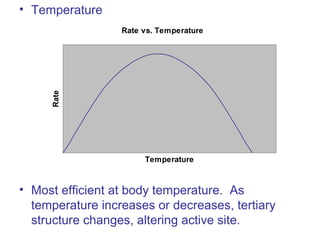
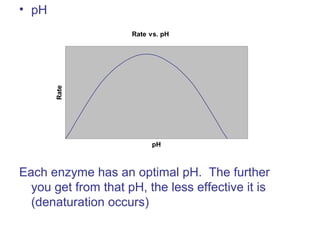
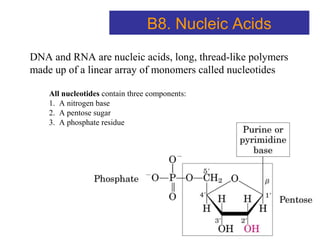
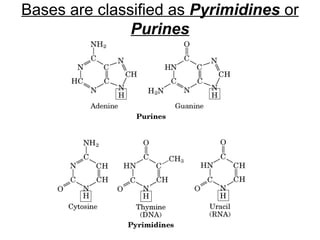
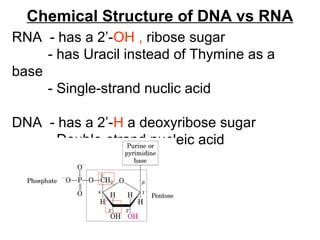
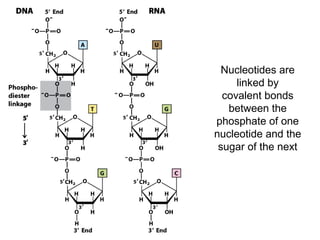
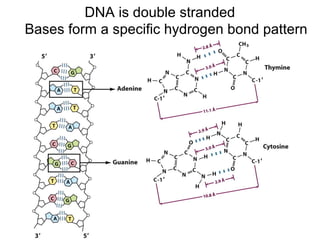
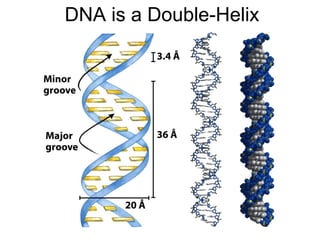
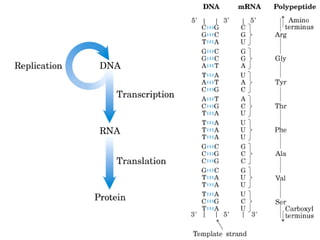
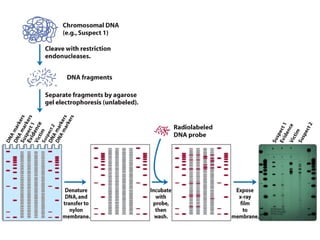
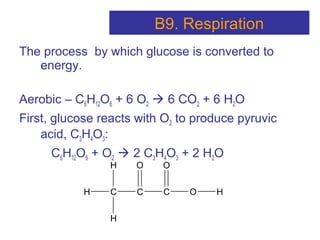
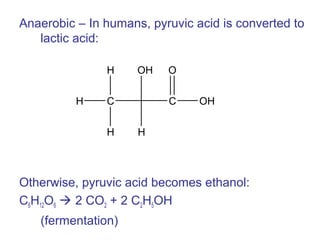
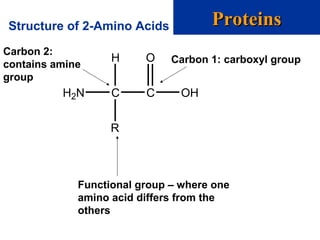
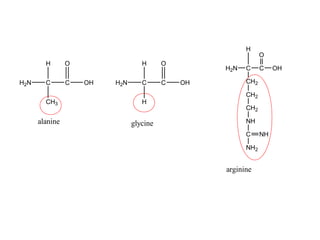
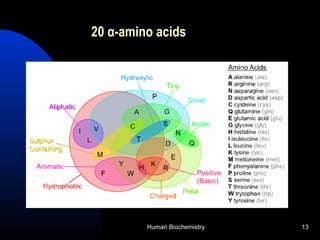
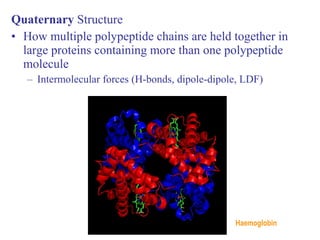
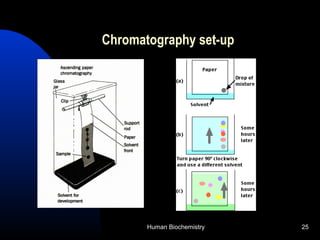
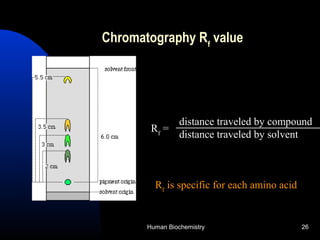
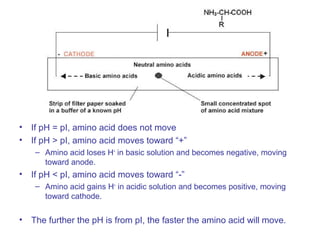
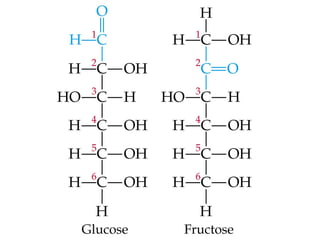
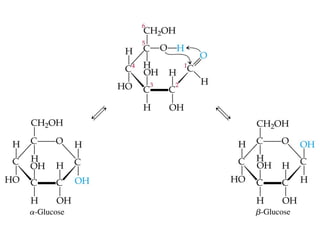
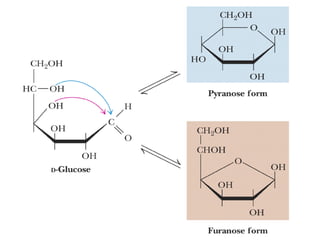
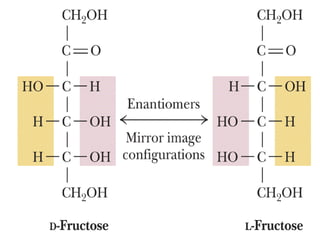
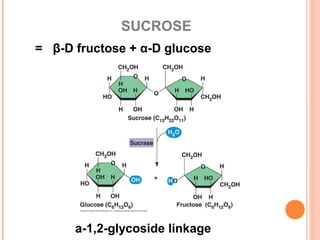
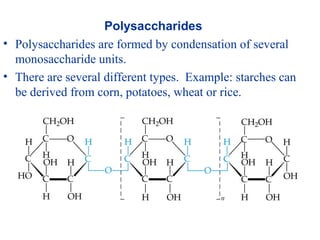
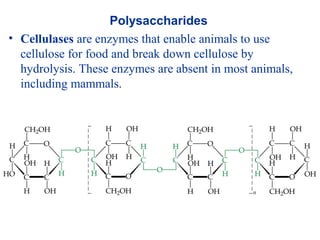
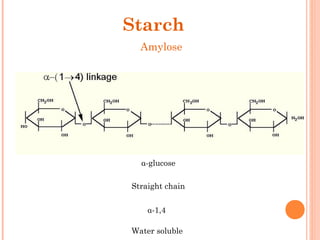
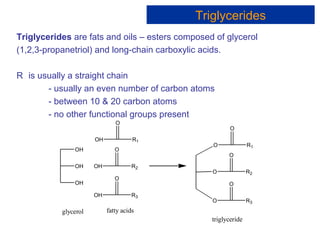
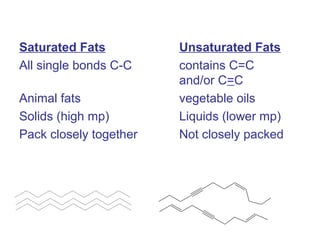
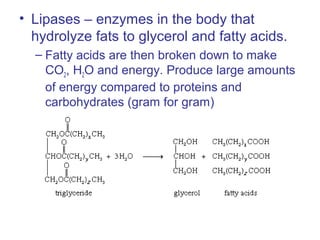
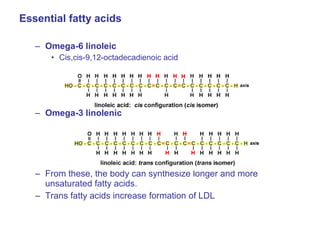
The document provides an overview of key topics in biochemistry including energy from food, proteins, carbohydrates, lipids, and nucleic acids. Specifically, it discusses how calorimetry can be used to determine the energy content of foods, the structures and functions of amino acids, proteins, carbohydrates like glucose and starch, and lipid molecules like triglycerides. It also briefly outlines analysis techniques for proteins like chromatography and electrophoresis. The document serves as an introductory guide to understanding the basic building blocks and energy sources in living organisms.










