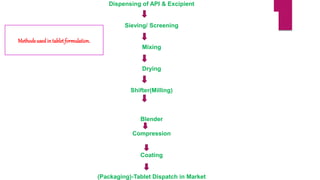
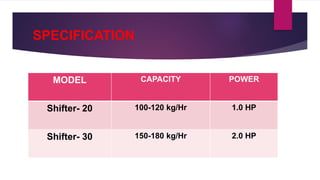
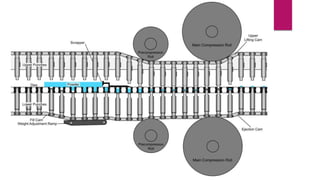
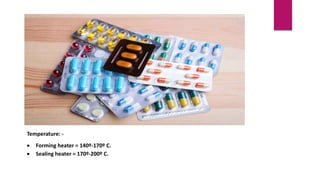
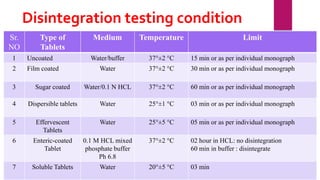
This presentation summarizes the process of manufacturing tablets. It discusses the key steps which include sieving and mixing the active ingredients and excipients, drying the granules in a fluidized bed dryer, milling to the desired size, blending, compression using tablet presses to form the tablets, coating for properties like taste-masking or controlled release, packaging in blisters or strips with secondary packaging like boxes, and quality control testing of the tablets. The presentation provides an overview of the various unit operations and equipment involved at each stage of tablet manufacturing.