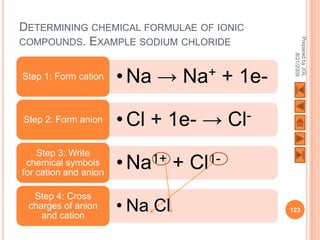
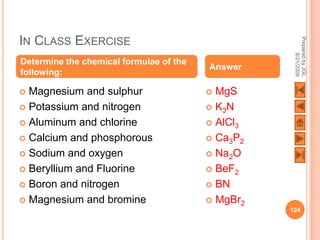
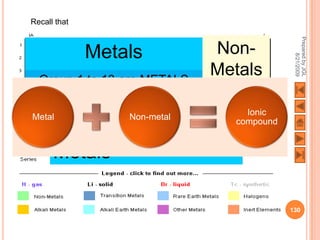
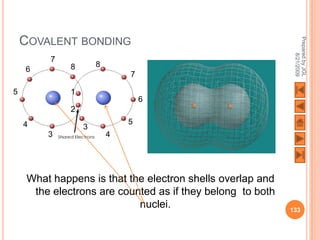
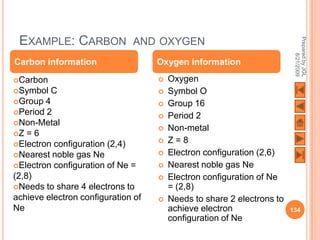
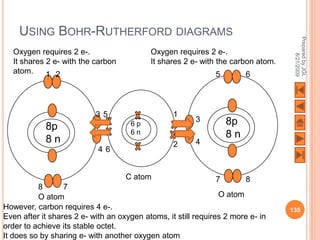
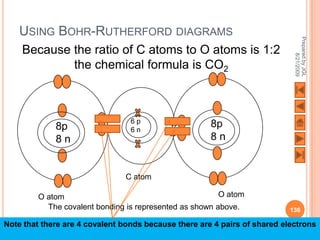
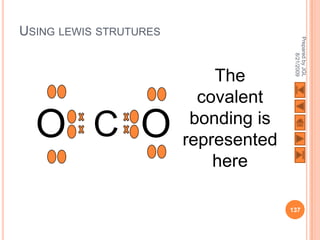
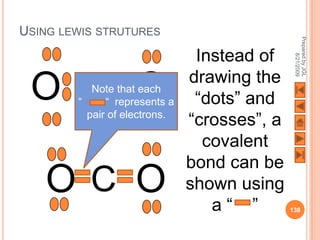
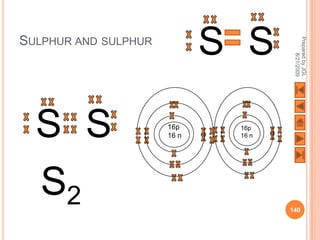
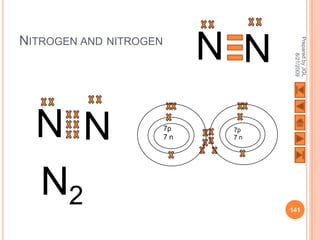
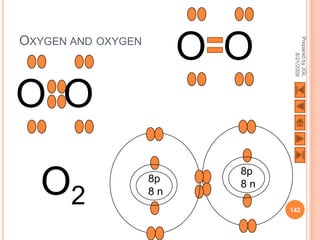
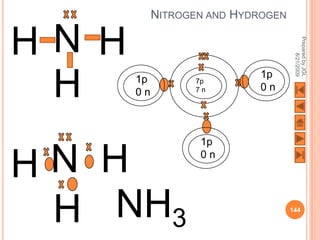
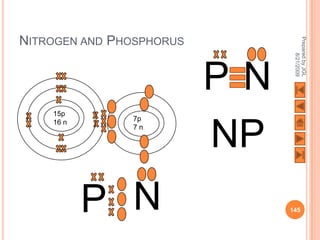
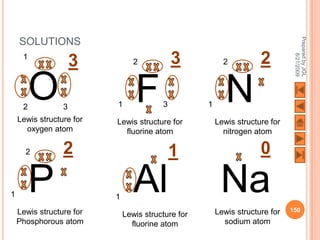
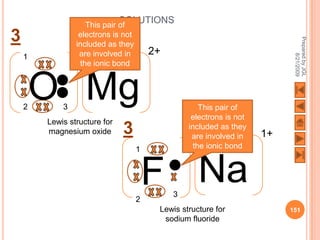
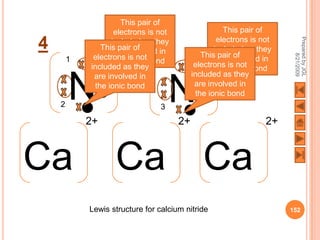
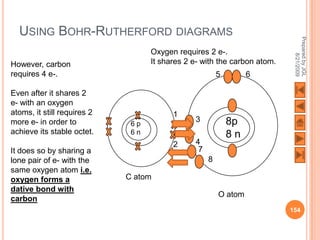
