The document provides an overview of Earth's atmosphere including:
- Its composition of nitrogen (78%), oxygen (21%), and trace amounts of other gases.
- It describes the layers of the atmosphere from lowest to highest - troposphere, stratosphere, mesosphere, thermosphere, and exosphere.
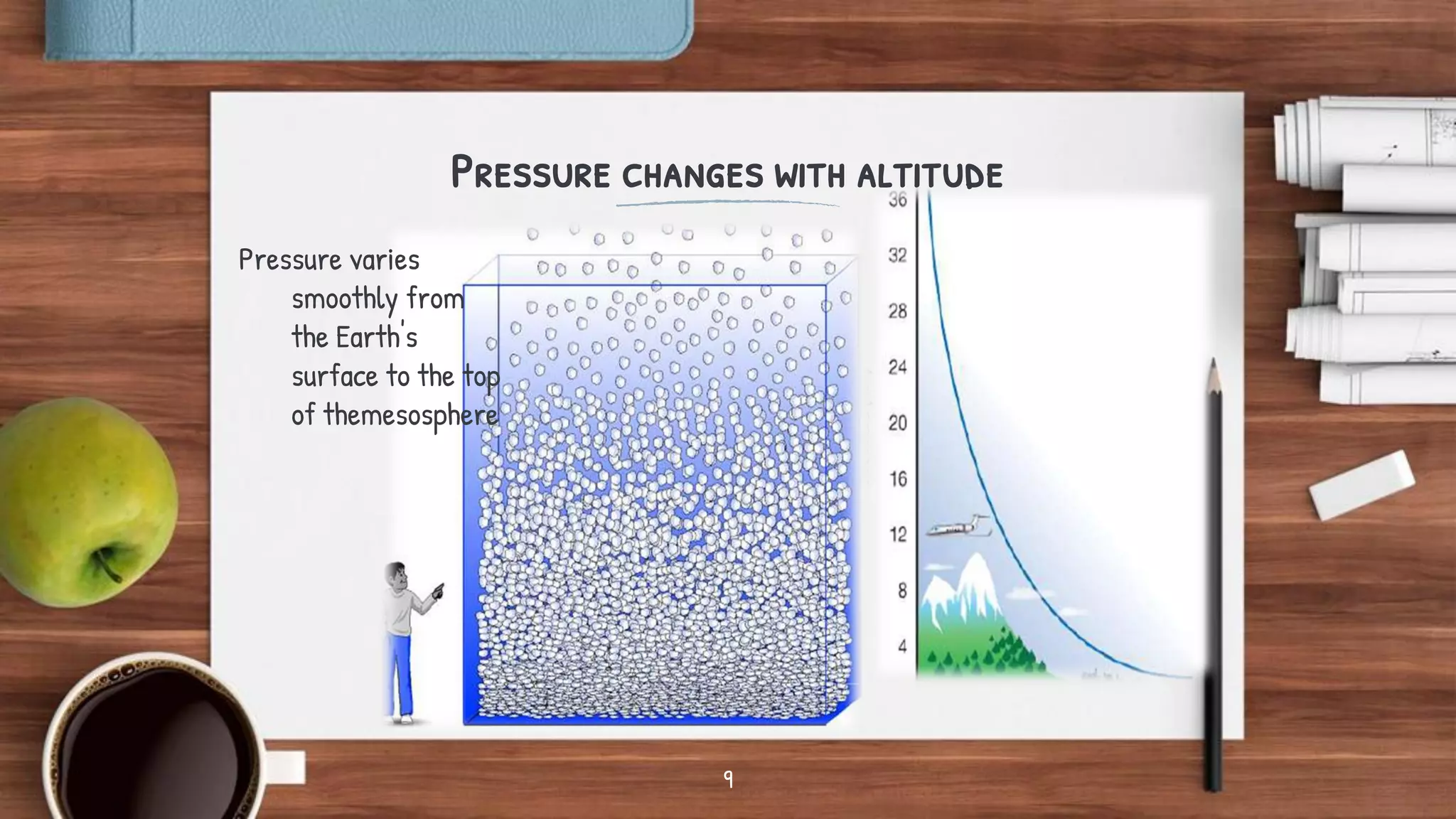
