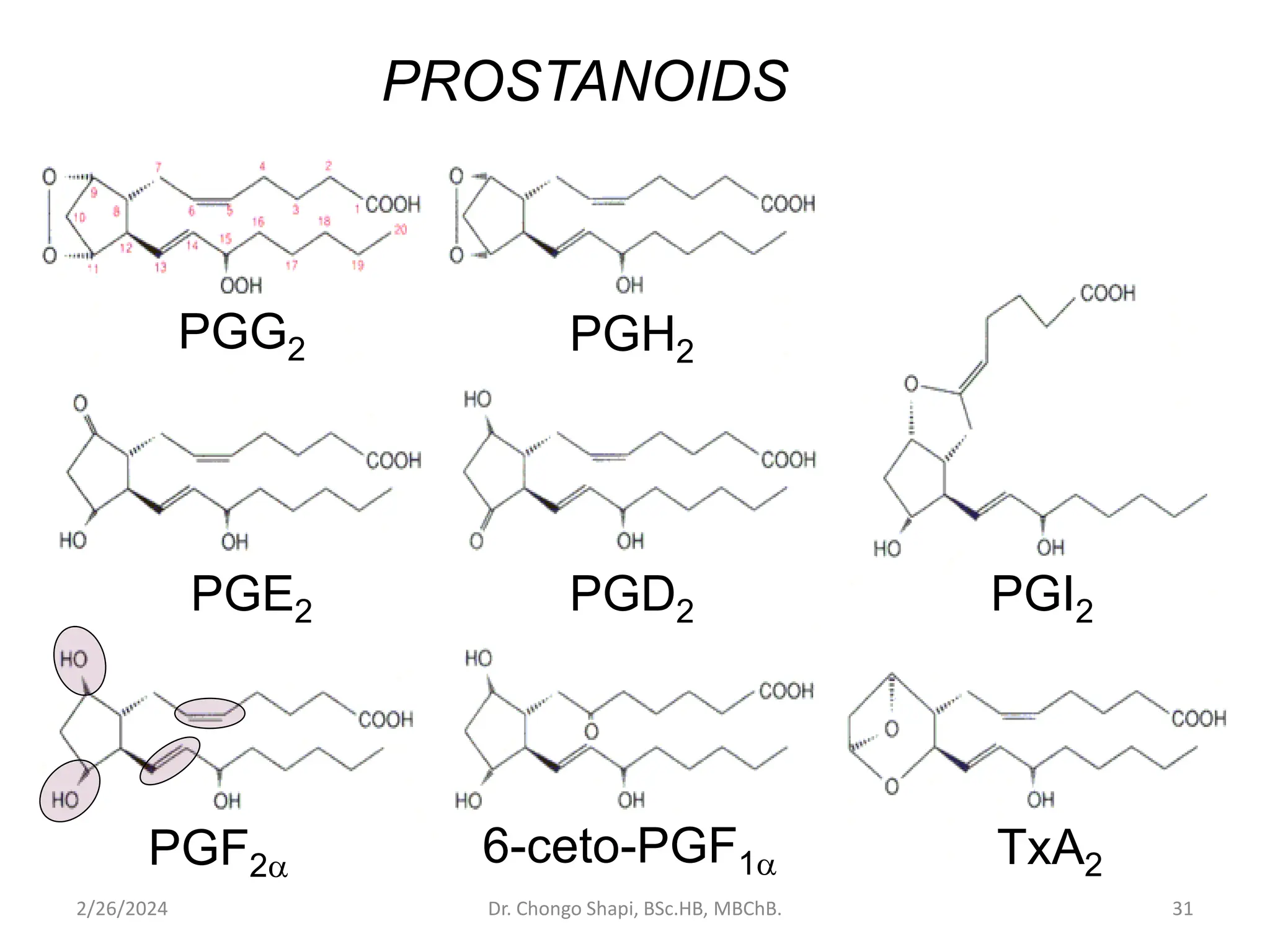
This document discusses lipids and fatty acids. It defines lipids and outlines their structural features and classification. Lipids are classified into simple lipids, compound lipids, and derived lipids. The document discusses the biomedical importance of lipids as important dietary constituents, building materials, and as carriers of fat-soluble vitamins. It also summarizes the different types of fatty acids including saturated, unsaturated, essential fatty acids, and eicosanoids derived from polyunsaturated fatty acids.