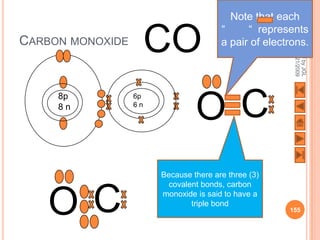
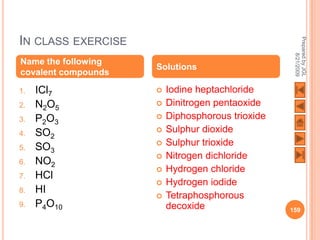
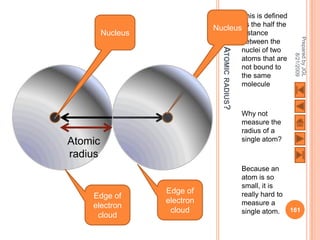
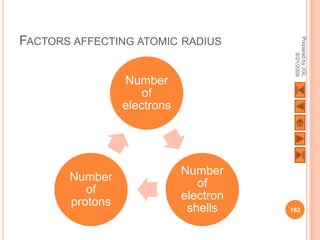
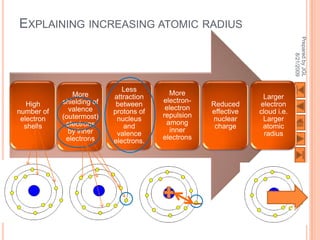
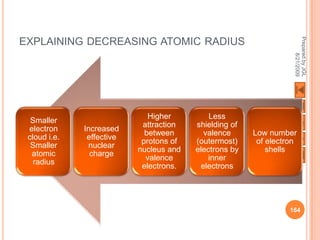
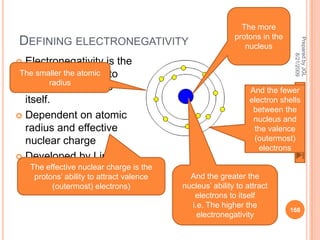
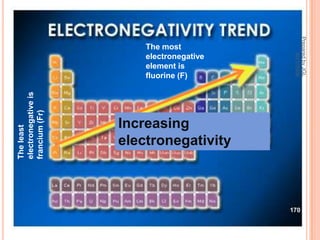
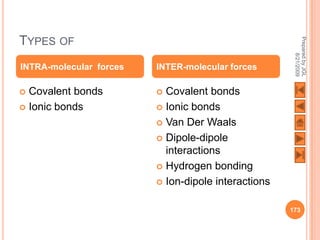
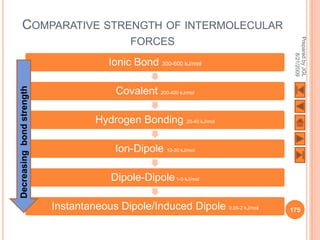
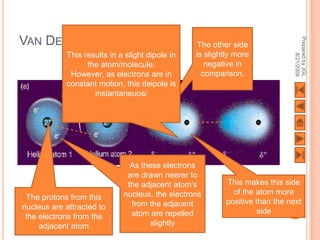
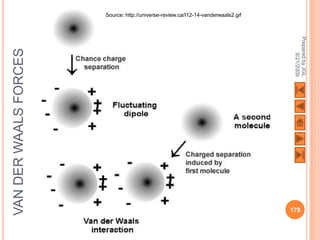
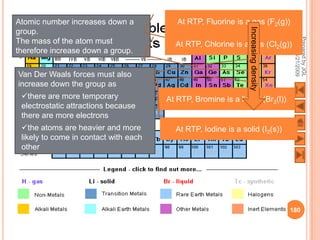
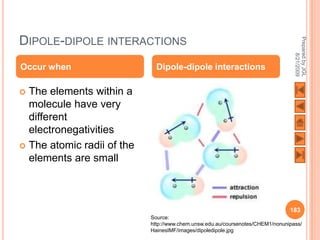
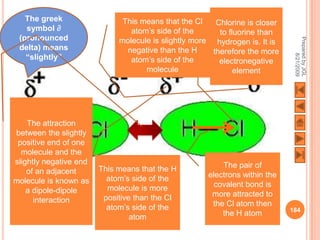
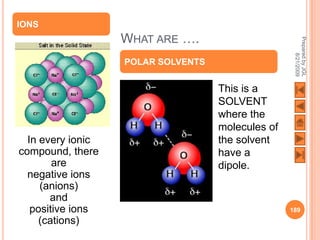
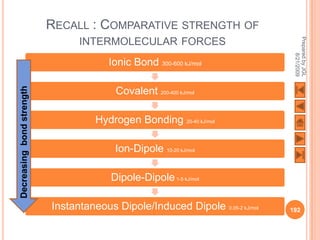