![Jan 3, 2010 Human Biochemistry 14
Properties of amino acids
(amphoteric)
H+
+ H2
N-CHR-COO-
← H3
N+
-CHR-COO-
→ H3
N+
-CHR-COOH + OH-
>At low pH
>Extra H+
reacts with OH-
>[OH-
] drops
>Equilibrium shifts to the right
>H3
N+
-CHR-COOH form
>positive charge
>At high pH
>Extra OH-
reacts with H+
>[H+
] drops
>Equilibrium shifts to the left
>H2
N-CHR-COO-
form
>negative charge
>At isoelectric point
>identical ionizations
>only zwitterion
>H3
N+
-CHR-COO-
form
>no net charge](https://image.slidesharecdn.com/biochemistry97-150303012307-conversion-gate01/85/Biochemistry-97-14-320.jpg)
![Jan 3, 2010 Human Biochemistry 15
Properties of amino acids
(buffer)
H3
N+
-CHR-COO-
+ H+
→ H3
N+
-CHR-COOH + H2
O>when H+
is added
>equilibrium shifts to right
>[H+
] drops
>pH remains the same
>buffer action
>when OH-
is added
>equilibrium shifts to left
>[OH-
] drops
>pH remains the same
>buffer action
H2
O + H2
N-CHR-COO-
← OH-
+ H3
N+
-CHR-COO-](https://image.slidesharecdn.com/biochemistry97-150303012307-conversion-gate01/85/Biochemistry-97-15-320.jpg)


