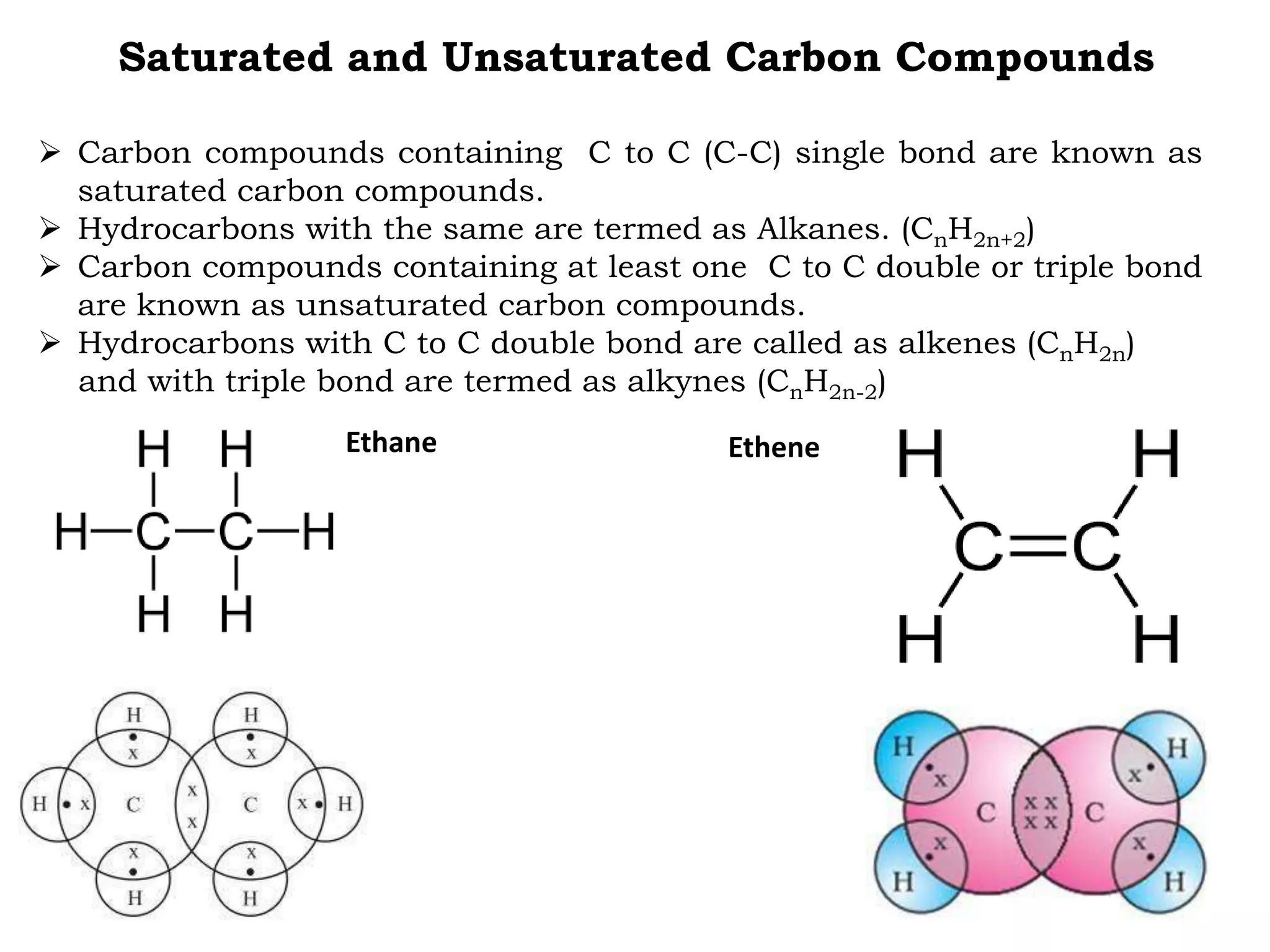
The document provides an overview of carbon, its properties, and its compounds, highlighting its significance in various fields such as structural biology and medicine. It discusses carbon's allotropes, bonding characteristics, and the versatility of carbon due to its ability to form covalent bonds and catenate, leading to a vast array of carbon compounds. Additionally, it describes saturated and unsaturated hydrocarbons, basic chemical reactions involving carbon compounds, and specific examples like ethanol and acetic acid.



![ As we Write down electronic configuration
of Carbon i.e
6C = 1s2 2s2 2p2 or [He] 2s2 2p2
We could see carbon is left out with 4 more
electrons to attain nearest noble gas
configuration of Neon, and as we know by
virtue every element tries to obtain noble
gas configuration to attain stability either
by gaining or losing of electrons
(Individually or by support)
Carbon could form C4- anion but for a atom or nucleus with 6 electrons to
hold 4 more electrons is highly difficult and in the same way to form C4+
cation the successive ionization energies are drastically high and won’t
allow the formation.
So in order to overcome these conditions and to attain stability carbon
will share its valance electrons with other elements , and both the
elements on bonding attains nearest noble gas configuration
This sharing will continue until the valancy get satisfied either by the
same atoms or other involved in reaction](https://image.slidesharecdn.com/carbonanditscompounds-230220170036-66d7f0d2/75/Carbon-and-It-s-Compounds-pptx-4-2048.jpg)