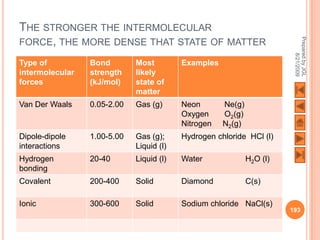
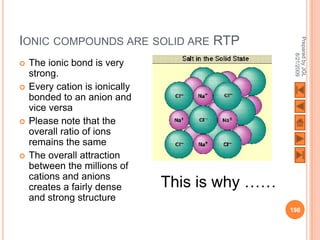
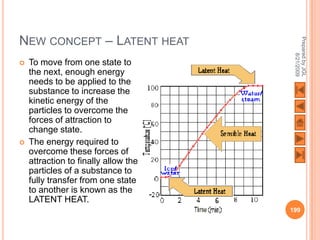
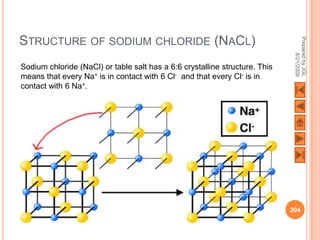
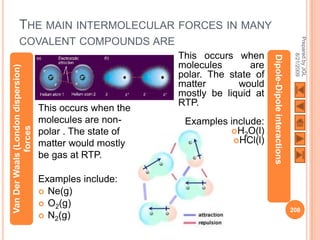
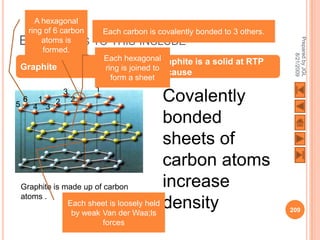
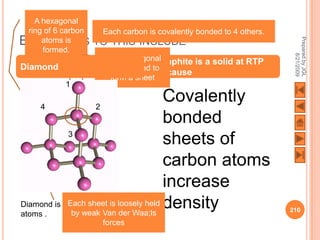
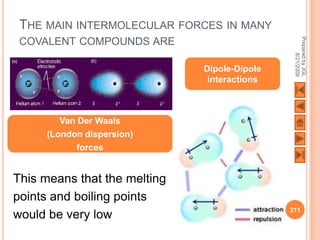
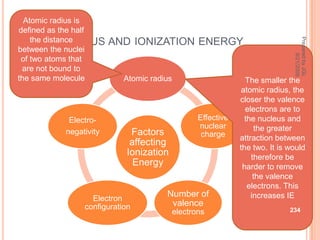
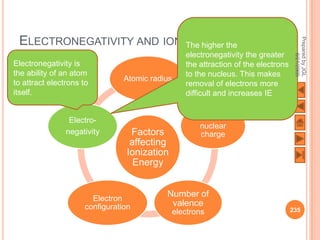
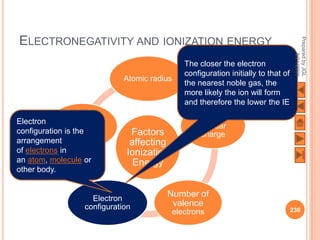
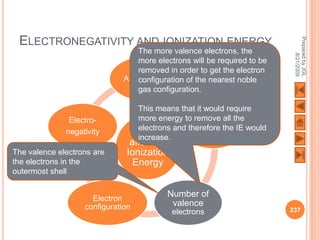
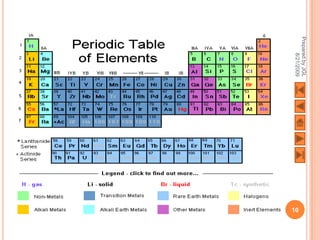
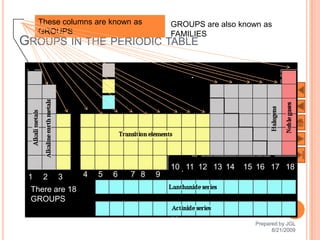
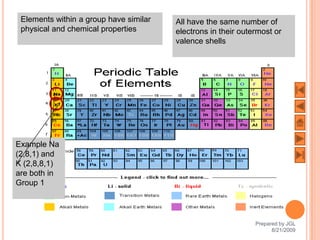
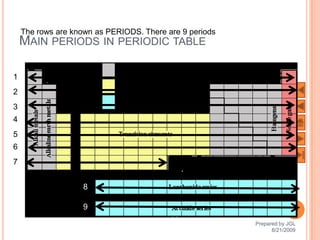
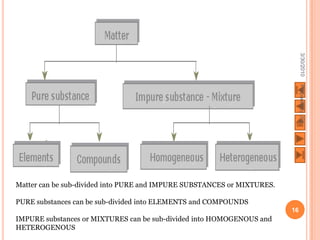
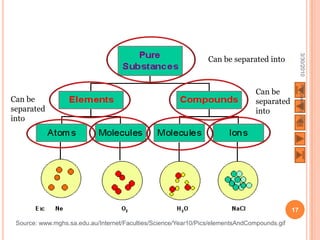
