This document discusses bladder cancer. Some key points:
1. Bladder cancer is the most common tumor of the urinary tract and the second most common cause of cancer death.
2. Bladder cancer presents at a muscle-invasive stage in 20-40% of cases.
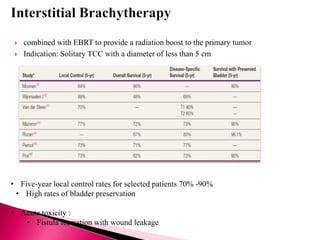
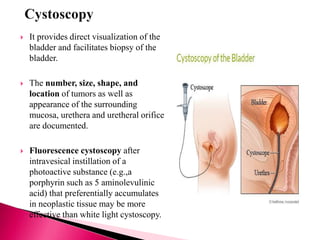
3. Diagnosis involves cystoscopy, urine cytology, imaging like CT/MRI, and biopsy.
4. Treatment depends on stage - non-muscle invasive cancers receive transurethral resection and intravesical therapy while muscle-invasive cancers require radical cystectomy and urinary diversion.































![Indications
Residual disease after initial TURBT
When specimen contained no muscle
High-grade and/or T1 tumor
Timing and strategy:
Most recommend 2-6 weeks after initial TUR
Should include resection of primary tumor site
Evidence: 2nd look TURBT in T1 /HG tumor
1/2 will have residual disease on 2nd look [EAU 2010]
Under stage is more if muscle is absence (50% vs 15%) [Herr JU 1999]
1/4 will have upstage [Herr JU1999]
1/3 will have to change management [Herr JU 1999]
20% increase 5yr DFS [Germen observational study 2003]
European Association of Urology Guidelines 2015.](https://image.slidesharecdn.com/caurinarybladdermanagement-160928090807/85/Ca-urinary-bladder-management-33-320.jpg)

















![Standard PLND
Proximal: Bifurcation of common iliac artery
Distal: Circumflex iliac vein
Lateral :Gentitofemoral nerve
Medial: Bladder wall
Pelvic floor and hypogastric vesse
Anything more (up to bifurcation of aorta and above) can be called an extended
PLND.
Includes b/l obturator, internal, external, common iliac and presacral nodes as well
as nodes at the aortic bifurcation may also Extend to IMA
Evidence[1-3] suggests that a more extended lymphadenectomy is beneficial in
both lymph node–positive and lymph node–negative patients with bladder cancer.
The Bladder Cancer Collaborative Group recommends 10-14 lymph nodes should
be removed at time cystectomy
52
1. Herr HW, Bochner BH, Dalbagni G, et al. Impact of the number of lymph nodes retrieved on outcome in patients with muscle invasive bladder
cancer. J Urol2002;167(3):1295–1298.
2. . Leissner J, Ghoneim MA, bol-Enein H, et al. Extended radical lymphadenectomy in patients with urothelial bladder cancer: results of a
prospective multicenter study. J Urol 2004;171(1):139–144.
3. . Stein JP. The role of lymphadenectomy in patients undergoing radical cystectomy for bladder cancer. Curr Oncol Rep 2007;9(3):213–221.](https://image.slidesharecdn.com/caurinarybladdermanagement-160928090807/85/Ca-urinary-bladder-management-51-320.jpg)