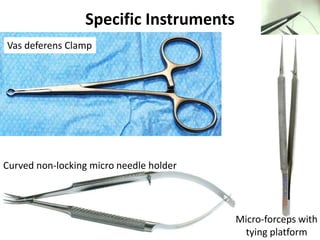
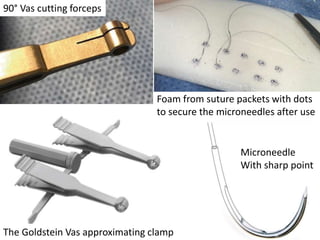
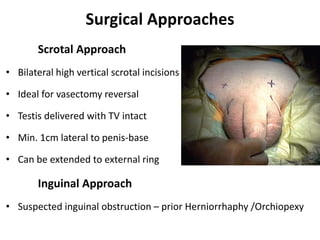
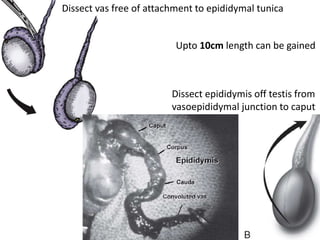
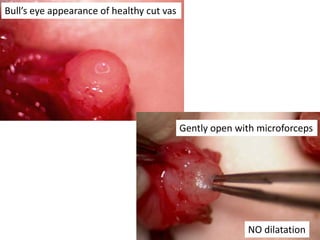
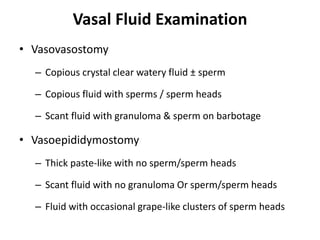
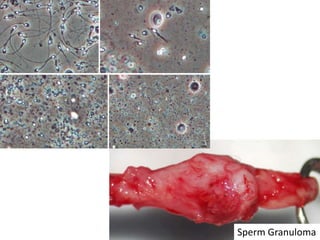
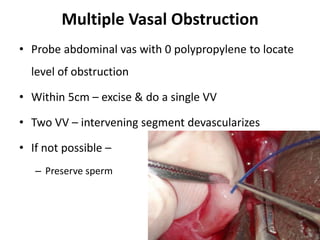
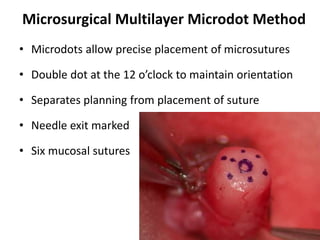
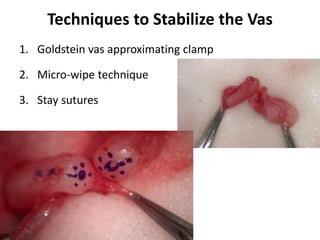
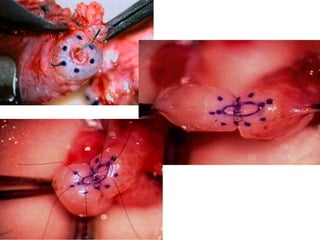
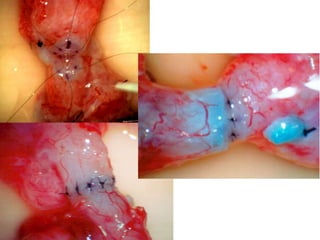
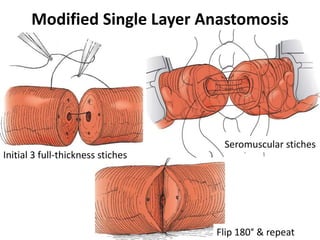
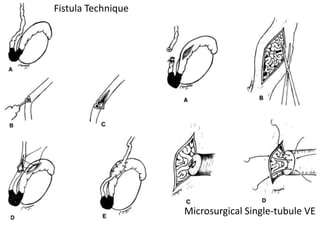
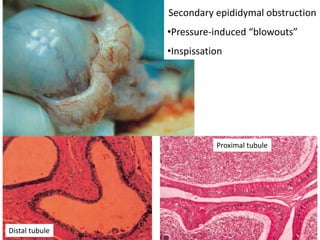
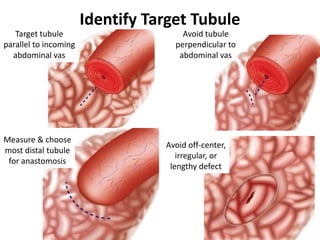
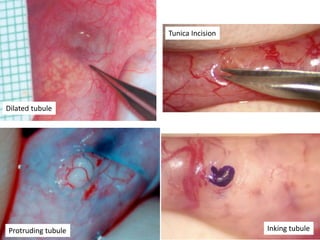
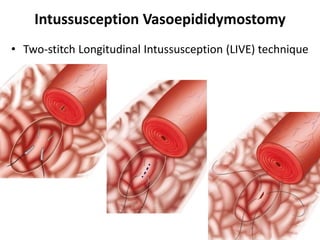
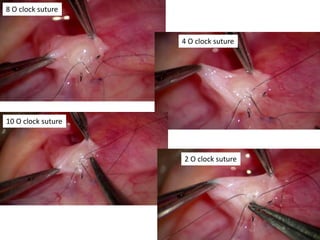
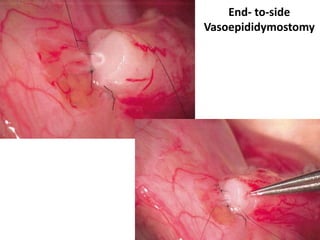
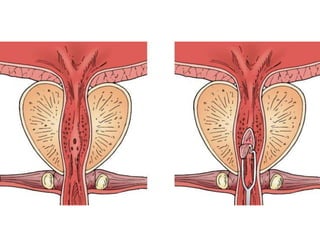
This document provides an overview of surgical techniques for treating obstructive azoospermia, including vasovasostomy (VV), vasoepididymostomy (VE), and transurethral resection of the ejaculatory ducts (TURED). It discusses the history, indications, preparation, approaches, techniques, principles, and outcomes of VV and VE. VV is the standard treatment for vasectomy reversal while VE is indicated when there is no sperm in the vas deferens. Both require microsurgical skills for reconnecting the vas deferens or epididymal tubule with success rates over 70% for VV and 50% for VE. TURED may be considered for e