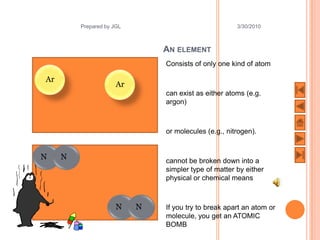
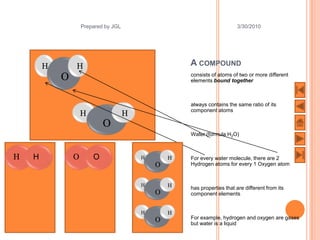
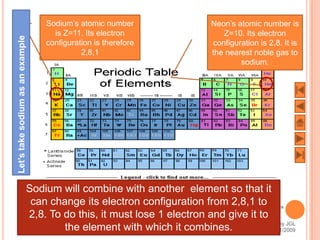
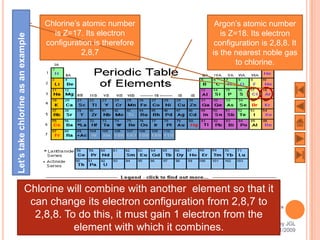
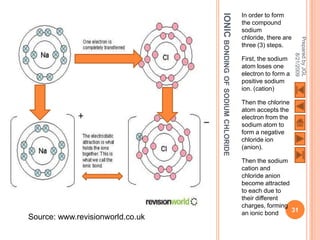
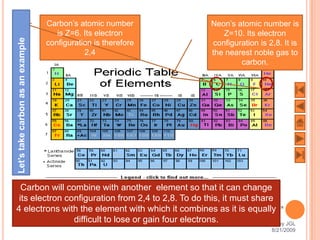
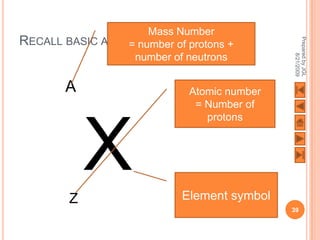
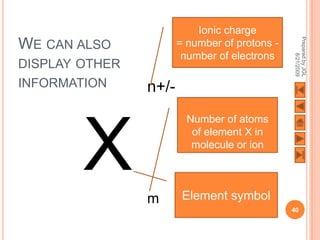
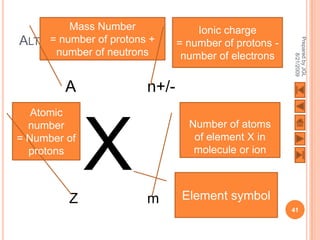
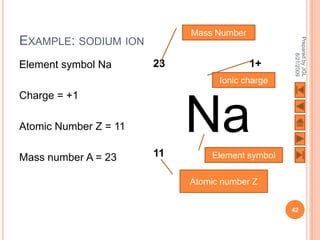
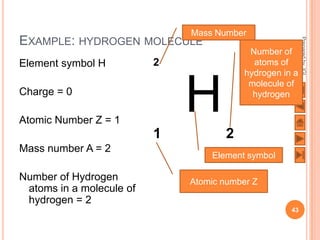
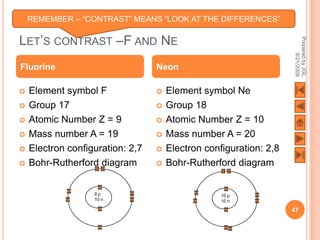
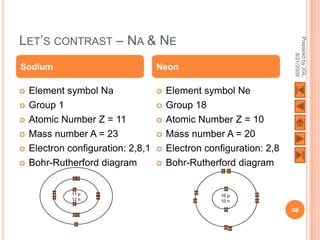
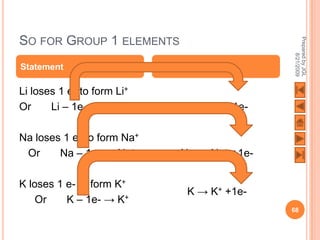
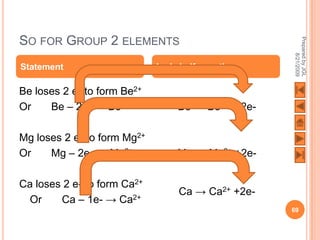
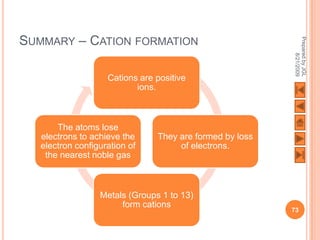
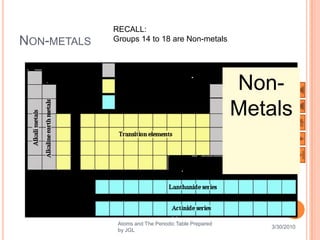
