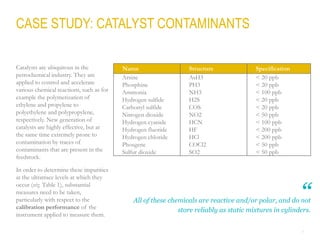
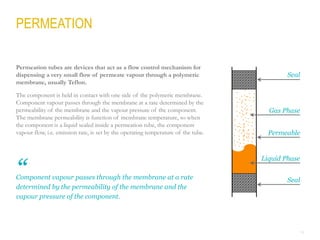
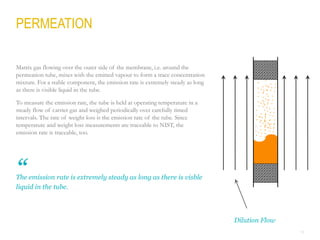
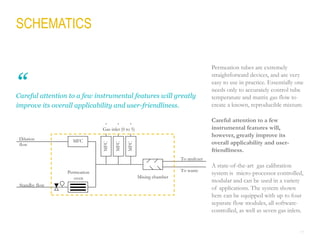
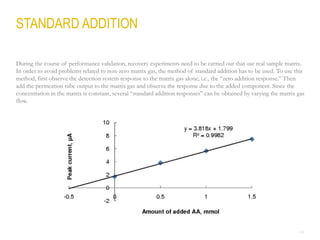
This document provides a comprehensive guide on the calibration challenges in trace and ultratrace gas analysis, focusing on permeation principles and reliable gas standard preparation techniques. It emphasizes the importance of method development, validation, and quality control for accurate gas analysis results. Various calibration strategies, including the use of permeation tubes and dilution methods, are discussed in detail to improve measurement accuracy.