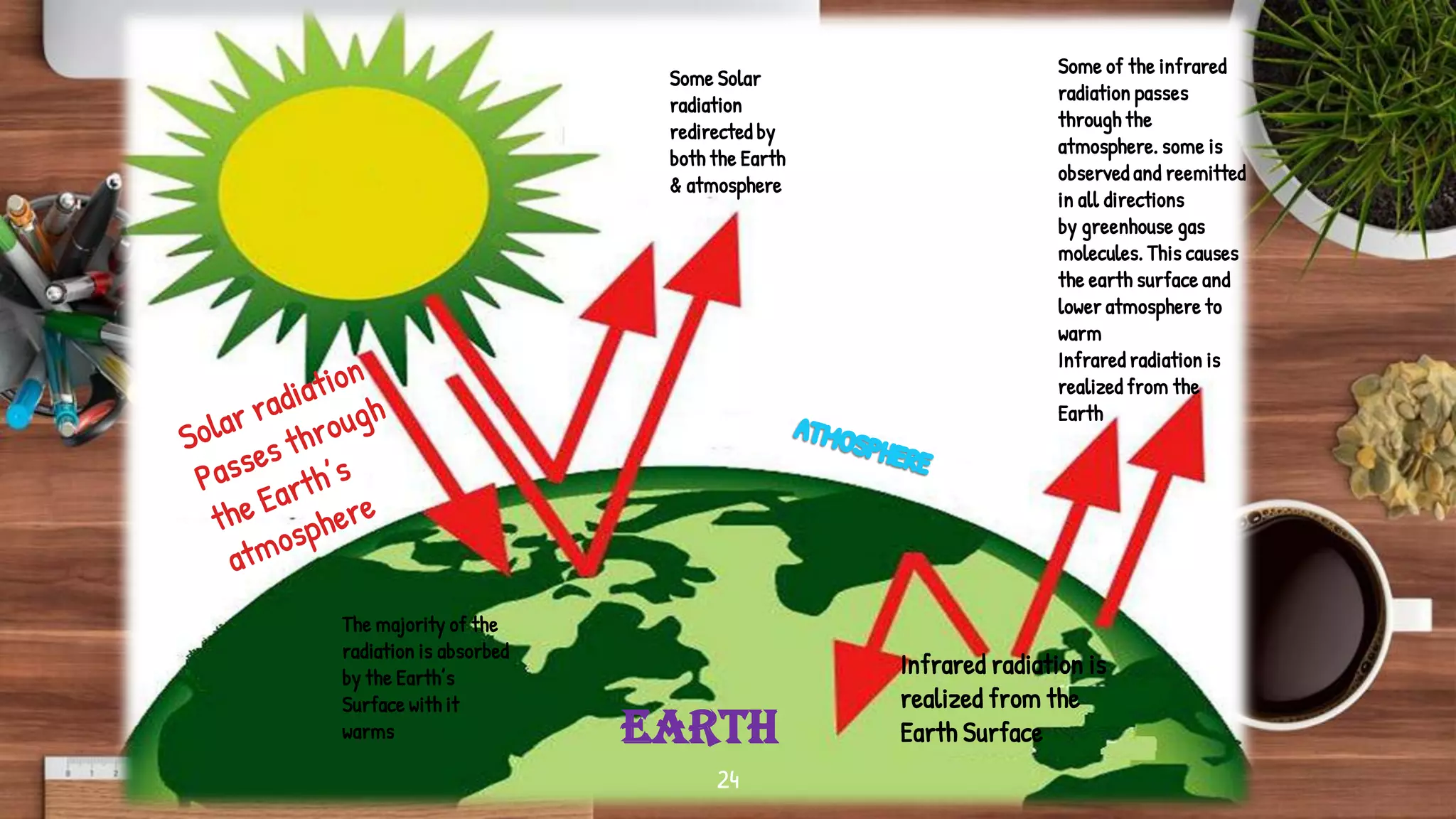
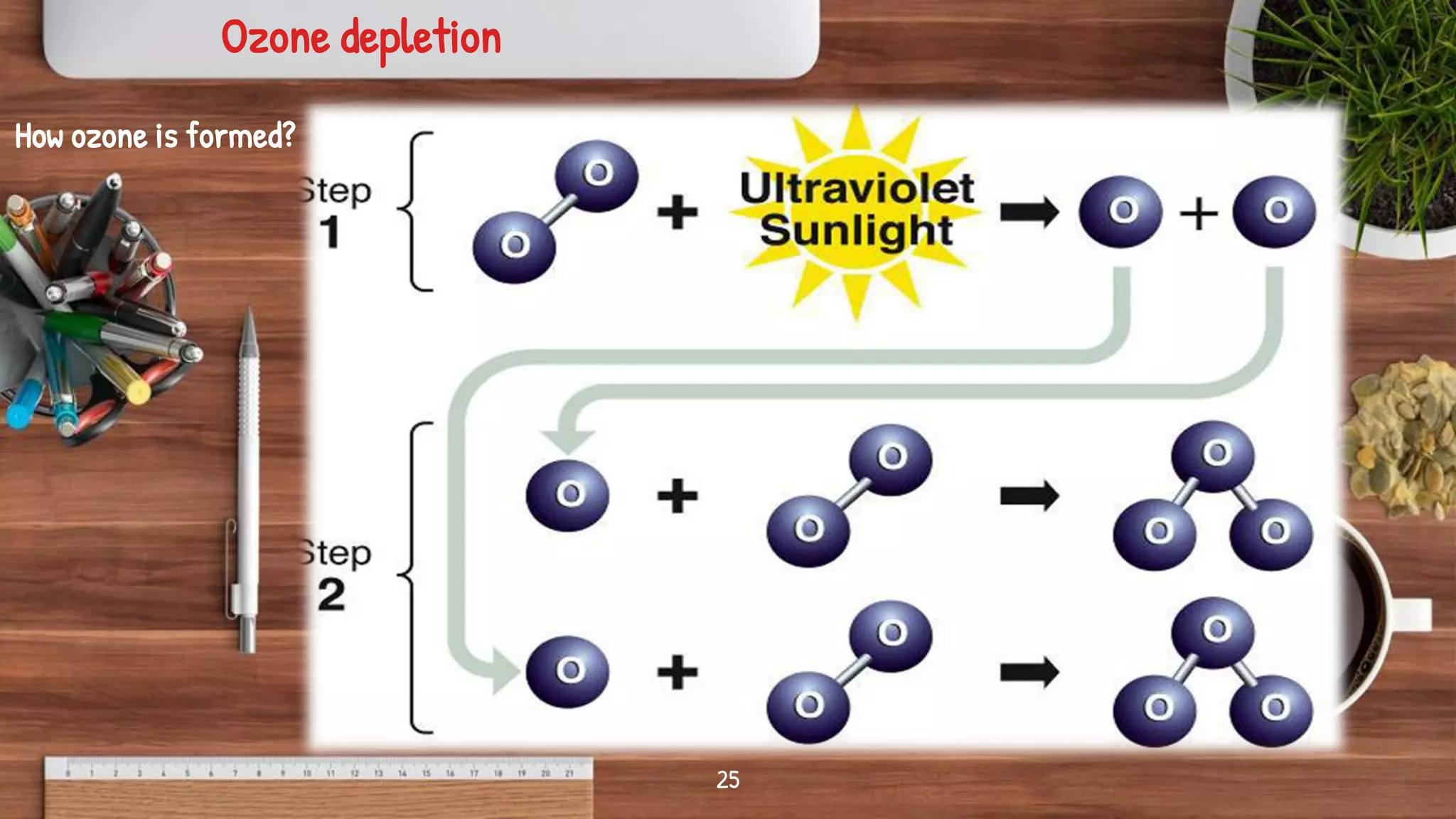
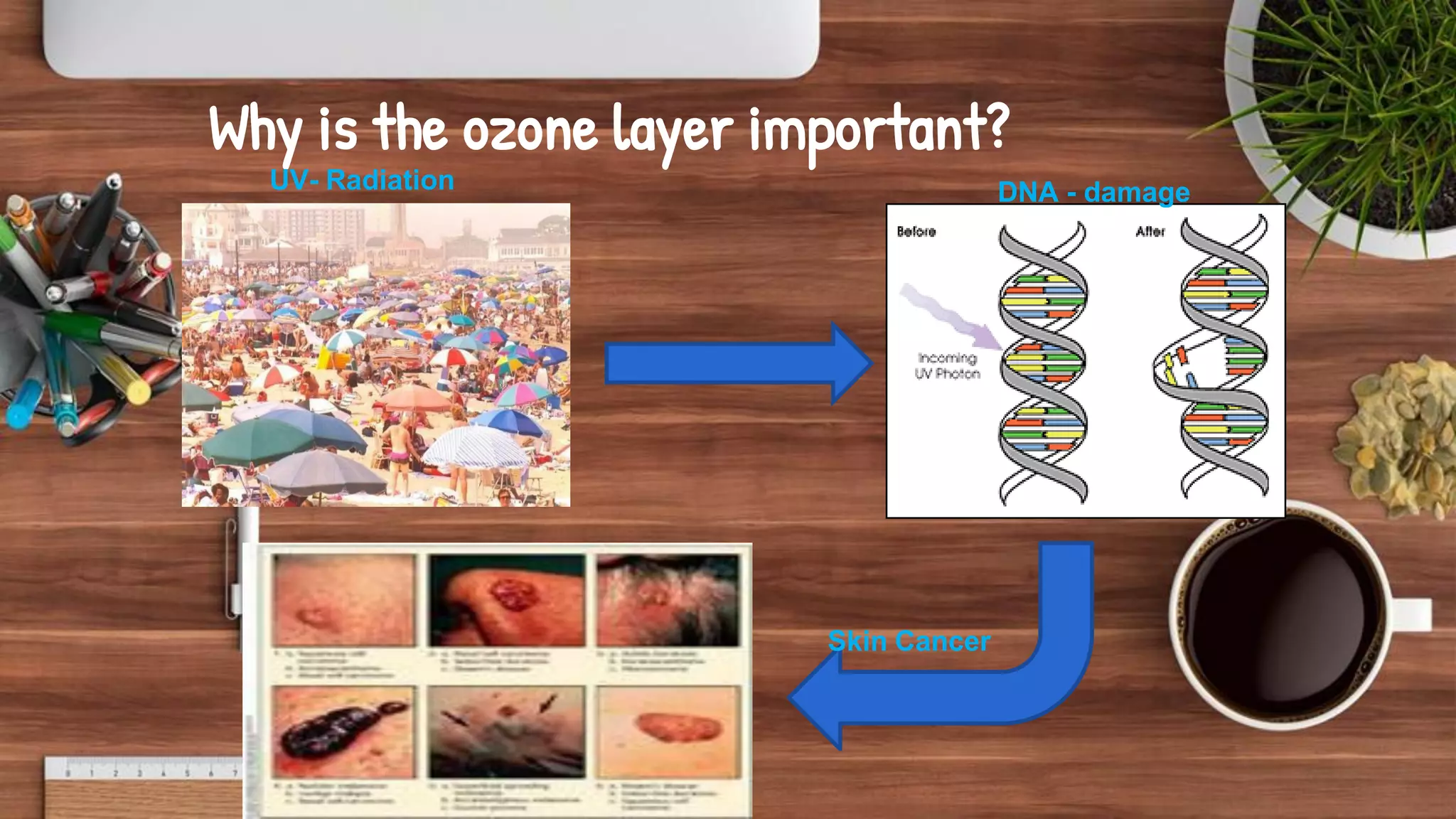
- Key topics covered include the nitrogen cycle, how pressure decreases with altitude, measuring pressure, atmospheric chemistry, and issues like acid rain, the greenhouse effect, and ozone depletion.