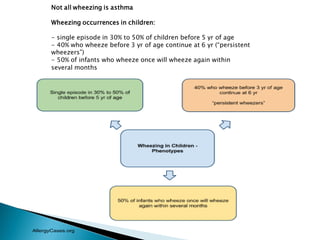
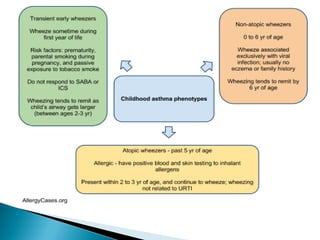
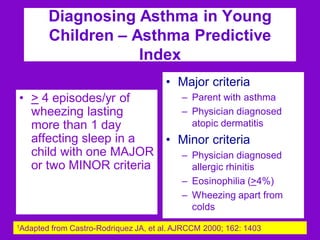
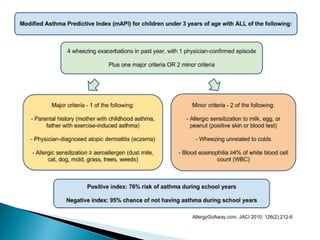
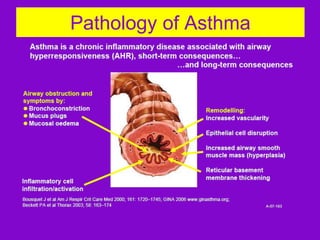
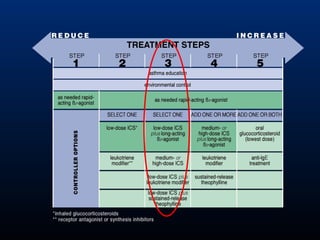
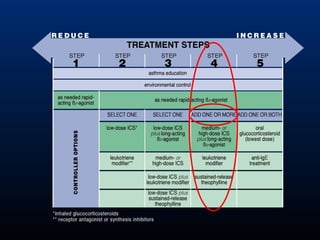
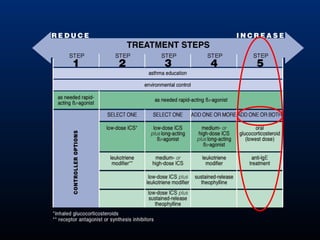
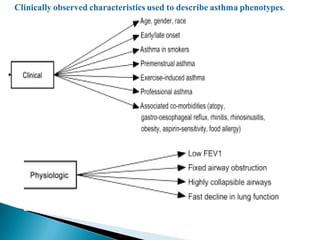
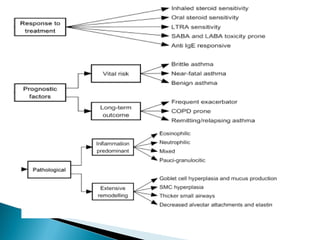
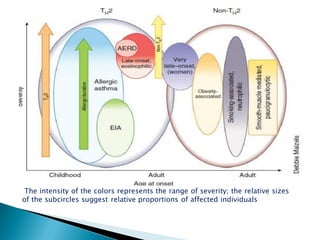
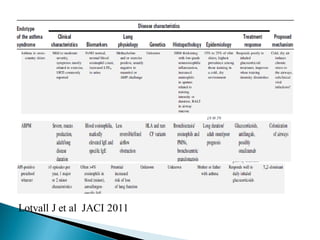
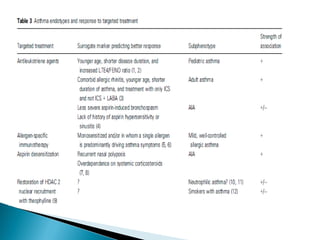
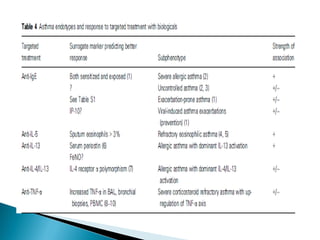
This document discusses various asthma phenotypes or endotypes that have been identified based on differences in clinical characteristics, biomarkers and treatment responses. The two main endotypes discussed are TH2-high asthma and non-TH2 asthma. TH2-high asthma includes early-onset allergic asthma, late-onset eosinophilic asthma and exercise-induced asthma. It is characterized by eosinophilia, TH2 biomarkers and good response to corticosteroids and anti-TH2 targeted therapies. Non-TH2 asthma includes obesity-related asthma, neutrophilic asthma and smoking asthma. It has fewer clinical allergies and TH2 biomarkers, and poorer responses to corticosteroids. Distinct clinical features, genetics,



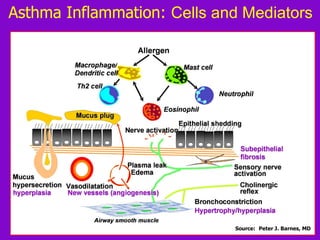
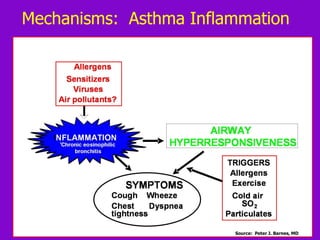
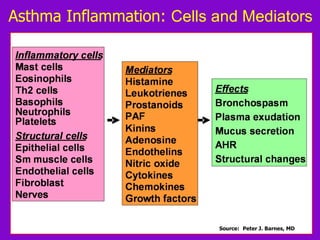
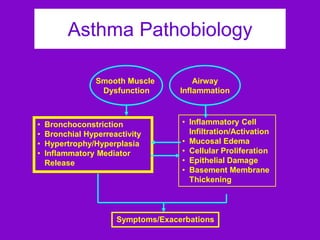












































![ACOS
The mixed COPD–asthma phenotype was
defined as an airflow obstruction that is not
completely reversible accompanied by
symptoms or signs of an increased
reversibility of the obstruction.[7] In other
guidelines, these patients are described as
'patients with COPD and prominent asthmatic
component' or as asthma that complicates
COPD.](https://image.slidesharecdn.com/asthmaupdates-131208173205-phpapp01/85/Asthma-updates-66-320.jpg)