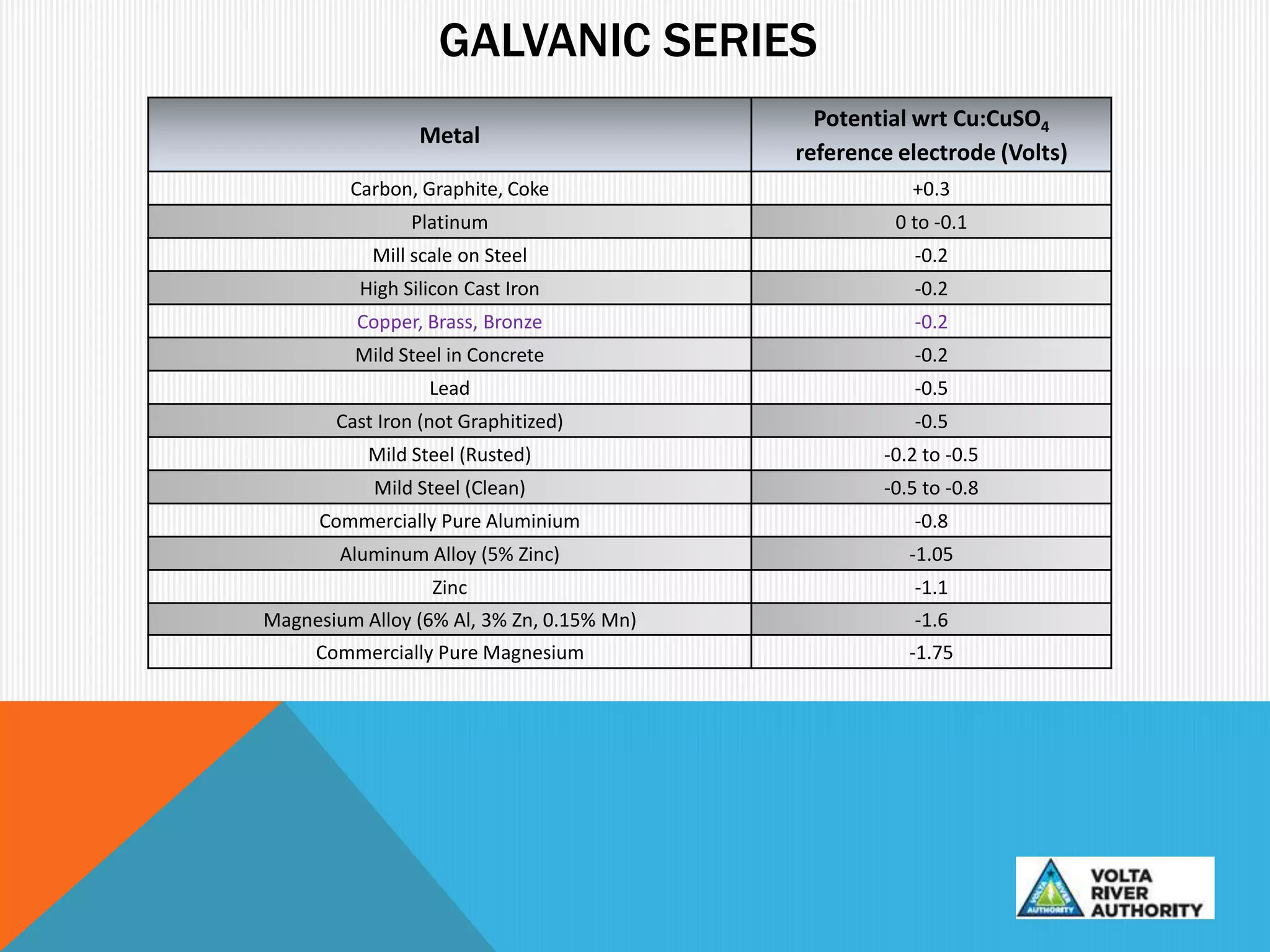
This document provides information on corrosion and cathodic protection. It discusses how corrosion degrades metals through chemical or electrochemical reactions. Corrosion costs over $2 trillion annually worldwide. Cathodic protection works by reducing the potential difference between anodic and cathodic sites on a metal surface to stop corrosion. There are two main types of cathodic protection systems - galvanic systems which use sacrificial anodes and impressed current systems which use an external current source. Both systems have advantages and limitations for different corrosion protection applications.