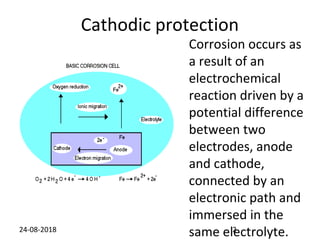
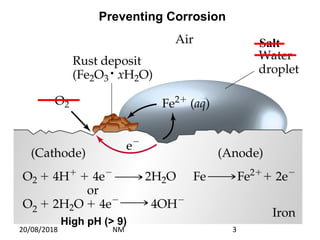
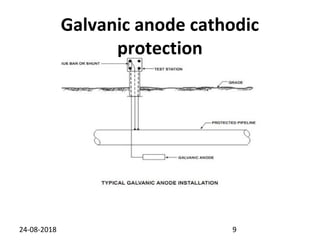
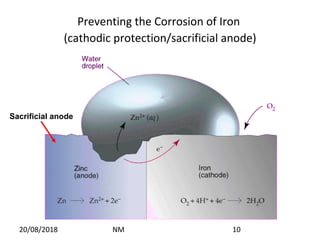
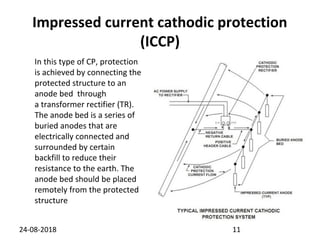
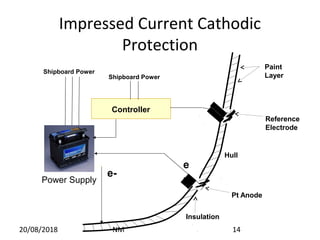
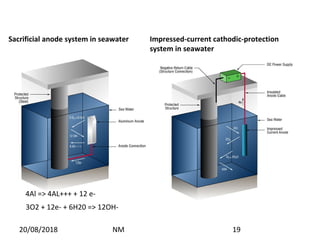
Cathodic protection is a method used to protect steel structures like pipelines, ships, and platforms from corrosion. There are two main types: galvanic anode cathodic protection which uses sacrificial anodes to create a potential difference; and impressed current cathodic protection which uses an external power source and inert anodes. Both types aim to polarize the protected structure negatively to prevent corrosion from occurring. Cathodic protection is commonly used on buried pipelines to mitigate corrosion where coatings may be damaged.