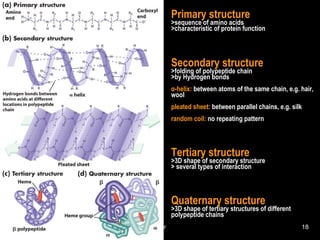
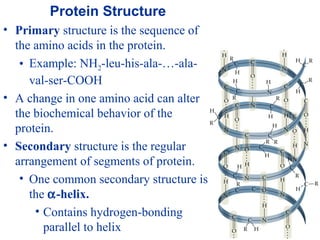

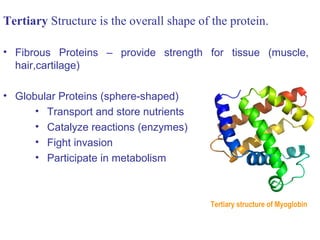
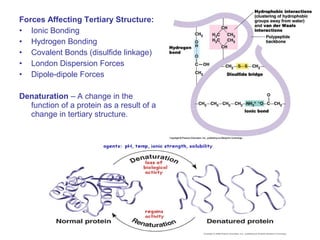








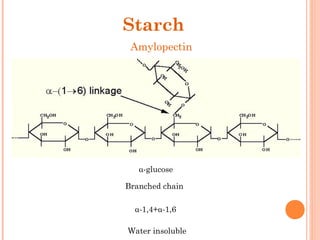
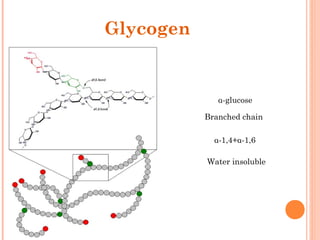
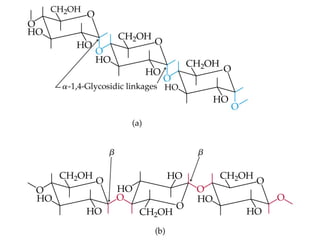
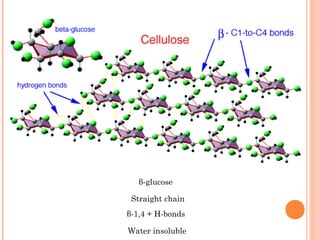