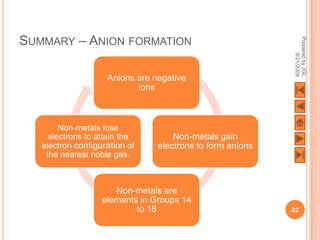
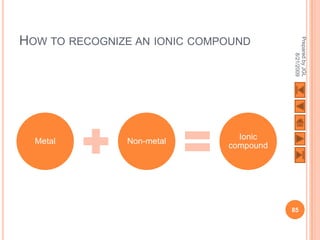
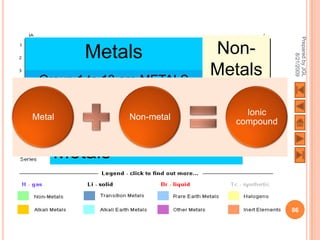
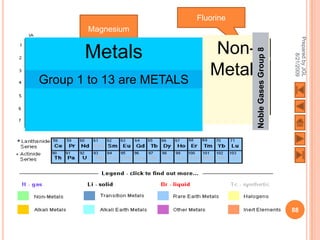
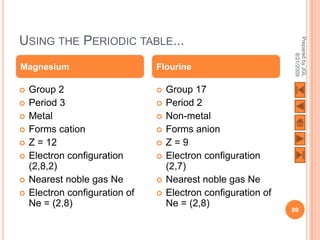
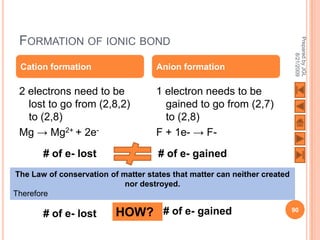
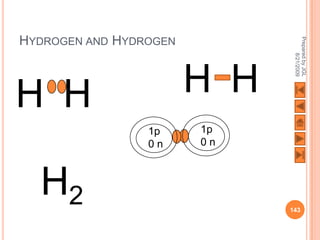The document outlines a syllabus on structure and bonding, including concepts of ionic, covalent, and metallic bonding, aimed at helping students understand the behavior of elements and compounds. It covers the formation of ions through electron gain or loss, naming conventions, and the properties associated with different types of bonding, along with the periodic table's role in understanding these phenomena. Key objectives include recognizing