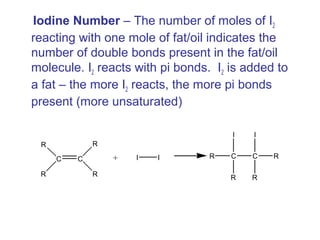

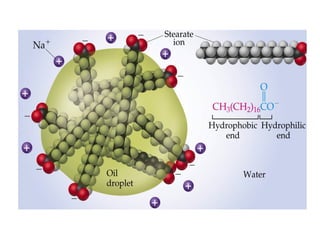


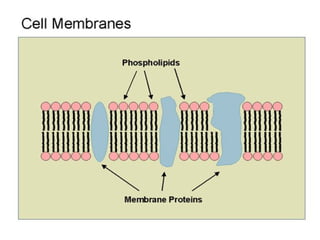







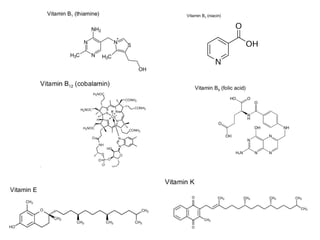










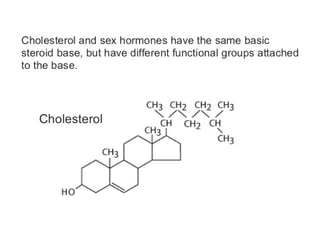

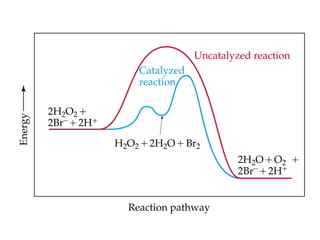
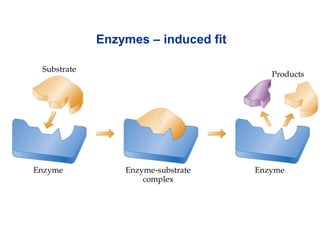
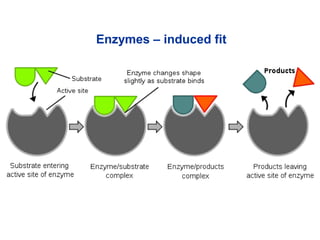
![• Cofactor – A substance that attaches to an
enzyme in order to increase its effectiveness.
– Coenzyme – an organic cofactor.
• Factors that affect the rate of a catalyzed
reaction
– [enzyme] – first order Rate = k[E]
• As concentration of enzyme increases, more substrate
molecules can be catalyzed
Rate vs. [Enzyme]
[Enzyme]
RateofReaction](https://image.slidesharecdn.com/biochemistry97-150303012307-conversion-gate01/85/Biochemistry-97-88-320.jpg)
![• [Substrate]
• As concentration increases, rate increases
(first order kinetics) up to a point – enzyme
saturation. When all enzyme molecules are
working at capacity, rate levels off.
Rate vs. [Substrate]
[Substrate]
RateofReaction](https://image.slidesharecdn.com/biochemistry97-150303012307-conversion-gate01/85/Biochemistry-97-89-320.jpg)
![Rate vs. [Substrate]
[Substrate]
RateofReaction
Vmax
Vmax = The maximum rate of a reaction for a particular
enzyme concentration.
Km = Michaelis-Menten constant
Represents approximate [substrate] in human
body under normal conditions
Equal to [substrate] at ½ Vmax
Km](https://image.slidesharecdn.com/biochemistry97-150303012307-conversion-gate01/85/Biochemistry-97-90-320.jpg)

![2. Non-competitive inhibition – Inhibitor binds to
the enzyme at a site other than the active site.
This causes the shape of the active site to
change, so that the enzyme will no longer fit
into the active site properly. (heavy metal ions)
Rate vs. [substrate] with Inhibition
[Substrate]
Rate
Uninhibited Competitive Non-competitive](https://image.slidesharecdn.com/biochemistry97-150303012307-conversion-gate01/85/Biochemistry-97-92-320.jpg)