ion formation, explaining different types of bonding, and predicting properties based on these concepts.


















































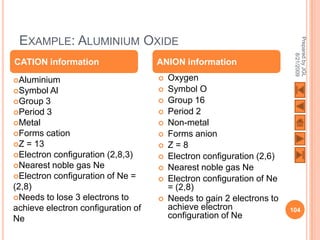



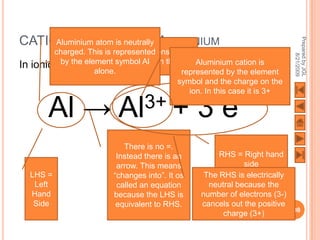
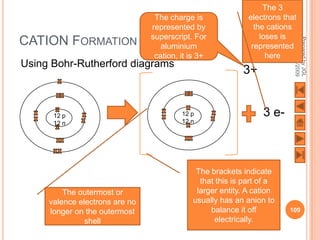


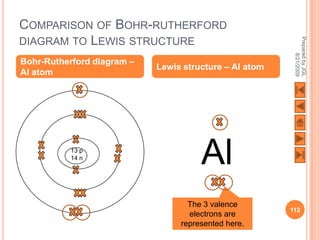
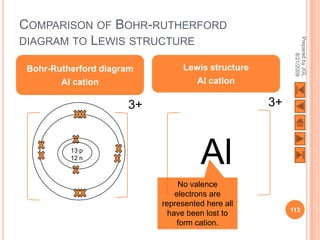
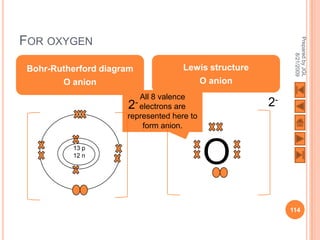



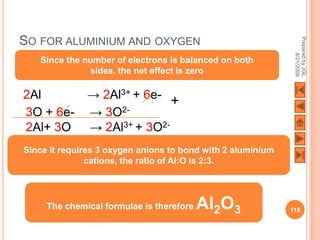
![USING LEWIS (DOT-AND-CROSS) DIAGRAMS
Prepared by JGL
2 -
8/21/2009
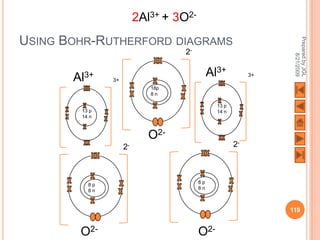
[Al] 3+ O [Al] 3+
2- 2-
O O
2Al3+ + 3O2-
120](https://image.slidesharecdn.com/bonding-100331004049-phpapp01/85/Bonding-ionic-covalent-metallic-120-320.jpg)